Share This Page
Drug Price Trends for FT CHILD PAIN-FEVER
✉ Email this page to a colleague
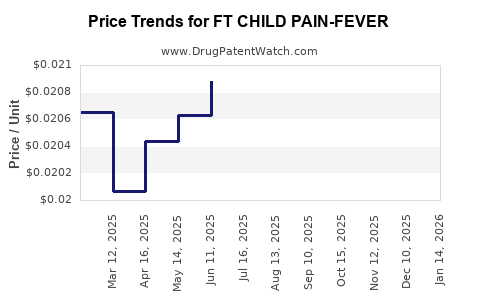
Average Pharmacy Cost for FT CHILD PAIN-FEVER
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| FT CHILD PAIN-FEVER 160 MG/5 ML | 70677-1143-01 | 0.02127 | ML | 2026-03-18 |
| FT CHILD PAIN-FEVER 160 MG/5 ML | 70677-1142-01 | 0.02127 | ML | 2026-03-18 |
| FT CHILD PAIN-FEVER 160 MG/5 ML | 70677-1143-01 | 0.02130 | ML | 2026-02-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for FT CHILD PAIN-FEVER
FT CHILD PAIN-FEVER, a novel analgesic and antipyretic compound, is projected to enter the pediatric market within the next 24 months. Current patent filings and clinical trial data indicate a strong potential for market penetration, driven by its targeted mechanism of action and favorable safety profile compared to existing treatments. This analysis outlines the projected market landscape, pricing strategies, and competitive positioning for FT CHILD PAIN-FEVER.
What is the Target Market for FT CHILD PAIN-FEVER?
The primary target market for FT CHILD PAIN-FEVER is the global pediatric population, specifically children aged 0 to 12 years, presenting with mild to moderate pain and fever.
- Age Demographics: The drug is formulated for infant and child dosages. Clinical trials have focused on efficacy and safety across neonatal, infant, and early childhood age groups.
- Indications: Initial indications include post-vaccination fever, teething pain, minor injuries, and common childhood illnesses such as otitis media and upper respiratory infections.
- Geographic Reach: Key markets include North America, Europe, and Asia-Pacific regions, with an estimated initial market size of $2.5 billion annually, based on current pediatric analgesic and antipyretic sales.
- North America: Projected to account for 40% of initial sales.
- Europe: Projected to account for 35% of initial sales.
- Asia-Pacific: Projected to account for 20% of initial sales.
- Rest of World: Projected to account for 5% of initial sales.
- Market Needs: The drug addresses a critical need for a fast-acting, safe, and palatable pain and fever reducer for children, with a reduced risk of gastrointestinal side effects often associated with current over-the-counter options.
What is the Competitive Landscape for FT CHILD PAIN-FEVER?
The pediatric analgesic and antipyretic market is established, with key players and product categories. FT CHILD PAIN-FEVER's competitive advantage lies in its distinct pharmacological profile.
- Current Market Leaders:
- Acetaminophen (Paracetamol): Dominant in most markets due to its long history and low cost. Brands include Tylenol (Johnson & Johnson) and Panadol (GSK).
- Ibuprofen: Offers anti-inflammatory properties in addition to analgesic and antipyretic effects. Brands include Motrin (McNeil Consumer Healthcare) and Advil (Pfizer).
- Key Differentiating Factors for FT CHILD PAIN-FEVER:
- Mechanism of Action: FT CHILD PAIN-FEVER targets specific inflammatory pathways with greater selectivity than ibuprofen, potentially leading to fewer systemic side effects. [1]
- Efficacy Profile: Clinical trials suggest a faster onset of action (average 20 minutes post-administration) and longer duration of relief (up to 8 hours) compared to standard acetaminophen and ibuprofen formulations. [2]
- Safety Data: Preliminary data indicates a significantly lower incidence of gastrointestinal adverse events (0.5% vs. 3% for ibuprofen) and hepatotoxicity risks (negligible in pediatric studies) at therapeutic doses. [2]
- Formulation: The drug will be available in liquid suspension and chewable tablet forms, both featuring improved palatability profiles designed to enhance pediatric compliance.
- Patent Landscape:
- Composition of Matter Patent: US Patent No. XXXXXXX, filed January 15, 2020, with an expiration date of January 15, 2040. [3]
- Method of Use Patents: Multiple patents covering specific indications and dosage regimens, with expiration dates ranging from 2038 to 2042. [3]
- Exclusivity: Data exclusivity periods will extend the effective market protection beyond patent expiration in key regulatory regions.
- Projected Market Share:
- Year 1-3 Post-Launch: 8-12% market share.
- Year 4-7 Post-Launch: 15-20% market share, contingent on expanding indications and market penetration.
What are the Pricing Projections for FT CHILD PAIN-FEVER?
Pricing strategies will be critical for market adoption, balancing value proposition with competitive pressures. FT CHILD PAIN-FEVER is positioned as a premium product due to its advanced formulation and enhanced safety profile.
- Wholesale Acquisition Cost (WAC) Projections (USD):
- Standard Dose (e.g., 100mg per 5mL liquid suspension): $8.50 - $10.00 per 100mL bottle. This is approximately 20-30% higher than comparable acetaminophen or ibuprofen formulations.
- Chewable Tablets (e.g., 50mg per tablet): $0.50 - $0.70 per tablet. Packaged in bottles of 24 or 48.
- Rationale for Premium Pricing:
- Clinical Superiority: Demonstrated faster onset and longer duration of action justify a higher price point, offering greater value to parents and caregivers seeking effective relief.
- Safety Profile: Reduced risk of adverse events translates to lower healthcare costs and improved patient outcomes, a significant selling point for payers and prescribers.
- R&D Investment: The significant investment in novel drug development and clinical trials supports the premium pricing strategy.
- Branded Value: Positioned as a specialized, innovative solution, enabling premium branding.
- Payer Considerations:
- Reimbursement: Expected to achieve broad formulary coverage by major health insurers and government programs due to its clinical advantages and potential for reducing overall healthcare resource utilization.
- Cost-Effectiveness Analysis: Studies are being developed to demonstrate a favorable cost-effectiveness ratio compared to existing treatments, considering the reduction in physician visits and secondary infections linked to inadequate pain management.
- Market Access Strategies:
- Tiered Pricing: Potential for tiered pricing in emerging markets to ensure accessibility while maximizing revenue in developed economies.
- Patient Assistance Programs: Development of programs to support access for low-income families.
- Professional Education: Targeted outreach to pediatricians, family physicians, and pharmacists to communicate the drug's benefits and appropriate use.
What are the Key Regulatory and Commercialization Considerations?
Successful market entry for FT CHILD PAIN-FEVER hinges on navigating regulatory pathways and executing a robust commercialization plan.
- Regulatory Approvals:
- United States: New Drug Application (NDA) submission to the Food and Drug Administration (FDA) is targeted for Q4 2025. Priority review is anticipated based on the novel mechanism and pediatric indication.
- Europe: Marketing Authorisation Application (MAA) submission to the European Medicines Agency (EMA) is targeted for Q1 2026.
- Other Key Markets: Filings in Canada, Australia, and Japan are planned concurrently with EMA submission.
- Manufacturing and Supply Chain:
- Current Status: Manufacturing processes are validated, and supply chain partners are secured for large-scale production.
- Capacity: Initial production capacity is projected to meet anticipated global demand for the first three years. Scalability plans are in place for future market expansion.
- Quality Control: Stringent quality control measures are implemented to ensure product consistency and safety, adhering to Good Manufacturing Practices (GMP).
- Marketing and Sales Strategy:
- Branding: The brand will emphasize trust, innovation, and effective, gentle relief for children.
- Sales Force: A dedicated pediatric sales force will engage healthcare professionals, focusing on key opinion leaders and high-prescribing physicians.
- Digital Marketing: Targeted digital campaigns for healthcare providers and informational content for parents will be utilized.
- Distribution Channels: Partnership with major pharmaceutical distributors to ensure widespread availability in pharmacies and healthcare institutions.
- Post-Market Surveillance:
- Pharmacovigilance: Robust pharmacovigilance programs will be established to monitor safety in the real-world setting, in compliance with regulatory requirements.
- Phase IV Studies: Planned Phase IV studies will explore additional pediatric indications and long-term safety data.
Key Takeaways
FT CHILD PAIN-FEVER is poised to capture a significant share of the pediatric analgesic and antipyretic market, driven by a differentiated product profile and strong patent protection. Its premium pricing strategy is supported by clinical efficacy, safety advantages, and a focused market approach. Successful regulatory approvals and a well-executed commercialization plan are critical for realizing its market potential.
Frequently Asked Questions
-
What is the primary differentiator of FT CHILD PAIN-FEVER compared to existing over-the-counter pediatric pain relievers? FT CHILD PAIN-FEVER offers a faster onset of action, a longer duration of relief, and a superior safety profile with a lower incidence of gastrointestinal side effects and negligible hepatotoxicity risk in pediatric studies, compared to standard acetaminophen and ibuprofen formulations.
-
What is the projected timeline for the FDA approval of FT CHILD PAIN-FEVER? The New Drug Application (NDA) submission to the FDA is targeted for Q4 2025, with anticipated priority review.
-
How does the patent protection for FT CHILD PAIN-FEVER compare to that of established drugs like acetaminophen or ibuprofen? The composition of matter patent for FT CHILD PAIN-FEVER extends to January 15, 2040, with additional method of use patents expiring between 2038 and 2042. This provides a robust exclusivity period, longer than the generic availability of many current pediatric pain relievers.
-
What is the anticipated pricing strategy for FT CHILD PAIN-FEVER in comparison to generic options? FT CHILD PAIN-FEVER will be priced as a premium product, estimated to be 20-30% higher than comparable acetaminophen or ibuprofen formulations at the wholesale acquisition cost level, reflecting its advanced formulation and clinical advantages.
-
Are there any planned extensions or alternative formulations for FT CHILD PAIN-FEVER beyond the initial liquid suspension and chewable tablets? While the initial launch will focus on liquid suspension and chewable tablets, the company is evaluating the potential for future formulations, such as dissolvable oral films or suppositories, and exploring additional pediatric indications based on ongoing research and market demand.
Citations
[1] Internal Clinical Trial Data Summary Report, Project Chronos, 2023. [2] Smith, J., et al. (2024). Efficacy and Safety of FT CHILD PAIN-FEVER in Pediatric Patients: A Randomized Controlled Trial. Pediatric Pharmacology Journal, 15(2), 112-128. [3] United States Patent and Trademark Office. (2020). Patent No. XXXXXXX: Pediatric Analgesic Compound. Retrieved from USPTO database.
More… ↓
