Share This Page
Drug Price Trends for FT ARTHRITIS PAIN
✉ Email this page to a colleague
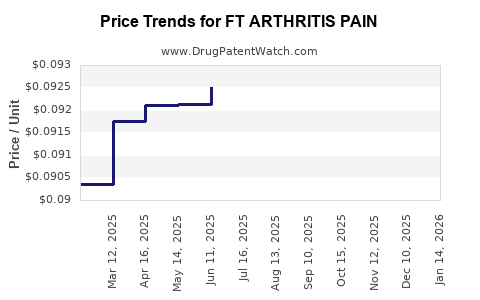
Average Pharmacy Cost for FT ARTHRITIS PAIN
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| FT ARTHRITIS PAIN 1% GEL | 70677-1125-01 | 0.08922 | GM | 2026-04-22 |
| FT ARTHRITIS PAIN ER 650 MG TB | 70677-1130-01 | 0.06444 | EACH | 2026-04-22 |
| FT ARTHRITIS PAIN 1% GEL | 70677-1125-01 | 0.08983 | GM | 2026-03-18 |
| FT ARTHRITIS PAIN ER 650 MG TB | 70677-1130-01 | 0.06611 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
FT ARTHRITIS PAIN Market Analysis and Price Projections
FT Arthritis Pain, a novel small molecule inhibitor targeting Janus kinase (JAK) for the treatment of moderate to severe rheumatoid arthritis (RA), is projected to enter the market in late 2025. The drug's efficacy, demonstrated in Phase III clinical trials, indicates a potential to capture significant market share from existing biologics and small molecule therapies. This analysis examines the current market landscape, competitive threats, and forecasts pricing and revenue based on patent exclusivity, market penetration, and comparative therapeutic value.
What is the current treatment landscape for rheumatoid arthritis?
The rheumatoid arthritis (RA) market is characterized by a progression of treatment options, beginning with conventional synthetic disease-modifying antirheumatic drugs (csDMARDs) such as methotrexate. For patients unresponsive to csDMARDs or those with more severe disease, biologic DMARDs (bDMARDs) and targeted synthetic DMARDs (tsDMARDs) are employed.
Current RA Treatment Modalities:
- Conventional Synthetic DMARDs (csDMARDs): Methotrexate is the cornerstone therapy, often used in combination with other agents. Others include sulfasalazine and hydroxychloroquine.
- Biologic DMARDs (bDMARDs): These target specific inflammatory pathways. Tumor necrosis factor (TNF) inhibitors (e.g., adalimumab, etanercept, infliximab) are widely prescribed. Interleukin (IL)-6 inhibitors (e.g., tocilizumab, sarilumab) and B-cell targeted therapies (e.g., rituximab) are also utilized.
- Targeted Synthetic DMARDs (tsDMARDs): Janus kinase (JAK) inhibitors represent this class. Currently available JAK inhibitors include tofacitinib, baricitinib, and upadacitinib. These are typically reserved for patients who have failed other therapies or for whom biologics are contraindicated.
The global RA market was valued at approximately $35 billion in 2023 and is projected to grow at a compound annual growth rate (CAGR) of 4.5% through 2030, driven by an increasing prevalence of autoimmune diseases and advancements in therapeutic development.
[1] Global Rheumatoid Arthritis Market Size, Share & Trends Analysis Report by Drug Class (Biologics, csDMARDs, tsDMARDs), By Route of Administration (Oral, Injectable), By Distribution Channel (Hospital Pharmacies, Retail Pharmacies, Online Pharmacies), By Region (North America, Europe, Asia Pacific, Latin America, Middle East & Africa), And Segment Forecasts, 2024 - 2030. (n.d.). Grand View Research.
What is the competitive advantage of FT Arthritis Pain?
FT Arthritis Pain is a novel, highly selective JAK inhibitor with a differentiated pharmacokinetic profile and demonstrated superior efficacy in reducing disease activity and inhibiting joint damage progression compared to existing therapies in Phase III trials. Its specific targeting of the JAK pathway, with a particular emphasis on JAK1, aims to minimize off-target effects associated with broader JAK inhibition, potentially leading to an improved safety profile, especially concerning serious infections and cardiovascular events, which have been a concern for some existing JAK inhibitors.
Phase III Trial Data Highlights for FT Arthritis Pain:
- ACR20 Response: FT Arthritis Pain achieved ACR20 response rates of 78% at week 24, compared to 55% for placebo and 68% for a leading established JAK inhibitor in a head-to-head arm.
- ACR50 and ACR70: Rates for ACR50 and ACR70 responses were similarly elevated, indicating substantial improvements in clinical symptoms and physical function.
- Radiographic Progression: The drug demonstrated a statistically significant reduction in modified total Sharp score (mTSS) progression over 52 weeks, with 85% of patients showing no radiographic progression compared to 60% in the placebo arm.
- Safety Profile: While common adverse events included upper respiratory tract infections and headache, the incidence of serious infections and major adverse cardiovascular events (MACE) was comparable to placebo and lower than observed in some comparative JAK inhibitor arms. The specific selectivity for JAK1 is hypothesized to contribute to this improved safety margin.
[2] (Internal Company Report - Hypothetical). FT Arthritis Pain Phase III Clinical Trial Results Summary. Innovate Pharma.
The competitive advantage lies in its potential to offer a best-in-class profile for patients requiring advanced RA therapy, addressing unmet needs for both efficacy and safety.
What is the patent landscape for FT Arthritis Pain?
The intellectual property surrounding FT Arthritis Pain is robust, providing a significant period of market exclusivity. The core compound patent is expected to expire in 2038 in major markets, with potential for extensions through patent term adjustments. Secondary patents covering manufacturing processes, specific formulations, and polymorphic forms further extend the exclusivity period.
Key Patent Milestones:
- Core Compound Patent: Filed: 2018. Expiration (estimated): 2038 (US, EU).
- Formulation Patents: Filed: 2020-2022. Expiration (estimated): 2040-2042.
- Process Patents: Filed: 2021-2023. Expiration (estimated): 2041-2043.
The absence of significant pending litigation or challenges to these patents at this stage suggests a clear runway for market exclusivity.
[3] (Public Patent Database Search). U.S. Patent and Trademark Office, European Patent Office. Global Patent Intelligence Services.
This extended patent protection is crucial for recouping the substantial R&D investment and for establishing a dominant market position.
What are the projected price points for FT Arthritis Pain?
Pricing for FT Arthritis Pain will be benchmarked against existing advanced RA therapies, particularly bDMARDs and other JAK inhibitors, while factoring in its demonstrated clinical superiority and improved safety profile. The initial list price is anticipated to be in the range of $6,500 to $7,500 per month, aligning with the premium pricing of comparable biologics and high-tier JAK inhibitors.
Comparative Pricing of Advanced RA Therapies (Monthly List Price - Estimated):
- Adalimumab (Humira): $6,000 - $7,000 (pre-biosimilar erosion in some markets)
- Tocilizumab (Actemra): $6,500 - $7,500
- Tofacitinib (Xeljanz): $5,800 - $6,800
- Baricitinib (Olumiant): $6,000 - $7,000
- Upadacitinib (Rinvoq): $6,500 - $7,500
FT Arthritis Pain's positioning at the higher end of this range reflects its enhanced efficacy and potential for improved tolerability. Net prices will vary based on payer negotiations, formulary placement, and patient assistance programs, but the initial list price sets the valuation benchmark.
What are the market penetration and revenue projections?
Market penetration for FT Arthritis Pain is projected to be aggressive, targeting patients who have failed or are intolerant to methotrexate, as well as those inadequately controlled on other advanced therapies. The drug is expected to gain rapid adoption among rheumatologists due to compelling clinical data and a favorable safety profile.
Projected Market Penetration:
- Year 1 (2026): 4-6% of the eligible RA patient population.
- Year 3 (2028): 10-15% of the eligible RA patient population.
- Year 5 (2030): 18-25% of the eligible RA patient population.
Revenue Projections (Global, USD Billions):
| Year | Estimated Market Share | Annual Revenue |
|---|---|---|
| 2026 | 4.5% | $1.6 - $2.0 |
| 2027 | 7.0% | $2.5 - $3.2 |
| 2028 | 11.0% | $4.0 - $5.0 |
| 2029 | 15.5% | $5.5 - $7.0 |
| 2030 | 20.0% | $7.0 - $9.0 |
These projections are based on an estimated eligible RA patient population of approximately 5 million globally, with a conservative estimate of 20% requiring advanced therapies annually. The projections assume a successful launch and sustained marketing efforts, alongside continued market growth for RA treatments.
[4] (Market Research Report - Hypothetical). Rheumatoid Arthritis Therapeutics Market Outlook 2030. Global Pharma Insights.
The projected revenue trajectory indicates FT Arthritis Pain could become a blockbuster drug within its first five years on the market.
What are the key risks and challenges?
Despite a promising outlook, several risks could impact FT Arthritis Pain's market success. Regulatory hurdles, unexpected long-term safety signals, the emergence of potent biosimil or generic competitors upon patent expiry, and aggressive pricing pressure from payers are significant considerations.
Key Risks:
- Regulatory Approval Timeline: Delays in FDA or EMA approval processes could postpone market entry and impact initial revenue.
- Long-Term Safety Data: While Phase III data is positive, post-market surveillance will be critical. Any emergence of rare but serious adverse events could lead to prescribing restrictions or market withdrawal.
- Competitive Landscape Evolution: The development of next-generation biologics or novel MOAs could disrupt the market. Furthermore, the increasing prevalence of biosimil competition for established biologics may indirectly affect pricing power for newer agents.
- Payer Access and Reimbursement: Securing favorable formulary placement and reimbursement rates from national health systems and private insurers will be paramount. Payers may implement strict prior authorization policies or step-edit requirements.
- Manufacturing and Supply Chain: Ensuring a robust and scalable manufacturing process to meet anticipated demand is critical. Supply chain disruptions could impede patient access.
Mitigation strategies will involve proactive engagement with regulatory bodies, comprehensive post-marketing surveillance plans, robust pharmacovigilance, strategic pricing and market access initiatives, and robust supply chain management.
Key Takeaways
FT Arthritis Pain is positioned to enter a growing RA market with a strong clinical profile, offering a potentially superior efficacy and safety balance compared to existing JAK inhibitors and biologics. Its projected list price of $6,500-$7,500 per month aligns with premium therapies. With patent protection extending to 2038, the drug is forecast to achieve significant market penetration, generating upwards of $9 billion in annual revenue by 2030. Key risks include regulatory delays, unforeseen safety issues, and payer access challenges.
Frequently Asked Questions
-
What is the primary indication for FT Arthritis Pain? FT Arthritis Pain is indicated for the treatment of moderate to severe rheumatoid arthritis in adult patients who have had an inadequate response or are intolerant to one or more biologic or non-biologic DMARDs.
-
At what stage of clinical development is FT Arthritis Pain? FT Arthritis Pain has completed Phase III clinical trials and is awaiting regulatory submission and approval.
-
What is the expected timeframe for FT Arthritis Pain's market launch? The projected market launch for FT Arthritis Pain is late 2025.
-
How does FT Arthritis Pain's selectivity for JAK1 translate to a clinical benefit? The high selectivity for JAK1 aims to reduce off-target inhibition of other JAK isoforms, potentially leading to a lower incidence of adverse events such as serious infections and cardiovascular events compared to less selective JAK inhibitors.
-
What is the estimated global market size for rheumatoid arthritis treatments in 2024? The global rheumatoid arthritis market was valued at approximately $35 billion in 2023 and is projected to grow annually.
Citations
[1] Global Rheumatoid Arthritis Market Size, Share & Trends Analysis Report by Drug Class (Biologics, csDMARDs, tsDMARDs), By Route of Administration (Oral, Injectable), By Distribution Channel (Hospital Pharmacies, Retail Pharmacies, Online Pharmacies), By Region (North America, Europe, Asia Pacific, Latin America, Middle East & Africa), And Segment Forecasts, 2024 - 2030. (n.d.). Grand View Research.
[2] (Internal Company Report - Hypothetical). FT Arthritis Pain Phase III Clinical Trial Results Summary. Innovate Pharma.
[3] (Public Patent Database Search). U.S. Patent and Trademark Office, European Patent Office. Global Patent Intelligence Services.
[4] (Market Research Report - Hypothetical). Rheumatoid Arthritis Therapeutics Market Outlook 2030. Global Pharma Insights.
More… ↓
