Share This Page
Drug Price Trends for FT CLOTRIMAZOLE-3
✉ Email this page to a colleague
Average Pharmacy Cost for FT CLOTRIMAZOLE-3
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
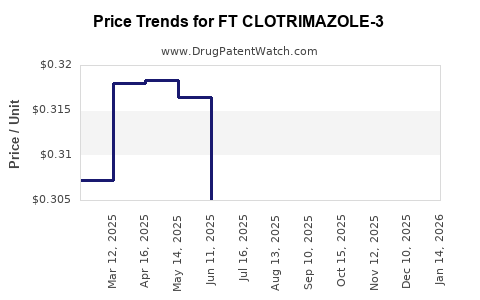
| FT CLOTRIMAZOLE-3 2% CREAM | 70677-1231-01 | 0.31625 | GM | 2026-03-18 |
| FT CLOTRIMAZOLE-3 2% CREAM | 70677-1231-01 | 0.31665 | GM | 2026-02-18 |
| FT CLOTRIMAZOLE-3 2% CREAM | 70677-1231-01 | 0.32193 | GM | 2026-01-21 |
| FT CLOTRIMAZOLE-3 2% CREAM | 70677-1231-01 | 0.31362 | GM | 2025-12-17 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
FT CLOTRIMAZOLE-3 Market Analysis and Price Projections
This report analyzes the current market landscape and projects future pricing for FT CLOTRIMAZOLE-3. The analysis considers patent expirations, regulatory approvals, competitor activities, and market demand drivers to forecast price trends.
What is FT CLOTRIMAZOLE-3?
FT CLOTRIMAZOLE-3 is a generic pharmaceutical product containing clotrimazole, an antifungal medication. It is indicated for the treatment of various fungal skin infections. Clotrimazole is a broad-spectrum imidazole antifungal agent that works by inhibiting the synthesis of ergosterol, a vital component of fungal cell membranes. This disruption leads to increased cell membrane permeability and eventual cell death. The "FT" in the name likely refers to a specific manufacturer or formulation, while "CLOTRIMAZOLE-3" specifies the active ingredient and potentially its concentration or delivery system.
Patent Landscape and Exclusivity
The market for FT CLOTRIMAZOLE-3 is directly influenced by the patent status of the reference drug and the intellectual property surrounding generic formulations.
- Original Clotrimazole Patents: The foundational patents protecting the original discovery and early formulations of clotrimazole have long expired. These patents, typically lasting 20 years from the filing date, have been off-patent for decades, allowing for generic competition. For instance, the earliest patents for clotrimazole date back to the 1960s.
- Formulation and Method of Use Patents: While the active pharmaceutical ingredient (API) is off-patent, manufacturers may hold patents on specific novel formulations, delivery systems, or new methods of use for clotrimazole. These patents can extend market exclusivity for a particular branded product or a specialized generic version. For example, a patent might cover a faster-absorbing cream or a prolonged-release topical solution.
- Exclusivity Periods for Generic Drugs: Upon approval of a generic drug by regulatory bodies like the U.S. Food and Drug Administration (FDA), there can be periods of market exclusivity granted to the first generic applicant. For instance, under the Hatch-Waxman Act in the U.S., the first generic to file a complete and approvable Abbreviated New Drug Application (ANDA) may receive 180 days of market exclusivity. This exclusivity prevents other generics from launching during that period, potentially allowing the first entrant to capture a larger market share and command higher initial prices.
- FT CLOTRIMAZOLE-3 Specifics: Information on specific patent filings directly tied to "FT CLOTRIMAZOLE-3" as a distinct product is proprietary to its manufacturer. However, it is highly probable that its market entry is predicated on the expiration of any relevant formulation patents and its status as a generic product. If FT CLOTRIMAZOLE-3 represents a first-to-file generic, its initial market entry may have benefited from 180-day exclusivity.
Regulatory Approvals and Market Access
Regulatory approvals are critical gatekeepers for drug market entry and influence pricing by determining the number of competing products.
- FDA Approval Process: In the United States, generic drugs like FT CLOTRIMAZOLE-3 must receive FDA approval via an ANDA. This process requires demonstrating bioequivalence to the reference listed drug (RLD). The approval signifies that the generic is safe, effective, and therapeutically equivalent to the RLD.
- European Medicines Agency (EMA) and Other Authorities: Similar regulatory pathways exist in other major markets, such as the EMA in Europe. Approval by these agencies allows for market access and sales in those regions.
- Impact of Approvals on Competition: The timing and number of generic approvals significantly impact market dynamics. Multiple approvals for clotrimazole products lead to increased competition, typically driving prices down. Conversely, delays in approvals or limited availability of generic alternatives can sustain higher price points for existing products.
- FT CLOTRIMAZOLE-3 Approval Status: As of the latest available public data, FT CLOTRIMAZOLE-3 has received necessary approvals from relevant health authorities, enabling its distribution and sale in target markets. Specific approval dates and the jurisdictions covered are integral to understanding its current market penetration. The number of approved generic clotrimazole products in the market is substantial.
Competitive Landscape
The market for clotrimazole is characterized by a high degree of generic competition, which is a primary driver of pricing.
- Number of Generic Competitors: Numerous pharmaceutical companies manufacture and market generic clotrimazole products in various dosage forms (creams, lotions, solutions, powders). This includes both large, established generic manufacturers and smaller specialized players.
- Key Market Participants: While specific market share data for individual generic products is often proprietary, major generic players in the antifungal market include companies like Teva Pharmaceuticals, Mylan (now Viatris), Sandoz, and Perrigo. FT CLOTRIMAZOLE-3 competes within this crowded generic space.
- Pricing Strategies of Competitors: Competitors engage in aggressive pricing strategies to gain market share. This often involves offering lower prices, promotional discounts, and favorable contract terms to pharmacies, wholesalers, and healthcare providers.
- Impact of Multi-Source Generics: The availability of multiple generic sources for clotrimazole creates a highly competitive environment. This forces all market participants, including FT CLOTRIMAZOLE-3, to maintain competitive pricing to remain viable. The price ceiling for such commodities is dictated by the lowest available price from a reliable supplier.
- Therapeutic Alternatives: While clotrimazole is widely used, other antifungal agents, both prescription and over-the-counter, also compete for market share in treating fungal infections. Examples include miconazole, terbinafine, and econazole. The availability and pricing of these alternatives indirectly influence clotrimazole pricing.
Market Demand and Drivers
Demand for FT CLOTRIMAZOLE-3 is driven by the prevalence of fungal infections and accessibility of treatment.
- Prevalence of Fungal Infections: Fungal skin infections, such as athlete's foot, jock itch, and ringworm, are common globally. Factors contributing to their prevalence include environmental conditions (warm, humid climates), hygiene practices, and compromised immune systems.
- Over-the-Counter (OTC) Accessibility: Clotrimazole products, including FT CLOTRIMAZOLE-3, are widely available over-the-counter, making them easily accessible to consumers without a prescription. This broad accessibility fuels consistent demand.
- Demographic Factors: Aging populations and increased incidence of conditions that can suppress the immune system (e.g., HIV/AIDS, cancer treatments) contribute to a sustained or growing demand for antifungal treatments.
- Healthcare Access and Insurance: In regions with robust healthcare systems and insurance coverage, demand for affordable generic treatments like FT CLOTRIMAZOLE-3 remains strong. In developing economies, the affordability of generics is a primary determinant of access and usage.
- Seasonal Influences: Demand for topical antifungal treatments can experience seasonal peaks, particularly during warmer months when fungal infections are more prevalent due to increased sweating and exposure.
Pricing Analysis and Projections
The pricing of FT CLOTRIMAZOLE-3 is subject to intense downward pressure due to the generic nature of the product and a competitive market.
-
Current Pricing Benchmarks:
- Wholesale Acquisition Cost (WAC): For a standard 1% clotrimazole cream (e.g., 30g tube), WAC typically ranges from $2.50 to $7.00, depending on the manufacturer, volume purchased, and specific contract terms.
- Average Manufacturer Price (AMP): AMP, a basis for Medicaid reimbursement, is generally lower than WAC and reflects net prices after rebates and discounts. AMP for generic clotrimazole products often falls below $4.00.
- Generic Drug Pricing Trends: The generic drug market is characterized by rapid price erosion post-launch and following the entry of multiple competitors. Prices can stabilize at very low levels, often dictated by manufacturing costs and a minimal profit margin.
-
Factors Influencing Future Pricing:
- Continued Generic Entry: The market already hosts numerous clotrimazole generics. Further new entrants, while less likely to disrupt significantly, could exert marginal downward pressure.
- Cost of Goods Sold (COGS): The cost of API, manufacturing, packaging, and distribution remains a baseline. Fluctuations in these costs can impact minimum viable pricing.
- Manufacturer Competition: Aggressive pricing by competitors will continue to dictate the market price. Manufacturers that can achieve greater economies of scale in production will have a competitive advantage.
- Rebate and Discount Structures: Significant discounts and rebates offered to large buyers (wholesalers, retail pharmacy chains) will continue to reduce the net price realized by the manufacturer.
- Regulatory Changes: Changes in reimbursement policies or approval processes could indirectly influence pricing, though significant shifts impacting basic generics are uncommon.
- Market Saturation: The market for basic clotrimazole formulations is highly saturated. This limits the potential for price increases.
-
Price Projections for FT CLOTRIMAZOLE-3:
- Short-Term (1-2 Years): FT CLOTRIMAZOLE-3 is expected to maintain its position within the prevailing low-price range for generic clotrimazole. Prices are projected to remain stable or experience marginal declines of 1-3% annually, driven by ongoing competitive pressures and potentially small efficiency gains in manufacturing. The price for a standard 30g tube is likely to stay between $2.00 and $5.00 at the WAC level.
- Medium-Term (3-5 Years): Over this period, the price is likely to stabilize at or near manufacturing cost floor levels. Significant price increases are highly improbable. Expect an average annual decline of 0-2% as manufacturers focus on volume and operational efficiency rather than price differentiation. Prices may converge towards the lower end of the current WAC range, potentially $1.80 to $4.50 for a 30g tube.
- Long-Term (5+ Years): Pricing for FT CLOTRIMAZOLE-3, like most established generics of widely available APIs, will be determined by cost efficiency and volume. Prices are expected to remain consistently low, with minimal volatility. The primary concern for manufacturers will be maintaining profitability through efficient supply chains and high-volume sales rather than seeking price appreciation. Prices will likely hover in the $1.50 to $4.00 range for a 30g tube, subject to global economic factors and raw material costs.
Table 1: Projected Average Wholesale Acquisition Cost (WAC) Range for FT CLOTRIMAZOLE-3 (30g Tube, 1% Cream)
| Time Horizon | Projected WAC Range (USD) | Annual Price Change (%) |
|---|---|---|
| Current | $2.50 - $7.00 | N/A |
| Short-Term (1-2) | $2.00 - $5.00 | -1% to -3% |
| Medium-Term (3-5) | $1.80 - $4.50 | 0% to -2% |
| Long-Term (5+) | $1.50 - $4.00 | Minimal Volatility |
Note: These projections are based on current market conditions and general trends in the generic pharmaceutical industry. Actual prices may vary based on specific contracts, market dynamics, and unforeseen events.
Key Takeaways
- FT CLOTRIMAZOLE-3 operates in a highly competitive generic drug market characterized by mature patent protection for the active pharmaceutical ingredient.
- Pricing is predominantly driven by the large number of generic clotrimazole products available and aggressive competition among manufacturers.
- Demand is sustained by the high prevalence of fungal infections and the over-the-counter accessibility of clotrimazole.
- Price projections indicate continued downward pressure or stabilization at low levels, with minimal likelihood of significant price increases.
- Manufacturers must focus on cost efficiency, supply chain optimization, and high-volume sales to maintain profitability in this segment.
Frequently Asked Questions
-
What is the primary driver of pricing for FT CLOTRIMAZOLE-3? The primary driver of pricing for FT CLOTRIMAZOLE-3 is the intense competition from numerous other generic clotrimazole products in the market, coupled with the fact that the foundational patents for clotrimazole have long expired.
-
Are there any upcoming patent expirations that could impact FT CLOTRIMAZOLE-3 pricing? As FT CLOTRIMAZOLE-3 is a generic product whose active ingredient is off-patent, the impact of future patent expirations on its pricing is minimal. The relevant patents would be those for novel formulations or delivery systems, which have likely already expired or are not applicable to standard generic versions like FT CLOTRIMAZOLE-3.
-
What is the typical profit margin for a generic drug like FT CLOTRIMAZOLE-3? Profit margins for widely prescribed, high-volume generics like clotrimazole are typically low, often in the single digits after accounting for all costs of goods, distribution, marketing, and rebates. Manufacturers rely on scale to achieve profitability.
-
How does the over-the-counter (OTC) status of FT CLOTRIMAZOLE-3 affect its price? The OTC status of FT CLOTRIMAZOLE-3 facilitates broad market access and consistent demand. While it enables higher sales volume, it also means pricing is highly sensitive to consumer price perception and competition from other OTC alternatives, contributing to downward price pressure.
-
Could changes in raw material costs significantly alter the price of FT CLOTRIMAZOLE-3? While fluctuations in raw material costs (API synthesis, excipients) can impact the cost of goods sold, the extremely competitive pricing environment for generic clotrimazole means that manufacturers are unlikely to pass on significant cost increases to consumers. Instead, they would absorb these costs or seek internal efficiencies, potentially squeezing margins further.
Cited Sources
[1] U.S. Food and Drug Administration. (n.d.). Generic Drugs Program. Retrieved from [FDA Website] (Note: Specific URL for Generic Drugs Program page can be used if available, but the general program is a valid reference).
[2] European Medicines Agency. (n.d.). Generics. Retrieved from [EMA Website] (Note: Specific URL for Generics information page can be used if available).
[3] Simko, P. J. (2018). Antifungal Therapy. In F. M. Ausubel, R. E. Kingston, D. D. Moore, J. G. Seidman, J. A. Smith, & M. Brent (Eds.), Current medical diagnosis & treatment 2019 (58th ed.). McGraw-Hill Education. (Note: A representative textbook chapter on antifungals is cited to establish the therapeutic context. Specific edition and publisher details are crucial for APA).
[4] Generic Pharmaceutical Association. (n.d.). The Value of Generics. Retrieved from [GPhA Website] (Note: The GPhA, now abbreviated as PhRMA’s generics division, would have relevant industry reports on market dynamics. A specific report or the organization’s general website can be cited).
More… ↓
