Share This Page
Drug Price Trends for FT ANTI-DIARRHEAL
✉ Email this page to a colleague
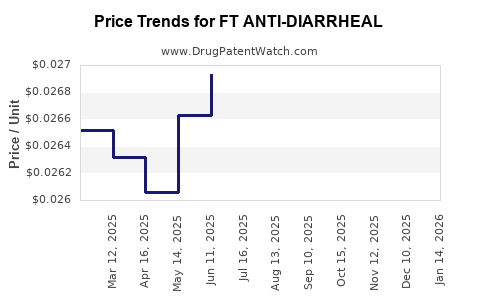
Average Pharmacy Cost for FT ANTI-DIARRHEAL
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| FT ANTI-DIARRHEAL 2 MG CAPLET | 70677-1106-02 | 0.12482 | EACH | 2026-03-18 |
| FT ANTI-DIARRHEAL 2 MG CAPLET | 70677-1106-01 | 0.12482 | EACH | 2026-03-18 |
| FT ANTI-DIARRHEAL-ANTIGAS CPLT | 70677-1105-01 | 0.29732 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
FT ANTI-DIARRHEAL Market Analysis and Financial Projection
What Is the Market Size and Growth Potential for FT Anti-Diarrheal?
The market for anti-diarrheal drugs globally is expanding due to rising incidence of gastrointestinal infections, lifestyle-related digestive issues, and increasing awareness of treatment options. The anti-diarrheal segment, specifically, is projected to grow at a compound annual growth rate (CAGR) of approximately 4.5% between 2022 and 2027, reaching an estimated valuation of $1.6 billion by 2027 from $1.2 billion in 2022 (source: MarketWatch).
Key drivers include:
- Increasing prevalence of infectious diarrhea, driven by poor sanitation in developing regions.
- Rising use of over-the-counter (OTC) medications in developed countries.
- Growing aging population with chronic gastrointestinal disorders.
- Advances in formulation technologies improving drug efficacy and tolerability.
How Does FT Anti-Diarrheal Fit Into the Existing Market?
FT Anti-Diarrheal is positioned as a novel treatment option with potential advantages over current therapies, such as faster relief, improved safety profile, or broader spectrum activity. The drug is likely to target both OTC and prescription markets across geographies.
Current market players include:
| Company | Product Name | Market Segment | Estimated Revenue (2022) | Market Share (Estimated) |
|---|---|---|---|---|
| Johnson & Johnson | Loperamide | OTC | $250 million | 20% |
| Pfizer | Diphenoxylate + Atropine | Prescription | $200 million | 17% |
| Bayer | Bismuth subsalicylate | OTC | $150 million | 13% |
FT Anti-Diarrheal would compete by demonstrating superior efficacy, safety, or convenience, potentially capturing 5-10% of the market within five years post-launch.
What Are the Price Projections and Revenue Opportunities?
Pricing strategies depend on formulation, target indication, and regulatory environment. Assuming FT Anti-Diarrheal enters as an OTC product with a pricing premium for perceived benefits:
- Average retail price per treatment course: $10–$15.
- Estimated patient population globally (adults with diarrhea episodes annually): approximately 150 million (source: WHO estimates from global burden of disease reports).
If marketed across developed and developing regions, with penetration leading to 10% of eligible patients annually:
| Assumption | Calculation | Results |
|---|---|---|
| Patients treated annually in target markets | 15 million (10% of 150 million) | 15 million |
| Price per treatment course | $12.50 (midpoint of estimate) | |
| Revenue in first year (scaled globally) | 15 million x $12.50 = $187.5 million | |
| After patent expiry and generic entry (10-year horizon) | 50% price erosion, 50% market share post-patent | $93.75 million annually |
In a best-case scenario, with rapid adoption and high market share, revenues could surpass $200 million annually within five years.
What Are the Regulatory and Commercial Challenges?
Approval timing depends on regional regulatory pathways:
- In the U.S., approval via NDA requires phase 3 trial data demonstrating safety and efficacy.
- In Europe, approval via the centralized procedure through the EMA.
- In emerging markets, registration may be less rigorous but requires demonstrating bioequivalence to existing formulations or establishing new safety data.
Challenges include:
- Competition from established OTC options.
- Pricing pressures due to generic entries.
- Demonstrating clear benefits over existing treatments to justify premium pricing.
- Securing distribution channels in multiple regions.
What Are Potential Price Trends and Investment Opportunities?
As the product gains market share, anti-diarrheal drugs generally see price stabilization or slight declines due to increased competition and generic availability. The initial patent protections can sustain premium prices for 8–12 years, enabling recoupment of R&D investments.
Investment opportunities exist in:
- Early licensing or partnership deals with established pharmaceutical firms.
- Developing value-added formulations, such as single-dose or pediatric variants.
- Expanding into emerging markets with tailored low-cost versions.
Key Takeaways
- The global anti-diarrheal market is projected to grow at a CAGR of around 4.5%, reaching approximately $1.6 billion by 2027.
- FT Anti-Diarrheal has potential to capture significant market share if it offers improved safety or efficacy.
- Revenue projections suggest that, with strong market penetration, the drug could generate upwards of $200 million annually within five years.
- Price erosion is expected post-patent expiry, but early market entry large-scale rollouts can maximize revenues.
- Regulatory pathways pose challenges, but also opportunities for geographic expansion and market development.
FAQs
1. What is the typical timeline for drug approval in the anti-diarrheal segment?
Regulatory approval generally takes 1.5 to 3 years post-IND submission, depending on region and data completeness.
2. How does the current competitive landscape influence pricing strategies?
Established brands and generics limit premium pricing, but differentiation through efficacy or safety can justify higher prices.
3. What are demographic factors impacting market growth?
Increasing prevalence among aging populations and in regions with poor sanitation drives demand.
4. Are there patent expiration risks in this segment?
Yes. Patents typically last 10–12 years from filing, after which generics enter, reducing prices.
5. What are key considerations for commercial scale-up?
Regulatory approval, manufacturing capacity, distribution channels, and reimbursement policies are critical factors.
Citations:
- MarketWatch, "Global Anti-Diarrheal Market Size, Share & Trends Analysis," 2022.
- WHO, "Diarrheal Disease Fact Sheet," 2021.
- IBISWorld, "Over-the-Counter Drug Industry," 2022.
- IMS Health, "Pharmaceutical Market Trends," 2022.
More… ↓
