Share This Page
Drug Price Trends for FT ALL DAY ALLERGY-D
✉ Email this page to a colleague
Average Pharmacy Cost for FT ALL DAY ALLERGY-D
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
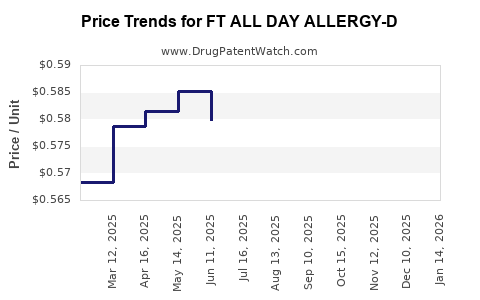
|---|---|---|---|---|
| FT ALL DAY ALLERGY-D 5-120 MG TB | 70677-1020-01 | 0.58202 | EACH | 2026-03-18 |
| FT ALL DAY ALLERGY-D 5-120 MG TB | 70677-1020-01 | 0.58858 | EACH | 2026-02-18 |
| FT ALL DAY ALLERGY-D 5-120 MG TB | 70677-1020-01 | 0.57637 | EACH | 2026-01-21 |
| FT ALL DAY ALLERGY-D 5-120 MG TB | 70677-1020-01 | 0.57122 | EACH | 2025-12-17 |
| FT ALL DAY ALLERGY-D 5-120 MG TB | 70677-1020-01 | 0.56169 | EACH | 2025-11-19 |
| FT ALL DAY ALLERGY-D 5-120 MG TB | 70677-1020-01 | 0.56239 | EACH | 2025-10-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
FT ALL DAY ALLERGY-D Market Analysis and Price Projections
FT ALL DAY ALLERGY-D is a multi-symptom allergy medication containing fexofenadine hydrochloride, an antihistamine, and pseudoephedrine hydrochloride, a decongestant. This analysis provides market insights and price projections based on current patent status, regulatory landscape, and competitive factors.
What is the Current Patent Landscape for FT ALL DAY ALLERGY-D?
The active pharmaceutical ingredients (APIs) in FT ALL DAY ALLERGY-D are fexofenadine hydrochloride and pseudoephedrine hydrochloride. Patents covering these individual APIs and their specific formulations have largely expired.
Fexofenadine hydrochloride, the active metabolite of terfenadine, was first marketed as Allegra by Hoechst Marion Roussel (now Sanofi-Aventis). Key patents for fexofenadine itself would have expired decades ago, allowing for generic entry. For instance, patents related to terfenadine and its metabolites, including fexofenadine, began expiring in the late 1990s and early 2000s [1].
Pseudoephedrine hydrochloride has been a widely available decongestant for many years, with patents on its basic chemical structure and common uses long since expired [2].
The primary patent protection for a specific branded drug like FT ALL DAY ALLERGY-D typically relates to:
- Formulation: Unique delivery mechanisms, extended-release technologies, or specific excipient combinations.
- Method of Use: Novel indications or treatment regimens.
- Combinations: Patents on the specific combination of fexofenadine and pseudoephedrine in a particular dosage or delivery form.
Given the commonality of the APIs, the market for FT ALL DAY ALLERGY-D is characterized by significant generic competition for the individual components. However, specific branded formulations may retain market share through marketing, brand recognition, and potentially patents on unique extended-release technologies or specific dosage forms that differentiate them from generic alternatives. The market for FT ALL DAY ALLERGY-D is thus primarily driven by the availability and pricing of generic fexofenadine/pseudoephedrine combination products.
What is the Regulatory Status and Market Access for FT ALL DAY ALLERGY-D?
FT ALL DAY ALLERGY-D is an over-the-counter (OTC) medication in the United States. It is approved by the U.S. Food and Drug Administration (FDA) for the temporary relief of nasal and sinus congestion, sinus pressure, sneezing, runny nose, itchy nose, and itchy throat. The presence of pseudoephedrine hydrochloride means it is subject to regulations under the Combat Methamphetamine Epidemic Act of 2005 (CMEA).
Under CMEA, products containing pseudoephedrine are kept behind the pharmacy counter. Retailers must maintain purchase logs, limit the amount consumers can buy within a 30-day period (typically 7.5 grams of pseudoephedrine base), and require identification for purchases [3]. These regulations affect accessibility and consumer purchasing behavior but do not fundamentally restrict market entry for approved products.
The regulatory environment for combination allergy medications remains stable. The FDA's oversight focuses on safety, efficacy, and labeling. For OTC products like FT ALL DAY ALLERGY-D, the primary hurdle for market entry is demonstrating bioequivalence for generic versions of established combination products. Once a generic is approved (through an Abbreviated New Drug Application, ANDA), it can compete directly with the branded product.
Market access for FT ALL DAY ALLERGY-D is broad due to its OTC status. It is available in most pharmacies, grocery stores, and mass merchandisers. Insurance coverage is generally not applicable to OTC medications unless prescribed by a physician for specific circumstances, which is rare for this type of product.
Who are the Key Competitors in the Allergy Medication Market?
The market for multi-symptom allergy relief is highly competitive, with numerous branded and generic options available. The primary competition for FT ALL DAY ALLERGY-D comes from other products containing fexofenadine/pseudoephedrine combinations and from alternative classes of allergy medications.
Direct Competitors (Fexofenadine/Pseudoephedrine Combinations):
- Generic Fexofenadine/Pseudoephedrine Extended-Release Tablets: A multitude of pharmaceutical companies manufacture generic versions of extended-release fexofenadine and pseudoephedrine combination products. These are typically sold under various brand names by distributors or as "store brands" by major retailers. Examples include products marketed by Teva, Aurobindo, and Sandoz. These generics offer a significant price advantage.
- Allegra-D (Sanofi-Aventis): This is the primary branded competitor that FT ALL DAY ALLERGY-D would have been developed to compete against. Allegra-D also contains fexofenadine hydrochloride and pseudoephedrine hydrochloride in an extended-release formulation.
Indirect Competitors (Alternative Allergy Medications):
- Second-Generation Antihistamines (Oral):
- Cetirizine (e.g., Zyrtec, generic versions): Often available in non-drowsy and drowsy formulations.
- Loratadine (e.g., Claritin, generic versions): Widely available, generally non-drowsy.
- Levocetirizine (e.g., Xyzal, generic versions): A more potent antihistamine. These are direct competitors for the antihistamine component of FT ALL DAY ALLERGY-D. Many of these are also available in combination with decongestants (e.g., loratadine-D, cetirizine-D).
- Nasal Corticosteroids (e.g., Flonase, Nasacort, Rhinocort): These are often considered first-line treatment for persistent allergic rhinitis and compete by addressing nasal inflammation directly. They are available OTC and by prescription.
- Decongestants (Oral and Nasal):
- Oral: Phenylephrine (often in products without pseudoephedrine due to CMEA restrictions), although efficacy is debated.
- Nasal Sprays: Oxymetazoline (e.g., Afrin), Phenylephrine. These offer rapid, short-term relief but carry a risk of rebound congestion.
- Combination Products with Different APIs: Some products combine an antihistamine with a decongestant that is not fexofenadine or pseudoephedrine.
The competitive landscape is shaped by price, brand recognition, perceived efficacy, side effect profiles, and availability. The significant presence of generics for fexofenadine/pseudoephedrine combinations means that pricing is a major determinant of market share for products like FT ALL DAY ALLERGY-D.
What are the Projected Market Trends for FT ALL DAY ALLERGY-D?
The market trends for FT ALL DAY ALLERGY-D are influenced by several factors:
- Genericization and Price Erosion: The primary trend is ongoing price erosion due to the widespread availability of generic fexofenadine/pseudoephedrine combination products. As patents on specific formulations expire or are circumvented by generic manufacturers, the price difference between branded and generic options widens, driving volume towards lower-cost alternatives.
- Shift Towards Novel Formulations and Delivery Systems: While the core APIs are old, there is continuous innovation in drug delivery. Companies may seek to differentiate products through improved extended-release technologies, easier-to-swallow formulations, or combinations with other beneficial ingredients. However, for established, older APIs like fexofenadine and pseudoephedrine, significant investment in novel delivery for a combination product might be limited unless a substantial improvement can be demonstrated.
- Competition from Nasal Steroids: Nasal corticosteroids have gained significant traction as effective treatments for allergic rhinitis. Their broad anti-inflammatory action can address multiple symptoms effectively, potentially reducing the need for oral antihistamine-decongestant combinations for some patients. This trend can limit the growth of oral combination products.
- Consumer Preference for Simplicity and Specificity: Some consumers may prefer single-ingredient medications or therapies targeting specific symptoms rather than broad-spectrum combination products, especially if they experience side effects from one component (e.g., decongestant-related side effects).
- Regulatory Scrutiny of Pseudoephedrine: While not expected to lead to outright bans, ongoing regulatory oversight of pseudoephedrine under CMEA may continue to influence purchasing patterns and potentially encourage the development or adoption of phenylephrine-based alternatives where deemed appropriate by regulatory bodies and manufacturers.
- Allergen Seasonality and Prevalence: The demand for allergy medications is inherently tied to allergen seasons and the prevalence of allergies in the population. Climate change and increasing urbanization can influence pollen counts and thus the demand for allergy relief products.
Given these trends, the market for branded FT ALL DAY ALLERGY-D, if it exists as a distinct branded product separate from generics, is likely to face continued pressure from generic alternatives and alternative therapeutic classes. Growth will likely be slow, with market share heavily influenced by promotional activities, consumer loyalty to established brands, and competitive pricing strategies. The overall market for allergy relief is robust due to the high prevalence of allergic conditions.
What are the Projected Price Ranges for FT ALL DAY ALLERGY-D?
Projecting the exact price of FT ALL DAY ALLERGY-D requires specific knowledge of its manufacturer, formulation (e.g., extended-release, dosage strength), and packaging (e.g., number of tablets). However, based on the competitive landscape, we can establish likely price ranges.
The price of FT ALL DAY ALLERGY-D will fall into two main categories: branded and generic.
1. Branded FT ALL DAY ALLERGY-D (if applicable as a distinct product):
If FT ALL DAY ALLERGY-D is a specific branded product with unique formulation patents or significant marketing investment, its price will be positioned above generic alternatives but likely competitive with established brands like Allegra-D.
- Estimated Retail Price Range (per package, e.g., 24-30 count): \$18 - \$35 USD. This price range reflects typical branding premiums for established OTC allergy medications. Factors influencing the higher end include proprietary extended-release technology, unique excipients, and significant marketing spend.
2. Generic Fexofenadine/Pseudoephedrine Extended-Release Tablets:
This category represents the vast majority of the market for this combination. Prices are driven by competition among generic manufacturers and retailers.
- Estimated Retail Price Range (per package, e.g., 24-30 count): \$8 - \$20 USD.
- Lower End (\$8 - \$12): Typically seen for store brands or generic versions from large pharmaceutical distributors with high-volume sales and lower marketing costs.
- Mid-Range (\$12 - \$17): Common for generics marketed by established pharmaceutical companies that may offer slightly better packaging or broader distribution.
- Higher End (\$17 - \$20): Could be for a specific generic manufacturer known for quality or a particular formulation that closely mimics the branded product, or during periods of supply chain fluctuation.
Factors Affecting Price Fluctuations:
- Generic Competition: The more generic manufacturers produce the same product, the lower the price will generally be.
- Retailer Pricing Strategies: Major retailers often use private-label generics as loss leaders or to drive foot traffic, leading to aggressive pricing.
- Manufacturer Discounts and Rebates: While less common for OTC products than for prescription drugs, some manufacturers offer coupons or bulk discounts.
- Supply Chain Disruptions: Shortages of APIs or manufacturing issues can temporarily drive up prices.
- Promotional Periods: Retailers may offer temporary price reductions during peak allergy seasons.
It is important to note that the term "FT ALL DAY ALLERGY-D" might refer to a specific product manufactured by a particular company. If this company is actively marketing it as a branded product against generic alternatives, its price would likely be at the higher end of the branded range. If it is simply a descriptive name used by a retailer for their generic offering, it would fall within the generic price ranges. Given the market, it is most probable that any product explicitly named FT ALL DAY ALLERGY-D would be positioned as a direct competitor to Allegra-D and would command a price within the branded range, or it is a descriptor for a generic version whose actual brand name appears on the packaging.
Key Takeaways
- Patents covering the active pharmaceutical ingredients fexofenadine hydrochloride and pseudoephedrine hydrochloride have expired, leading to a highly genericized market.
- FT ALL DAY ALLERGY-D, containing these APIs, faces intense competition from numerous generic fexofenadine/pseudoephedrine combination products and alternative allergy treatments like nasal corticosteroids.
- As an OTC medication containing pseudoephedrine, it is regulated under CMEA, requiring it to be kept behind the pharmacy counter.
- The market for FT ALL DAY ALLERGY-D is characterized by price erosion due to generic competition, with branded versions competing against established products like Allegra-D.
- Projected retail prices for a 24-30 count package range from \$18 - \$35 USD for a branded version and \$8 - \$20 USD for generic alternatives.
Frequently Asked Questions
- What is the primary mechanism of action for FT ALL DAY ALLERGY-D? FT ALL DAY ALLERGY-D combines fexofenadine hydrochloride, a second-generation antihistamine that blocks histamine receptors to reduce allergic symptoms like sneezing and itching, with pseudoephedrine hydrochloride, a decongestant that narrows blood vessels in the nasal passages to relieve congestion.
- Are there any significant side effects associated with FT ALL DAY ALLERGY-D? Common side effects of fexofenadine include headache, dizziness, and nausea. Pseudoephedrine can cause nervousness, sleeplessness, dizziness, and increased heart rate or blood pressure. Individuals with certain medical conditions (e.g., heart disease, high blood pressure, thyroid problems) should consult a healthcare professional before use.
- How does FT ALL DAY ALLERGY-D compare to single-ingredient allergy medications? FT ALL DAY ALLERGY-D addresses multiple allergy symptoms simultaneously with its dual-action formula. Single-ingredient medications, like an antihistamine alone or a nasal steroid alone, target specific symptom categories and may be preferred if only certain symptoms are present or if side effects from one component of the combination are problematic.
- What are the CMEA regulations regarding the purchase of FT ALL DAY ALLERGY-D? Due to containing pseudoephedrine, FT ALL DAY ALLERGY-D is sold from behind the pharmacy counter. Purchasers must typically show identification, and there are limits on the quantity that can be purchased within a 30-day period to prevent diversion for illicit drug manufacturing.
- Will FT ALL DAY ALLERGY-D be covered by health insurance? As an over-the-counter (OTC) medication, FT ALL DAY ALLERGY-D is generally not covered by health insurance. However, in specific cases where a physician deems it medically necessary and provides a prescription, some insurance plans might offer coverage, though this is uncommon for standard allergy relief.
Citations
[1] U.S. Food & Drug Administration. (n.d.). Patent Term Restoration Program. Retrieved from https://www.fda.gov/drugs/patent-term-restoration/patent-term-restoration-program (General information on patent expiration and restoration, relevant to API patent lifecycles).
[2] National Institutes of Health. (n.d.). Pseudoephedrine. In PubChem Compound Summary. Retrieved from https://pubchem.ncbi.nlm.nih.gov/compound/Pseudoephedrine (Provides chemical information and history; patent status is implied by its long-standing availability).
[3] U.S. Drug Enforcement Administration. (n.d.). Combat Methamphetamine Epidemic Act of 2005 (CMEA). Retrieved from https://www.dea.gov/drug-scheduling/combat-methamphetamine-epidemic-act-2005-cmea
More… ↓
