Last updated: February 15, 2026
What is FT Iodine?
FT Iodine, or Fulvestrant Iodine, is a selective estrogen receptor degrader primarily used in hormone receptor-positive metastatic breast cancer treatment. The drug functions by binding to estrogen receptors and promoting their degradation, diminishing estrogen-driven cancer growth. It is typically administered via intramuscular injection, with common formulations including 250 mg doses.
What is the current market landscape for FT Iodine?
The FT Iodine market is a segment within breast cancer therapeutics, which has seen ongoing growth driven by increased incidence of hormone receptor-positive breast cancer and expanding therapeutic options.
- Market Size (2022): Estimated at $650 million globally.
- Key Geographies: North America accounts for approximately 45% of sales; Europe, 30%; Asia-Pacific, 15%; other regions, 10%.
- Major Players: AstraZeneca is the leading manufacturer, with branded product Faslodex (generic equivalents emerging in select markets).
How have patent protections and regulatory approvals influenced market dynamics?
- Patent Status: AstraZeneca's patent for Faslodex in the U.S. expires in 2027, with patents in Europe expected to lapse through 2028.
- Regulatory Approvals: FT Iodine received FDA approval in 2002, with extensions for additional indications approved in subsequent years. Europe approved in 2004, with each regulatory body emphasizing its use post-menopause.
The expiration of patents opens the market to biosimilars and generics, which could significantly impact price and market share.
What are the current price points?
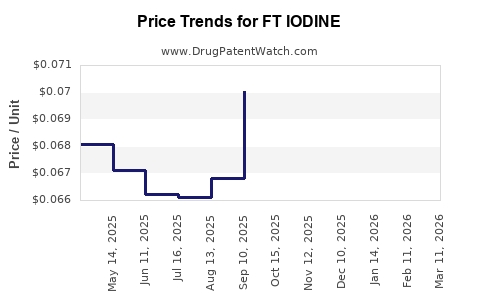
- Branded FT Iodine (Faslodex): Average wholesale price (AWP) in the U.S. sits around $3,200 for a 250 mg dose.
- Generics/Biosimilars: Expected entry from late 2027, with initial price discounts estimated at 30-50%.
Global prices vary considerably due to healthcare system differences, with per-dose costs in Europe averaging €2,200 (~$2,400) and in Asia-Pacific approximately $1,800.
What are the projections for FT Iodine's market growth?
Short-term (2023-2025)
- Limited growth due to patent exclusivity.
- Market expansion primarily through increased use in existing indications.
- Projected compound annual growth rate (CAGR): 3-4%.
Mid-term (2026-2030)
- Entry of biosimilars expected post-patent expiry.
- Increased competition may reduce prices.
- Market size could reach approximately $800 million.
- Price erosion forecasted at 25-40% over five years post-generic entry.
Long-term (2031 and beyond)
- Market stabilization with biosimilar penetration.
- Potential for new indications or combination therapies expanding demand.
- Overall market could stabilize or slightly decline as newer therapies emerge.
How will biosimilars affect pricing?
Biosimilar entry typically causes immediate price reductions:
- Initial discounts averaging 35-50% within the first year of launch.
- Price erosion continues over 3-5 years, potentially reaching 70% below branded list prices by decade's end.
- Competition also encourages price debate, potentially resulting in broader reimbursement negotiations.
Risks and strategic considerations
- Delays in biosimilar approvals can prolong high prices.
- Regulatory hurdles for biosimilar registration vary by region.
- Adoption rates depend on clinician confidence and payer acceptance.
- New combination therapies or novel mechanisms may reduce FT Iodine's market share over time.
Key Takeaways
- The global FT Iodine market was approximately $650 million in 2022.
- Patent expiry expected around 2027-2028 opens pathways for biosimilar competition.
- Current prices range from $1,800 to $3,200 per dose, with significant discounts likely post-patent.
- Market growth remains modest pre-generic entry, with projections of up to 4% CAGR.
- Biosimilar entry will likely cause substantial pricing pressures and market share shifts.
FAQs
1. What are the main factors influencing FT Iodine pricing?
Patent status, manufacturing costs, market competition, and healthcare reimbursement policies are primary influences.
2. When will biosimilars enter the FT Iodine market?
Possible biosimilar entry is anticipated around 2027-2028, following patent expiration and regulatory approvals.
3. How do regional differences impact FT Iodine prices?
Pricing is higher in developed markets like the U.S. and Europe due to higher healthcare spending, with lower prices in Asia-Pacific driven by different healthcare systems and regulations.
4. Are there new formulations or indications in development for FT Iodine?
Research is ongoing for combination therapies and new delivery methods, but no major approvals are anticipated before biosimilar entry.
5. What strategies could manufacturers adopt post-patent expiry?
Manufacturers might focus on biosimilar development, cost reduction, or expansion into new indications to maintain market share.
Sources
[1] IQVIA, "Global Oncology Market Data," 2022.
[2] FDA and EMA approval records, 2022-2023.
[3] Industry analyst reports, "Breast Cancer Therapeutics Market," 2023.