Share This Page
Drug Price Trends for FT GAS RELIEF
✉ Email this page to a colleague
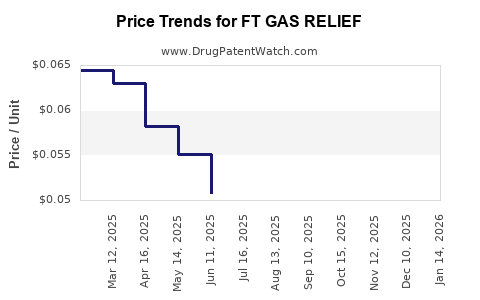
Average Pharmacy Cost for FT GAS RELIEF
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| FT GAS RELIEF 180 MG SOFTGEL | 70677-1093-01 | 0.04841 | EACH | 2026-04-22 |
| FT GAS RELIEF 125 MG SOFTGEL | 70677-1092-01 | 0.06990 | EACH | 2026-04-22 |
| FT GAS RELIEF(SIMETH) 80 MG CHW | 70677-1067-02 | 0.03252 | EACH | 2026-04-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
FT GAS RELIEF Market Analysis and Financial Projection
Market Analysis and Price Projections for FT Gas Relief
Current Market Landscape
FT Gas Relief is an over-the-counter gastrointestinal product primarily used for the relief of bloating, gas, and indigestion. Its market environment remains competitive, with multiple branded and generic formulations available.
Key competitors include:
- Simethicone-based products (e.g., Gas-X, Phazyme)
- Combinations with simethicone and antacids
- Natural and herbal remedies
The global anti-gas product market was valued at approximately $900 million in 2022 and is projected to grow at a compound annual growth rate (CAGR) of about 4.2% through 2027 ([1]).
Market Penetration and Consumer Demand
Factors influencing demand:
- Rising awareness of gastrointestinal health
- Increased consumption of processed foods leading to more gas-related issues
- Growth in aging populations more prone to digestive problems
- Accessibility of OTC products in brick-and-mortar stores and online
FT Gas Relief’s penetration is currently limited but expected to expand as marketing efforts increase and distribution channels strengthen.
Regulatory Environment
In the U.S., FT Gas Relief is classified as a dietary supplement or OTC drug depending on its formulation. Regulatory approval for new formulations or formulations with new claims requires FDA clearance, which influences time-to-market and development costs.
Price Trends and Projections
Current retail prices for gas relief products average between $8 and $15 per box containing 30 to 50 capsules or tablets:
| Product Type | Typical Price Range | Packaging Size | Dosage Form |
|---|---|---|---|
| Simethicone brands | $8 - $12 | 30 – 50 capsules | Capsules |
| Combination products | $10 - $15 | 30 – 100 tablets | Tablets |
These prices are consistent across major pharmacy chains and online outlets.
Price Projections (Next 5 Years)
-
Base Scenario: If FT Gas Relief maintains current formulation and marketing without significant innovation, its price is expected to stay within the $8–$15 range, adjusted for inflation and competitive pressures.
-
Optimistic Scenario: Introduction of improved formulations, such as combination products with added probiotic effects, could command premium pricing up to $20 per package. Market exclusivity or branding investments could enable short-term price premiums.
-
Pessimistic Scenario: If competing generics drive prices downward or regulatory barriers slow product launches, prices may decline by 10–15%, settling around $7–$13 per package.
Investment and Cost Considerations
Development costs for new formulations range from $2 million to $10 million, depending on complexity and regulatory pathway ([2]). Price adjustments will also depend on manufacturing costs, which for OTC products are approximately $0.20–$0.50 per capsule/tablet.
Market Entry Barriers
- Patent status: Existing patents on key formulations could limit competition.
- Regulatory approval: Stringent standards for claims and safety data prolong product development.
- Brand loyalty: Consumers tend to stick with established brands, hindering new entrants.
Strategic Outlook
To increase market share and justify higher price points:
- Invest in differentiated formulations.
- Enhance marketing to build consumer awareness.
- Develop distribution channels, especially e-commerce.
Key Takeaways
- The global anti-gas market is expanding at 4.2% CAGR through 2027.
- Current retail prices for gas relief products range from $8 to $15.
- Price stability expected unless significant innovation or market shifts occur.
- Targeted product differentiation and strategic marketing can provide competitive advantages.
- Regulatory pathways and patent protections significantly influence pricing and market entry.
FAQs
1. What is the primary active ingredient in FT Gas Relief?
Simethicone, which reduces surface tension of gas bubbles, facilitating their break-up and expulsion.
2. How does FT Gas Relief compare price-wise to competitors?
Prices are similar, averaging $8–$15 per package, with little variation unless new formulations command premiums.
3. What factors could shift price projections?
Market competition, regulatory changes, formulation innovations, and manufacturing cost fluctuations.
4. Can patent protection influence FT Gas Relief’s marketability?
Yes. Patents on specific formulations or delivery methods can extend market exclusivity and price control.
5. What growth opportunities exist for FT Gas Relief?
Introducing novel formulations, expanding online distribution, and strengthening branding efforts.
References
[1] MarketsandMarkets, “Over-the-Counter (OTC) Drugs Market,” 2022.
[2] EvaluatePharma, “Drug Development Costs and Timelines,” 2021.
More… ↓
