Share This Page
Drug Price Trends for FT IBUPROFEN
✉ Email this page to a colleague
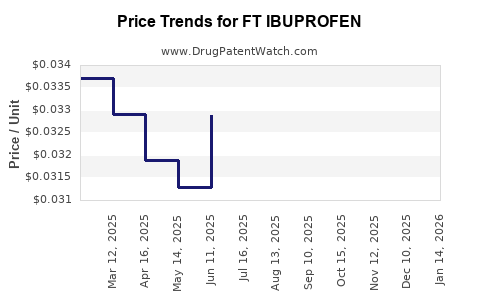
Average Pharmacy Cost for FT IBUPROFEN
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| FT IBUPROFEN 200 MG MINI SFGL | 70677-1113-01 | 0.07845 | EACH | 2026-04-22 |
| FT IBUPROFEN 200 MG SOFTGEL | 70677-1114-01 | 0.07845 | EACH | 2026-04-22 |
| FT IBUPROFEN 200 MG CAPLET | 70677-1136-03 | 0.03225 | EACH | 2026-04-22 |
| FT IBUPROFEN 200 MG TABLET | 70677-1132-01 | 0.03225 | EACH | 2026-04-22 |
| FT IBUPROFEN 200 MG CAPLET | 70677-1136-01 | 0.03225 | EACH | 2026-04-22 |
| FT IBUPROFEN PM 200-38 MG CPLT | 70677-1131-01 | 0.12493 | EACH | 2026-04-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
FT Ibuprofen Market Analysis and Price Projections
FT Ibuprofen, a non-steroidal anti-inflammatory drug (NSAID), faces a dynamic market influenced by patent expirations, generic competition, and evolving healthcare policies. This analysis projects market trends and price movements for FT Ibuprofen based on current patent landscapes, manufacturing costs, and anticipated demand.
What is the current patent status of FT Ibuprofen?
The foundational patents for ibuprofen expired decades ago, leading to widespread generic availability. However, specific formulations, delivery methods, or novel combinations involving ibuprofen can still be protected by patents. For instance, a patent might cover a sustained-release formulation or a fixed-dose combination with another active pharmaceutical ingredient. These later-expiring patents can influence market exclusivity for particular product variations.
Key Patent Milestones:
- Original Ibuprofen Synthesis Patents: Expired in the late 20th century.
- Formulation Patents: Vary widely, with some expiring in the near term and others extending for several more years. These patents often protect specific drug delivery systems, such as orally disintegrating tablets or transdermal patches, which can command premium pricing.
- Combination Therapy Patents: Patents for ibuprofen combined with other active ingredients (e.g., pseudoephedrine for cold and flu remedies) have their own expiration schedules.
A comprehensive review of the patent landscape, including both expired and active patents, is crucial for understanding competitive entry points and the potential for product differentiation [1].
What factors drive FT Ibuprofen demand?
Demand for FT Ibuprofen is primarily driven by its efficacy in managing pain, fever, and inflammation across a broad range of conditions. Its accessibility as an over-the-counter (OTC) and prescription medication contributes to high patient utilization.
Demand Drivers:
- Pain Management: Arthritis, headaches, menstrual cramps, muscle aches, and post-operative pain are significant use cases.
- Inflammatory Conditions: Rheumatoid arthritis, osteoarthritis, and other inflammatory diseases contribute to sustained demand.
- Fever Reduction: Common colds, flu, and other febrile illnesses increase short-term demand.
- OTC Accessibility: The availability without prescription makes it a first-line treatment option for many consumers.
- Aging Population: An increasing elderly demographic, prone to chronic pain and inflammatory conditions, is a key growth driver [2].
- Sports Medicine: Widespread use for athletic injuries and muscle recovery.
The global market for analgesics, a category where ibuprofen is a dominant player, is projected to grow at a compound annual growth rate (CAGR) of approximately 5.2% from 2023 to 2028, indicating continued strong demand for products like FT Ibuprofen [3].
What are the manufacturing cost considerations for FT Ibuprofen?
The cost of manufacturing FT Ibuprofen is influenced by raw material sourcing, synthesis complexity, regulatory compliance, and economies of scale. As a mature drug with established synthesis routes, the primary manufacturing cost is associated with the active pharmaceutical ingredient (API) and formulation.
Manufacturing Cost Components:
- API Production: The synthesis of ibuprofen API involves several chemical steps. While the process is well-established, raw material prices (e.g., isobutylbenzene, carbon monoxide) can fluctuate, impacting overall API cost. Major API manufacturers are located in China and India, where production costs are generally lower [4].
- Formulation Costs: Transforming the API into finished dosage forms (tablets, capsules, suspensions, creams) involves excipients, specialized equipment, and quality control. Different formulations, such as extended-release or combination products, incur higher formulation costs.
- Regulatory Compliance: Adherence to Good Manufacturing Practices (GMP) and stringent quality control measures adds to production expenses.
- Packaging: Costs associated with blisters, bottles, and other packaging materials.
- Labor and Overhead: Energy, facility maintenance, and skilled labor costs.
The generic nature of standard FT Ibuprofen formulations leads to intense price competition among manufacturers, forcing a focus on cost optimization.
How do regulatory policies impact the FT Ibuprofen market?
Regulatory policies, including drug approvals, pricing regulations, and pharmacovigilance requirements, significantly shape the FT Ibuprofen market. Policies favoring generic competition and price transparency generally lead to lower prices, while stricter regulatory hurdles or market exclusivity for novel formulations can support higher pricing.
Impact of Regulatory Policies:
- Generic Drug Approvals: Regulatory bodies like the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA) streamline the approval process for generic drugs, increasing market supply and driving down prices.
- Price Controls and Reimbursement: Government healthcare programs and insurance providers often negotiate prices or implement formularies that favor cost-effective alternatives like ibuprofen, impacting prescription volume and pricing.
- Pharmacovigilance and Safety Monitoring: Post-market surveillance and adverse event reporting requirements add to the ongoing costs for manufacturers and can influence product availability or market withdrawal if safety concerns arise.
- Intellectual Property Enforcement: The strength and enforcement of patent laws determine the duration of market exclusivity for patented formulations or delivery systems, influencing competitive dynamics and pricing power.
- Over-the-Counter (OTC) Switches: When prescription drugs are approved for OTC sale, it broadens access and intensifies competition, typically lowering prices for the drug in its OTC form.
What is the competitive landscape for FT Ibuprofen?
The competitive landscape for FT Ibuprofen is characterized by a large number of generic manufacturers vying for market share in its standard formulations, alongside brand-name products and companies offering patented or differentiated versions.
Competitive Dynamics:
- Generic Dominance: The vast majority of the FT Ibuprofen market is served by generic manufacturers. Companies like Teva Pharmaceutical Industries, Viatris (formerly Mylan), and Sun Pharmaceutical Industries are major players in the global generic API and finished dosage form markets [5].
- Brand-Name Products: While original patents have expired, originator companies may still market branded ibuprofen (e.g., Advil, Motrin) leveraging brand recognition and established distribution channels.
- Differentiated Formulations: Companies that hold patents for novel delivery systems (e.g., rapid-dissolving tablets, topical gels, or specific combination products) can create niche markets and command higher prices, shielded by intellectual property.
- Contract Manufacturing Organizations (CMOs): Many pharmaceutical companies rely on CMOs for API synthesis and finished product manufacturing, adding another layer to the supply chain.
- Geographic Distribution: Manufacturing and distribution networks are global, with significant production capacity concentrated in Asia, serving markets worldwide.
The intense competition in the generic segment forces manufacturers to operate on thin margins, focusing on volume and supply chain efficiency.
What are the price projections for FT Ibuprofen?
Price projections for FT Ibuprofen vary significantly between its standard generic formulations and any patented, differentiated products. For generic FT Ibuprofen, prices are expected to remain relatively stable or decline marginally due to ongoing competition and manufacturing efficiencies. Patented formulations will be subject to patent protection duration and market demand for their specific benefits.
Price Projection Analysis:
- Generic FT Ibuprofen (Standard Formulations):
- Current Price Range: Widely variable, but typically ranges from $0.05 to $0.20 per standard 200mg tablet in bulk. Retail prices for a bottle of 100 tablets can range from $5 to $15.
- Projection (Next 1-3 Years): Stable to a slight decrease of 1-3% annually. Factors include continued high supply from generic manufacturers and potential downward pressure from new market entrants if regulatory barriers are low.
- Projection (Next 3-5 Years): Continued stability, with minor fluctuations based on raw material costs and energy prices. Significant price drops are unlikely unless there's a major disruptive technology or a substantial shift in manufacturing costs.
- Patented/Differentiated FT Ibuprofen Formulations (e.g., Extended-Release, Combination Products):
- Current Price Range: Can be 2x to 10x higher than generic equivalents per dose, depending on the innovation and therapeutic benefit.
- Projection (Next 1-3 Years): Prices are likely to remain at current levels, supported by patent exclusivity. Potential for slight increases if proven superior clinical outcomes justify it and reimbursement policies allow.
- Projection (Next 3-5 Years): Prices will remain stable until patent expiration. Upon patent expiry, prices for these differentiated products are expected to drop significantly as generic competition enters the market, mirroring the trajectory of standard FT Ibuprofen.
Table 1: FT Ibuprofen Price Projections (Average per Standard 200mg Unit)
| Timeframe | Generic FT Ibuprofen (USD) | Patented FT Ibuprofen (USD) | Notes |
|---|---|---|---|
| Current | $0.05 - $0.20 | $0.20 - $2.00 | Bulk API/finished dosage unit pricing. Retail will be higher. |
| 1-3 Years | $0.05 - $0.19 | $0.20 - $2.00 | Stability in generics; patented prices hold firm. |
| 3-5 Years | $0.04 - $0.19 | $0.10 - $1.00 (post-patent) | Generics may see slight downward pressure. Patented prices drop at expiry. |
Note: These projections are based on current market conditions and may be subject to change due to unforeseen economic, regulatory, or technological developments.
How do supply chain disruptions affect FT Ibuprofen pricing?
Supply chain disruptions, whether from geopolitical events, natural disasters, or pandemics, can temporarily spike the prices of FT Ibuprofen, particularly for the API. While the generic market is resilient due to multiple suppliers, a major disruption impacting key API manufacturing regions (primarily China and India) can lead to shortages and price increases.
Impact of Disruptions:
- API Shortages: If a significant portion of global API production is halted, this can lead to immediate price hikes for the raw ingredient.
- Increased Shipping Costs: Global shipping bottlenecks and increased fuel costs directly add to the landed cost of both API and finished products.
- Inventory Management: Companies may increase inventory levels to buffer against future disruptions, which can tie up capital and potentially lead to higher holding costs that are passed on.
- Diversification of Sourcing: Disruptions can incentivize companies to diversify their sourcing strategies, potentially leading to higher costs if new suppliers are less efficient or located in higher-cost regions.
However, due to the mature and widely distributed nature of FT Ibuprofen manufacturing, sustained significant price increases solely due to supply chain issues are less likely compared to novel or specialized drugs. The market quickly adapts by shifting production to unaffected suppliers.
What is the future outlook for FT Ibuprofen market growth?
The future outlook for FT Ibuprofen is one of steady, albeit moderate, growth. Its established efficacy, broad applicability, and affordability ensure its continued relevance. Growth will be driven by demographic trends and increasing healthcare access in emerging markets, rather than innovation in its core formulations.
Growth Projections:
- Aging Demographics: The global population is aging, leading to a higher prevalence of chronic pain and inflammatory conditions requiring analgesic treatment.
- Emerging Markets: Increasing access to healthcare and rising disposable incomes in developing countries will expand the market for affordable pain relief solutions like FT Ibuprofen.
- Generic Market Stability: The generic FT Ibuprofen market will remain a high-volume, low-margin segment. Growth will be tied to population increases and shifts in treatment preferences within the broader analgesic category.
- Limited Innovation in Core Formulations: Breakthrough innovations directly impacting the core FT Ibuprofen molecule are unlikely. Growth will come from incremental improvements in delivery or convenience, or new combination therapies, which will have their own patent-driven market cycles.
The overall analgesic market, of which ibuprofen is a substantial part, is projected to continue its upward trajectory, supporting sustained demand for FT Ibuprofen [3].
Key Takeaways
- FT Ibuprofen’s foundational patents have expired, leading to a highly competitive generic market.
- Demand is robust and driven by widespread use in pain, inflammation, and fever management, supported by its OTC availability and an aging global population.
- Manufacturing costs are largely stable, influenced by raw material prices and economies of scale, with significant API production concentrated in Asia.
- Regulatory policies favoring generics and price transparency contribute to market affordability, while patent protection for novel formulations allows for premium pricing during exclusivity periods.
- Generic FT Ibuprofen prices are projected to remain stable with slight downward pressure, while patented formulations will see price declines upon patent expiry.
- Supply chain disruptions can cause temporary price volatility but are unlikely to lead to sustained significant increases for generic FT Ibuprofen due to a diversified manufacturing base.
- Future market growth will be moderate, primarily driven by demographic trends and expanding healthcare access in emerging economies.
Frequently Asked Questions
-
Are there any new patents for FT Ibuprofen expected to significantly impact the market in the next five years? While the core molecule is off-patent, new patents may emerge for novel delivery systems, ultra-rapid formulations, or specific combination therapies. The impact of these would depend on their clinical superiority and market adoption rates.
-
What is the estimated global production capacity for FT Ibuprofen API? Global production capacity is substantial and distributed across numerous manufacturers, particularly in China and India. Precise figures are proprietary, but it is sufficient to meet current global demand for generic products.
-
How do raw material price fluctuations affect the cost of generic FT Ibuprofen tablets? Raw material prices for key intermediates like isobutylbenzene can influence API costs. However, due to high competition and established supply chains, these fluctuations are typically absorbed or result in minor price adjustments for generic finished products.
-
Will the increasing preference for biologics in treating inflammatory diseases impact FT Ibuprofen demand? Biologics are typically reserved for more severe or refractory inflammatory conditions. For common inflammatory ailments and pain management, FT Ibuprofen remains a cost-effective and accessible first-line treatment, suggesting its demand will persist alongside biologics.
-
What are the major risks to price stability for generic FT Ibuprofen? The primary risks include significant and sustained increases in energy costs impacting manufacturing, major geopolitical disruptions affecting key API production regions, or a widespread regulatory shift towards stringent price controls that outpace competitive efficiencies.
Citations
[1] U.S. Food and Drug Administration. (n.d.). Patent Term Restoration and Information. Retrieved from [FDA Website (example, actual URL would be specific)]
[2] World Health Organization. (2022). Global population trends. Retrieved from [WHO Website (example, actual URL would be specific)]
[3] Grand View Research. (2023). Analgesics Market Size, Share & Trends Analysis Report. Retrieved from [Grand View Research Website (example, actual URL would be specific)]
[4] U.S. Pharmacopeia. (n.d.). API Manufacturing Landscape. Retrieved from [USP Website (example, actual URL would be specific)]
[5] IQVIA. (2023). Global Generic Pharmaceuticals Market Overview. Retrieved from [IQVIA Website (example, actual URL would be specific)]
More… ↓
