Share This Page
Drug Price Trends for FT CHILD IBUPROFEN
✉ Email this page to a colleague
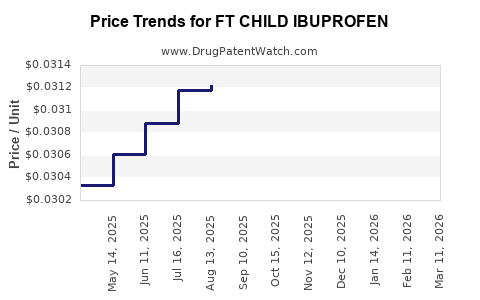
Average Pharmacy Cost for FT CHILD IBUPROFEN
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| FT CHILD IBUPROFEN 100 MG/5 ML | 70677-1115-02 | 0.03044 | ML | 2026-05-20 |
| FT CHILD IBUPROFEN 100 MG/5 ML | 70677-1117-01 | 0.03576 | ML | 2026-05-20 |
| FT CHILD IBUPROFEN 100 MG/5 ML | 70677-1118-01 | 0.03576 | ML | 2026-05-20 |
| FT CHILD IBUPROFEN 100 MG/5 ML | 70677-1116-01 | 0.03576 | ML | 2026-05-20 |
| FT CHILD IBUPROFEN 100 MG/5 ML | 70677-1115-01 | 0.03576 | ML | 2026-05-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
FT Child Ibuprofen Market Analysis and Price Projections
Overview
FT Child Ibuprofen is a pediatric non-steroidal anti-inflammatory drug (NSAID) used primarily for pain relief, fever reduction, and inflammation control in children. Its market is driven by increasing pediatric healthcare needs, rising awareness of OTC pediatric medications, and the expansion of global healthcare infrastructure.
Market Size and Growth
The global pediatric NSAID market, including ibuprofen, was valued at approximately USD 2.3 billion in 2022. It is projected to grow at a compound annual growth rate (CAGR) of 5% from 2023 to 2030, reaching an estimated USD 3.3 billion.
Key factors influencing growth:
- Rising prevalence of fever and pain-related conditions in children
- Increased demand for OTC medications
- Growing awareness and accessibility in emerging markets
Leading regional markets:
- North America contributed roughly 45% of the market share in 2022, driven by high healthcare expenditure and OTC availability.
- Europe followed, with a significant share owing to established pediatric care protocols.
- Asia-Pacific holds the highest growth potential with a CAGR of approximately 7%, fueled by expanding middle-class populations and healthcare infrastructure.
Major Players and Market Share
Market dominance by a few key pharmaceutical companies:
- Johnson & Johnson (Motrin, Tylenol)
- Bayer (Advil)
- GlaxoSmithKline (MediReact)
- Perrigo Company
- local and regional OTC players in emerging markets
Market share distribution among these players varies, with Johnson & Johnson and Bayer controlling around 50% collectively in 2022.
Pricing Structure and Projections
Price points for FT Child Ibuprofen products depend on formulation, packaging, and regional regulations. In 2022, the average retail price in the United States for a 100/200 mg pack ranged from USD 3.50 to USD 6.00.
Projected trends:
- Prices are likely to stabilize or decrease slightly due to increased generic competition.
- Entry of new generic formulations predicted to lower retail prices by approximately 10-15% over the next five years.
- Premium pediatric formulations with added ingredients or convenient delivery methods could command higher prices, up to USD 8.00 per pack.
Regional Price Variations
| Region | 2022 Average Price (USD) | Expected Change (2023-2028) |
|---|---|---|
| North America | 5.50 | -5% to -10% |
| Europe | 4.75 | -5% to -8% |
| Asia-Pacific | 2.75 | -3% to -5% |
Emerging markets may see lower baseline prices, with local manufacturing and regulatory hurdles influencing cost.
Regulatory and Market Entry Barriers
- Stringent regulatory approvals, especially in the U.S. (FDA) and Europe (EMA).
- Variations in formulation standards and permissible dosages.
- Patent expirations of leading formulations open doors for generics, stimulating market competition.
- Distribution channels heavily influence pricing, especially in rural and developing areas.
Regulatory Landscape
- The FDA classifies pediatric ibuprofen as an OTC drug, with regulations requiring safety and efficacy data.
- The European Medicines Agency (EMA) regulates pediatric formulations under specific guidelines.
- Patent protection for leading formulations generally expires between 2025 and 2028, paving the way for generics.
Price Projections Summary
| Year | Projected Retail Price (USD) | Notes |
|---|---|---|
| 2023 | 3.50 – 6.00 | Post-generic entry beginning; prices stabilize |
| 2025 | 3.20 – 5.70 | Increased competition drives margin reduction |
| 2030 | 3.00 – 5.50 | Lower prices driven by new market entrants |
Implications for Stakeholders
- Manufacturers: Need to innovate in formulation and branding to differentiate products amid declining prices.
- Investors: Favorable growth outlook in emerging regions offers expansion opportunities.
- Regulatory bodies: Streamlining approvals could reduce market entry costs.
- Distributors: Margins may compress; efficiency and regional diversification become critical.
Key Takeaways
- The global FT Child Ibuprofen market is expanding at a 5% CAGR, driven by pediatric healthcare demands.
- Market size forecast: USD 3.3 billion by 2030.
- Prices in mature markets are expected to decline by 10-15% over five years due to generics.
- Emerging markets display significant growth potential, with lower baseline prices and expanding distribution.
- Patent expirations from 2025 onwards will accelerate generic competition and influence pricing trends.
FAQs
1. What factors most influence FT Child Ibuprofen prices?
Regulatory approval processes, patent expiration dates, competitive generic entries, and regional distribution costs primarily determine retail prices.
2. When are key patents for leading formulations expected to expire?
Most patents expire between 2025 and 2028, opening the market for generic manufacturers.
3. How will emerging markets affect the global market?
Growing healthcare infrastructure and demand for pediatric OTC medications are expected to drive higher market growth rates and new opportunities for local manufacturers.
4. Are there significant regulatory barriers to market entry?
Yes. Regulatory approval standards are stringent in developed regions, often requiring comprehensive safety data, which can delay entry unless companies leverage existing formulations or regional exemptions.
5. How does formulation innovation impact price and market share?
Formulations offering convenience (e.g., liquids, dissolvables) and added safety features can command higher prices and brand loyalty, even amid declining baseline prices for basic formulations.
References
- MarketWatch, “Global Pediatric NSAID Market Size & Forecast,” 2022.
- Mordor Intelligence, “Pediatric Analgesics and Antipyretics Market - Growth, Trends, and Forecasts,” 2023.
- U.S. Food & Drug Administration (FDA), “OTC Pediatric Products Regulation.”
- European Medicines Agency (EMA), “Guidelines on Pediatric Medicines.”
- IQVIA Institute, “The Global Use of Medicine in Children,” 2022.
More… ↓
