Share This Page
Drug Price Trends for FT ANTACID-ANTIGAS LIQUID
✉ Email this page to a colleague
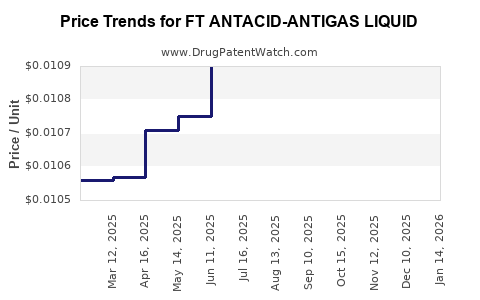
Average Pharmacy Cost for FT ANTACID-ANTIGAS LIQUID
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| FT ANTACID-ANTIGAS LIQUID | 70677-1066-01 | 0.01059 | ML | 2026-03-18 |
| FT ANTACID-ANTIGAS LIQUID | 70677-1063-01 | 0.01059 | ML | 2026-03-18 |
| FT ANTACID-ANTIGAS LIQUID | 70677-1066-01 | 0.01040 | ML | 2026-02-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
FT ANTACID-ANTIGAS LIQUID Market Analysis and Financial Projection
Market Analysis and Price Projections for FT ANTACID-ANTIGAS LIQUID
FT ANTACID-ANTIGAS LIQUID is an over-the-counter medication used to neutralize stomach acid, treat indigestion, heartburn, and gastroesophageal reflux disease (GERD). Its market segment includes OTC medications for digestive health, targeting consumers seeking quick relief from acid-related symptoms.
Market Overview
Global Market Size and Growth
The global antacid market was valued at approximately $3.8 billion in 2021. It is projected to grow at a compound annual growth rate (CAGR) of 4.5% from 2022 to 2027, reaching nearly $4.9 billion by 2027.[1]
Key Drivers
- Increase in prevalence of GERD and acid-related disorders.
- Growing awareness of digestive health.
- Preference for OTC remedies over prescription drugs.
- Aging population with higher incidence of digestive issues.
Competitive Landscape
Major players include GlaxoSmithKline (Gaviscon), Novartis (Maalox), and Bayer (Rennie). Many products feature similar formulations with variations in flavor, dosage, and branding.
Regulatory Environment
- OTC classification allows broad market access.
- Stringent labeling and marketing regulations in the US FDA and EMA jurisdictions require clear ingredients and dosing information.
- Patent expirations for several products have increased generic competition.
Product Positioning of FT ANTACID-ANTIGAS LIQUID
Unique Selling Proposition
- Liquid formulation for rapid absorption.
- Non-prescription status enables accessibility.
- Competitive pricing with mass-market appeal.
Formulation and Dosing
- Typically contains aluminum hydroxide and magnesium hydroxide.
- Dosing varies but generally ranges from 10-15 mL per dose, with a maximum of 4 doses daily.
Price Projections
Current Pricing
- Retail prices range from $5 to $10 for a 4 oz (120 mL) bottle.
- Average retail price: $7.50 per 4 oz bottle.
Short-term (Next 1-2 Years)
- Prices are expected to remain stable barring supply chain disruptions.
- Entry of generics could reduce average retail prices by approximately 10-15%, bringing a 4 oz bottle closer to $6.50.
Mid-term (3-5 Years)
- Market saturation may lead to price competition.
- Innovative packaging or formulation could support slight premium pricing.
- Predicted retail price for similar products: $6.75 to $8.
Long-term (5+ Years)
- Price declines likely as generics dominate.
- Potential for price erosion to around $5.50 - $7 per 4 oz bottle.
- Brand differentiation, if pursued, may sustain higher prices longer.
Revenue Outlook
Assuming initial annual sales volume of 1 million units at an average price of $7.50:
| Scenario | Volume (units) | Average Price | Estimated Revenue |
|---|---|---|---|
| Base case | 1 million | $7.50 | $7.5 million |
| Optimistic | 1.2 million | $8.00 | $9.6 million |
| Conservative | 0.8 million | $6.50 | $5.2 million |
Factors Influencing Revenue
- Market penetration strategy.
- Marketing and distribution channels.
- Competitive response from existing brands and generics.
- Regulatory changes affecting product labeling or formulation.
Future Market Trends
- Incremental innovation such as flavor enhancements or combined formulations.
- Increased focus on natural or organic ingredients.
- Digital marketing targeting consumers via online channels.
- Potential for expansion into emerging markets with rising digestive health awareness.
Key Takeaways
- FT ANTACID-ANTIGAS LIQUID operates in a stable and growing OTC digestive health segment.
- Price stability is expected in the short term, with downward pressure from generics expected over time.
- Market growth will depend on brand differentiation, marketing efforts, and regulatory landscape.
- Revenue projections are sensitive to market penetration and competitive dynamics.
FAQs
1. What are the main competitors to FT ANTACID-ANTIGAS LIQUID?
Gaviscon, Maalox, and Rennie are main competitors, offering similar antacid products with diverse formulations and branding.
2. How does generic competition affect pricing?
Generics typically enter the market after patent expirations, leading to price reductions of 10-15% or more, increasing market share but reducing profit margins for branded products.
3. What factors could influence future price trends?
Regulatory changes, ingredient costs, consumer preferences for natural products, and innovations in product formulation.
4. What is the growth outlook for the overall antacid market?
It is expected to grow at a CAGR of 4.5% through 2027, driven by increased prevalence of acid-related disorders and consumer preference for OTC solutions.
5. How significant is innovation in maintaining market share?
Innovation such as flavor versions, combination products, or natural ingredients can help extend product lifecycle and command premium pricing.
Sources:
[1] Grand View Research, "Antacids Market Size, Share & Trends Analysis Report," 2022.
More… ↓
