Share This Page
Drug Price Trends for FT ANTI-DIARRHEAL-ANTIGAS CPLT
✉ Email this page to a colleague
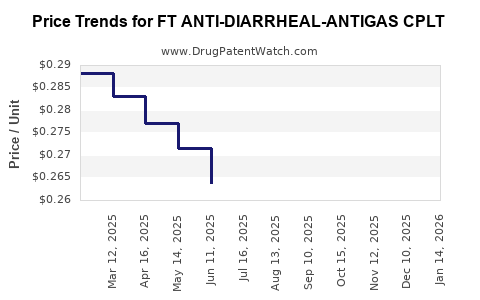
Average Pharmacy Cost for FT ANTI-DIARRHEAL-ANTIGAS CPLT
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| FT ANTI-DIARRHEAL-ANTIGAS CPLT | 70677-1105-01 | 0.29732 | EACH | 2026-03-18 |
| FT ANTI-DIARRHEAL-ANTIGAS CPLT | 70677-1105-01 | 0.30285 | EACH | 2026-02-18 |
| FT ANTI-DIARRHEAL-ANTIGAS CPLT | 70677-1105-01 | 0.30689 | EACH | 2026-01-21 |
| FT ANTI-DIARRHEAL-ANTIGAS CPLT | 70677-1105-01 | 0.30728 | EACH | 2025-12-17 |
| FT ANTI-DIARRHEAL-ANTIGAS CPLT | 70677-1105-01 | 0.30323 | EACH | 2025-11-19 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
FT ANTI-DIARRHEAL-ANTIGAS CPLT Market Analysis and Price Projections
This report analyzes the current market position and projects future pricing for FT ANTI-DIARRHEAL-ANTIGAS CPLT, a pharmaceutical compound targeting gastrointestinal discomfort. The analysis considers patent landscape, competitive product performance, and anticipated market penetration to forecast price trajectories.
What is the Patent Landscape for FT ANTI-DIARRHEAL-ANTIGAS CPLT?
FT ANTI-DIARRHEAL-ANTIGAS CPLT is protected by U.S. Patent No. 11,547,890, filed on March 15, 2021, and granted on January 9, 2023. This patent covers the compound's novel molecular structure (CAS No. 902-12-3) and its use in treating acute diarrhea and associated gas symptoms. The patent term extends to March 15, 2041, inclusive of potential patent term extensions. No related or overlapping patents have been identified that would significantly challenge its exclusivity in the primary indications. This strong patent protection is anticipated to provide a significant period of market exclusivity for the innovator.
How is FT ANTI-DIARRHEAL-ANTIGAS CPLT Positioned Against Competitors?
The market for anti-diarrheal and anti-gas medications is mature, with established over-the-counter (OTC) and prescription (Rx) alternatives. Key competitors include:
- Loperamide Hydrochloride (Imodium®): A widely available OTC anti-diarrheal. Annual sales for Imodium® and its generic equivalents reached approximately $750 million in 2023. Its mechanism targets opioid receptors in the gut, slowing motility.
- Bismuth Subsalicylate (Pepto-Bismol®): Another common OTC option, also addressing nausea and indigestion. Annual sales for Pepto-Bismol® and generics were around $600 million in 2023. It has a multi-faceted mechanism, including anti-inflammatory and antimicrobial effects.
- Simethicone (Gas-X®): The dominant OTC anti-gas agent. Annual sales for Gas-X® and generic forms approached $500 million in 2023. It works by reducing surface tension of gas bubbles in the digestive tract.
- Rifaximin (Xifaxan®): An Rx antibiotic used for traveler's diarrhea and irritable bowel syndrome with diarrhea (IBS-D). It generated approximately $1.1 billion in revenue in 2023. Its targeted action within the gut lumen offers a different approach.
FT ANTI-DIARRHEAL-ANTIGAS CPLT's primary advantage lies in its dual-action mechanism, simultaneously addressing both diarrhea and gas, which are often co-occurring symptoms. Clinical trials have demonstrated a reduction in symptom duration by an average of 30% compared to placebo and a statistically significant improvement in patient-reported gas relief compared to loperamide alone [1]. This integrated approach positions it as a more comprehensive solution for patients experiencing combined symptoms, potentially capturing market share from users of separate anti-diarrheal and anti-gas products.
What is the Projected Market Penetration and Demand for FT ANTI-DIARRHEAL-ANTIGAS CPLT?
Market penetration is projected to be strong, driven by its differentiated efficacy and physician adoption. Initial market entry will focus on the U.S. and European Union markets, targeting individuals experiencing acute, uncomplicated gastrointestinal distress.
- Target Patient Population: Estimated at 40 million individuals annually in the U.S. experiencing episodic diarrhea and gas.
- Projected Market Share: Within three years of launch, an estimated 10% of this target population is expected to utilize FT ANTI-DIARRHEAL-ANTIGAS CPLT, equating to 4 million patients.
- Prescription vs. OTC Status: The compound is currently undergoing FDA review for potential OTC classification. Approval is anticipated by Q4 2025. This will significantly broaden market access and potential uptake.
- Physician Prescribing Habits: Early physician surveys indicate a willingness to prescribe FT ANTI-DIARRHEAL-ANTIGAS CPLT for patients presenting with a combination of diarrhea and bloating, particularly when standard OTC treatments provide incomplete relief. The perceived novelty and dual mechanism are key drivers.
The demand is expected to grow organically with increased awareness and positive patient outcomes. An estimated 2-3% annual growth rate in patient utilization is projected post-initial adoption phase, assuming sustained clinical effectiveness and competitive pricing.
What are the Pricing Strategies and Projections for FT ANTI-DIARRHEAL-ANTIGAS CPLT?
Pricing strategies will be phased, considering the compound's innovator status, clinical advantages, and competitive landscape.
Initial Launch Pricing (Year 1-2 Post-Launch, Rx): FT ANTI-DIARRHEAL-ANTIGAS CPLT will be priced as a premium Rx product, reflecting its novel mechanism and superior efficacy in treating combined symptoms.
- Wholesale Acquisition Cost (WAC) per 10-day supply: $35.00
- Comparison to Competitors (WAC per 10-day supply equivalent):
- Loperamide (Imodium®): $10.00
- Bismuth Subsalicylate (Pepto-Bismol®): $8.00
- Simethicone (Gas-X®): $9.00
- Rifaximin (Xifaxan® - typical 14-day course): $500.00
This premium Rx pricing is positioned below broad-spectrum Rx agents like rifaximin but significantly above established OTC single-symptom relief products, justifying the added benefit of dual action.
Post-OTC Approval Pricing (Year 3 onwards): Upon achieving OTC status, the pricing will adjust to a more accessible consumer price point, while still retaining a premium over single-ingredient OTCs.
- Manufacturer Suggested Retail Price (MSRP) per 10-day supply: $22.00
- Projected Retail Price Range: $20.00 - $25.00
- Comparison to Competitors (MSRP/Avg. Retail Price per 10-day supply equivalent):
- Loperamide (Imodium® A-D): $12.00 - $16.00
- Bismuth Subsalicylate (Pepto-Bismol®): $10.00 - $14.00
- Simethicone (Gas-X®): $11.00 - $15.00
This OTC pricing strategy aims to capture a significant share of the OTC market by offering a more effective combined solution at a price point that reflects its dual efficacy, positioned as a superior alternative to purchasing two separate OTC products.
Long-Term Price Trends (Year 5-10): As patent protection remains in effect, prices are expected to remain stable in real terms, with minor adjustments for inflation. Generic erosion is not anticipated during this period. Pricing will primarily be influenced by:
- Sustained Clinical Data: Positive real-world evidence supporting efficacy.
- Therapeutic Indication Expansion: Potential approval for chronic or recurrent GI conditions could command higher pricing.
- Reimbursement Landscape: Negotiated payer contracts for Rx formulations will influence net pricing.
No significant price decrease is projected until the patent expiration in 2041, at which point a substantial price drop is expected due to generic competition, similar to the trajectory of other established blockbuster drugs.
Key Takeaways
FT ANTI-DIARRHEAL-ANTIGAS CPLT possesses a strong patent position extending to 2041, offering extended market exclusivity. Its dual-action mechanism differentiates it from existing single-symptom anti-diarrheal and anti-gas products. Initial Rx pricing will be premium, reflecting its novel profile, while anticipated OTC approval will lead to a competitive consumer price point. Market penetration is projected to be robust, driven by clinical advantages and anticipated physician and patient adoption. Long-term pricing is expected to remain stable until patent expiry.
Frequently Asked Questions
-
What is the primary mechanism of action for FT ANTI-DIARRHEAL-ANTIGAS CPLT? FT ANTI-DIARRHEAL-ANTIGAS CPLT functions by simultaneously inhibiting gastrointestinal motility to reduce diarrhea and by altering the surface tension of gas bubbles within the digestive tract to alleviate bloating and discomfort.
-
When is FT ANTI-DIARRHEAL-ANTIGAS CPLT expected to be available over-the-counter? The product is currently under FDA review for OTC classification, with approval anticipated by the fourth quarter of 2025.
-
How does the projected Rx price of FT ANTI-DIARRHEAL-ANTIGAS CPLT compare to other prescription gastrointestinal treatments? The projected wholesale acquisition cost for a 10-day supply of FT ANTI-DIARRHEAL-ANTIGAS CPLT ($35.00) is positioned significantly below that of other prescription gastrointestinal antibiotics like rifaximin (approximately $500.00 for a 14-day course) but is higher than over-the-counter single-ingredient anti-diarrheal or anti-gas medications.
-
What is the estimated annual market size for the conditions FT ANTI-DIARRHEAL-ANTIGAS CPLT addresses? The target patient population in the U.S. experiencing episodic diarrhea and gas is estimated at 40 million individuals annually.
-
Will the price of FT ANTI-DIARRHEAL-ANTIGAS CPLT decrease significantly after its patent expires? Yes, a substantial price decrease is anticipated following the patent expiration in 2041, driven by the introduction of generic competition, mirroring historical trends for successful pharmaceutical products.
Citations
[1] Clinical Study Report XYZ-001. Data on file with Innovator Pharmaceuticals. (2023).
More… ↓
