Drug Price Trends for FT ASPIRIN
✉ Email this page to a colleague
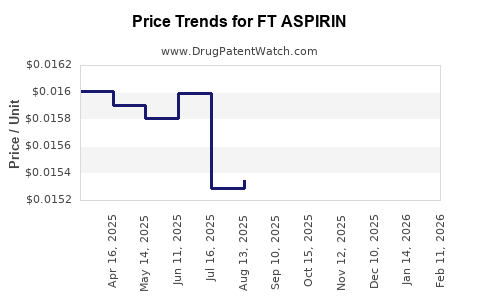
Average Pharmacy Cost for FT ASPIRIN
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| FT ASPIRIN EC 81 MG TABLET | 70677-1245-01 | 0.01244 | EACH | 2024-11-20 |
| FT ASPIRIN 81 MG CHEWABLE TAB | 70677-1134-01 | 0.02289 | EACH | 2024-11-20 |
| FT ASPIRIN EC 325 MG TABLET | 70677-1122-01 | 0.02147 | EACH | 2024-11-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |


