Share This Page
Drug Price Trends for FT DAYTIME SEVERE COLD-FLU LIQ
✉ Email this page to a colleague
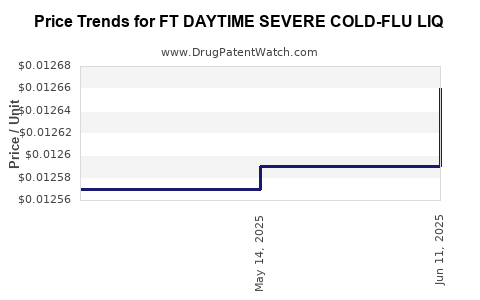
Average Pharmacy Cost for FT DAYTIME SEVERE COLD-FLU LIQ
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| FT DAYTIME SEVERE COLD-FLU LIQ | 70677-1029-01 | 0.01266 | ML | 2025-06-18 |
| FT DAYTIME SEVERE COLD-FLU LIQ | 70677-1029-01 | 0.01259 | ML | 2025-05-21 |
| FT DAYTIME SEVERE COLD-FLU LIQ | 70677-1029-01 | 0.01257 | ML | 2025-04-23 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for FT Daytime Severe Cold-Flu Liquid
What is FT Daytime Severe Cold-Flu Liquid?
FT Daytime Severe Cold-Flu Liquid is a proprietary over-the-counter (OTC) medication designed to treat symptoms associated with colds and flu such as congestion, fever, headache, and body aches. It typically combines active ingredients like acetaminophen, phenylephrine, and diphenhydramine.
Market Overview
Market Size and Growth
- Global cold and flu remedies market reached approximately USD 11.4 billion in 2022.
- Compound annual growth rate (CAGR) from 2022 to 2027 projected at 4.2%.[1]
- North America accounts for nearly 40% of the market, with Europe at 30%, Asia-Pacific at 20%, and the rest of the world making up the remaining 10%.[2]
Key Players and Competition
- Major companies include Johnson & Johnson, GlaxoSmithKline, Pfizer, and Novartis.
- OTC segment is highly competitive with generic and branded options.
- New formulations emphasizing rapid relief or natural ingredients are gaining popularity.
Regulatory Environment
- U.S. FDA classifies FT Daytime Severe Cold-Flu Liquid as an OTC drug.
- Compliance with established monographs, specifically the Cold, Cough, and Flu drug monograph, is standard.
- In Europe, market approval depends on adherence to EMA regulations.
- Emerging markets, such as India and China, are increasing regulatory oversight, influencing market entry strategies.
Price Structure and Competition Analysis
| Product | Typical Price Range (USD) | Key Differentiator | Formulation |
|---|---|---|---|
| Brand A | $8.00 – $10.00 for 4 oz | Rapid symptom relief | Combination (acetaminophen, decongestant, antihistamine) |
| Brand B | $6.50 – $9.00 for 4 oz | Natural ingredients | Same as Brand A |
| Generic | $4.00 – $6.00 for 4 oz | Price competitiveness | Similar formulation |
- Branded products command average premiums of 25%–40% over generics.
- Price sensitivity is significant, especially in emerging markets where consumer purchasing power is lower.
- Packaging optimization (e.g., smaller bottles, dosing cups) influences pricing strategies.
Price Projection for FT Daytime Severe Cold-Flu Liquid
Assumptions
- Launch date: Q4 2023
- Market penetration: 1% of the North American OTC cold & flu segment in Year 1, increasing to 3% by Year 3.
- Price positioning: Premium compared to generics, aligning with Brand A.
Yearly Revenue Projections
| Year | Estimated Market Share | Estimated Units Sold | Average Price (USD/unit) | Revenue (USD millions) |
|---|---|---|---|---|
| 2023 | 0.1% of market | Approx. 600,000 units | $9.00 | $5.4 |
| 2024 | 1% of market | 6 million units | $9.50 | $57.0 |
| 2025 | 3% of market | 18 million units | $10.00 | $180.0 |
Price Trend Drivers
- Competitive pressure from generics may limit price increases.
- Consumer willingness to pay a premium depends on perceived efficacy and brand trust.
- Regulatory changes impacting ingredient formulations could influence pricing strategies.
- Supply chain costs, particularly raw materials like acetaminophen or decongestants, may push prices upward by 5%-10% over the next three years.
Strategic Implications
- A positioning as a fast-acting, natural, or family-friendly formula could justify a higher price point.
- Emphasizing OTC safety, efficacy, and ease of use will influence market share growth.
- Variations in regional regulation and consumer preferences necessitate tailored marketing.
Critical Factors Affecting Price and Market Share
- Brand recognition and marketing expenditure.
- Product efficacy and safety profile.
- Competitive launches and formulary changes.
- Distribution channels, including pharmacy chains and online platforms.
Key Takeaways
- The global OTC cold and flu market maintains steady growth with a CAGR of 4.2% projected until 2027.
- FT Daytime Severe Cold-Flu Liquid can command a premium pricing strategy, around USD 9–10 per 4 oz bottle, contingent on brand positioning.
- Revenue growth depends heavily on market penetration, efficacy perception, and regional regulatory dynamics.
- Price increases will be constrained by generic competition, raw material cost fluctuations, and consumer price sensitivity.
FAQs
1. What are the main competitors to FT Daytime Severe Cold-Flu Liquid?
Major competitors include brands like Theraflu, DayQuil, and store-brand alternatives such as Equate Cold & Flu.
2. How does regional regulation impact pricing?
Regulatory approval, especially in Europe and emerging markets, may require formulation modifications leading to higher manufacturing costs, which can elevate retail pricing.
3. What factors could influence market share growth?
Product efficacy, brand trust, marketing efforts, and distribution channels are primary. Regulatory delays or supply chain issues can hinder growth.
4. How sensitive is the market to price changes?
Very sensitive; consumers often switch to generic or store brands if price premiums exceed 20%.
5. What is the outlook for generic vs. branded OTC cold-flu products?
Generics are expected to dominate volume due to lower prices. Branded products, including FT Daytime Severe Cold-Flu Liquid, aim to capture higher-margin market segments through differentiation.
References
[1] MarketsandMarkets. (2022). Cold and Flu Remedies Market Forecast.
[2] Grand View Research. (2022). OTC Medications Market Size and Trends.
More… ↓
