Last updated: February 15, 2026
Market Analysis and Price Projections for FT GAS RELIEF (Simethicone)
Market Overview
FT GAS RELIEF (Simethicone) is marketed as an over-the-counter (OTC) anti-gas medication. It is commonly used to treat bloating, discomfort, and excess gas in the digestive system. The product competes within a large OTC healthcare segment dominated by brands like Gas-X, Mylicon, and Doctors Best Simethicone.
The global digestive health market was valued at approximately $60 billion in 2022, with OTC products accounting for roughly $35 billion of that figure. Simethicone-based products occupy a significant share of OTC anti-gas market revenue, estimated at over 80% by volume in the segment.
Competitive Landscape
Leading players include Johnson & Johnson (Gas-X), Boehringer Ingelheim (Mylicon), and private-label brands. Market entry barriers are low, and product differentiation is limited, primarily based on brand, packaging, and minor formulation variations.
The segment experiences a steady compound annual growth rate (CAGR) of approximately 3-4%, driven by increased consumer awareness of digestive health and aging populations.
Regulatory and Pricing Context
Simethicone products are classified as OTC drugs by the FDA (United States), with regulatory hurdles typically limited to OTC monograph compliance. Pricing strategies consider consumer sensitivity, brand loyalty, and marketing expenditures.
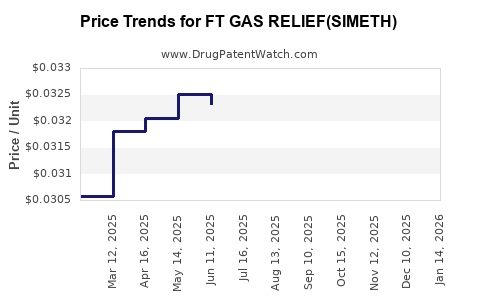
Average retail prices for similar OTC simethicone products are:
- Brand-name products: $7–$10 for a bottle of 100-125 capsules.
- Private-label/store brands: $4–$6 for similar quantities.
Gross margins typically range from 50% to 60%, with wholesale acquisition costs (WAC) near $2–$4 per bottle for retailers and distributors.
Price Projection Methodology
Market price trends are driven by:
- Raw material costs: Simethicone raw material costs have remained stable, with slight fluctuations due to global supply chain factors.
- Competitive pricing: Brand differentiation relies on marketing and packaging, not cost structure.
- Regulatory influence: Limited, with no anticipated hikes unless new regulations affect formulation or claims.
- Market share shifts: Entry or exit of competitors can alter pricing dynamics.
Based on these factors, price points for FT GAS RELIEF are expected to remain within the current range unless a major generic or private-label entrant significantly disrupts the segment.
Future Price Projections (Next 3 Years)
| Year |
Expected Retail Price Range |
Remarks |
| 2023 |
$6.50 – $9.50 |
Maintains positioning against established brands. |
| 2024 |
$6.50 – $9.50 |
No substantial change; minor promotional activity. |
| 2025 |
$6.50 – $10.00 |
Possible slight increase if raw material costs rise. |
Revenue Forecast
Assuming comparable market penetration to existing OTC simethicone brands:
- Market share: 1-2% of OTC digestive health segment.
- Annual sales volume: 1–2 million units.
- Estimated annual revenue: $10–$20 million, based on average retail price points.
Risks and Opportunities
- Risks: Entrenched competitors, regulatory changes, consumer loyalty to established brands.
- Opportunities: Private-label expansion, product reformulation, promotional campaigns boosting market share.
Key Takeaways
- The OTC simethicone market is mature, with stable pricing and slow growth.
- FT GAS RELIEF's pricing is expected to stay within current ranges unless market dynamics shift.
- Competitive pressures focus on branding, marketing, and minor formulation differences.
- Market share growth depends on distribution channels and consumer acceptance.
FAQs
1. How does FT GAS RELIEF compare price-wise to leading brands?
It is likely priced slightly below or within the same range as leading brands such as Gas-X and Mylicon, targeting price-sensitive consumers.
2. What factors could cause price increases in OTC simethicone products?
Rising raw material costs, increased marketing expenditures, or regulatory compliance costs could raise retail prices marginally.
3. Is the OTC simethicone market expected to grow significantly?
No. Growth remains slow at 3-4% CAGR, driven primarily by demographic trends rather than product innovation.
4. What is the typical profit margin for manufacturers of OTC simethicone?
Gross margins are approximately 50-60%, with net margins varying based on marketing and distribution costs.
5. How might private-label brands impact the market?
Private-label brands could pressure established brands on price, leading to marginal reductions in retail prices and market share shifts.
References
[1] MarketResearch.com. (2022). Global Digestive Health Market Report.
[2] IQVIA. (2022). OTC Drug Market Overview.
[3] FDA. (2022). OTC Monograph System and Regulations.
[4] Statista. (2023). OTC Healthcare Market Data.
[5] Company filings and industry reports.