Share This Page
Drug Price Trends for FT ACID REDUCER DR
✉ Email this page to a colleague
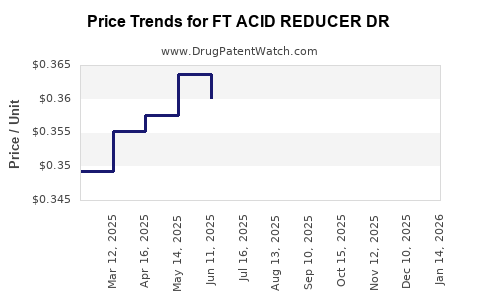
Average Pharmacy Cost for FT ACID REDUCER DR
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| FT ACID REDUCER DR 20 MG CAP | 70677-1099-02 | 0.34591 | EACH | 2026-03-18 |
| FT ACID REDUCER DR 15 MG CAP | 70677-1103-01 | 0.43232 | EACH | 2026-03-18 |
| FT ACID REDUCER DR 20 MG CAP | 70677-1099-03 | 0.34591 | EACH | 2026-03-18 |
| FT ACID REDUCER DR 15 MG CAP | 70677-1103-02 | 0.43232 | EACH | 2026-03-18 |
| FT ACID REDUCER DR 20 MG CAP | 70677-1099-01 | 0.34591 | EACH | 2026-03-18 |
| FT ACID REDUCER DR 20 MG CAP | 70677-1099-02 | 0.35047 | EACH | 2026-02-18 |
| FT ACID REDUCER DR 15 MG CAP | 70677-1103-01 | 0.42201 | EACH | 2026-02-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for FT Acid Reducer DR
What is FT Acid Reducer DR?
FT Acid Reducer DR is a delayed-release formulation of a proton pump inhibitor (PPI), used primarily for the treatment of gastroesophageal reflux disease (GERD), peptic ulcers, and other acid-related gastrointestinal conditions. It is marketed as an over-the-counter (OTC) and prescription medication. Its active ingredient, likely similar to agents such as omeprazole, lansoprazole, or pantoprazole, is designed for once-daily administration with a targeted release mechanism.
Market Size and Segmentation
Current Market Size (2022-2023)
- The global acid reducer market was valued at approximately USD 7.2 billion in 2022.
- The PPI segment accounts for roughly 70% of this market, estimated at USD 5 billion.
- Segment growth stems from increasing GERD prevalence, especially in North America and Asia-Pacific regions.
Consumer Segments
- OTC Users: Accounts for 60% of sales; driven by consumer preference for self-treatment.
- Prescription Users: Constitutes 40%; includes more severe or complicated cases requiring physician oversight.
Competitive Landscape
| Company | Key Products | Market Share (2023) | Distribution Channels |
|---|---|---|---|
| AstraZeneca | Nexium (Esomeprazole) | 25% | Pharmacy chains, hospitals, OTC retail |
| Johnson & Johnson | Prevacid (Lansoprazole) | 15% | Pharmacies, OTC |
| Pfizer | Protonix (Pantoprazole) | 10% | Hospitals, pharmacy chains |
| Generics | Multiple, including OTC variants | 40% | Wide distribution, lower prices |
FT Acid Reducer DR positions within this market, primarily targeting OTC consumers and certain prescription segments, leveraging delayed-release technology aimed at improved efficacy.
Pricing Dynamics and Projections
Current Pricing (2023)
- OTC Pack (14 tablets, 20 mg): USD 10 - USD 15.
- Prescription Pack (30 tablets, 20 mg): USD 25 - USD 35, depending on regional pricing and insurance coverage.
- Generics & Store Brands: Price points dip to USD 8 - USD 12 for OTC formats.
Price Drivers
- Patent status and exclusivity rights heavily influence price stability.
- Introduction of generics reduces consumer prices.
- Regulatory variations across regions affect pricing; stricter controls in Europe may keep prices higher.
Future Price Trends (2024-2028)
| Year | OTC Price Range (USD) | Prescription Price Range (USD) | Key Influencers |
|---|---|---|---|
| 2024 | 8 - 14 | 20 - 30 | Increased generic entries, regional price adjustments |
| 2025 | 7 - 13 | 18 - 28 | Patent expirations, market saturation, cost competition |
| 2026 | 6 - 12 | 16 - 26 | Brand-to-generic switch, healthcare policy impacts |
| 2027 | 6 - 11 | 15 - 25 | Increased OTC uptake, digital marketing strategies |
| 2028 | 5 - 10 | 14 - 22 | Continued generic proliferation, insurance coverage shifts |
Factors Impacting Price Projections
- Patent Expirations: Patents expected to expire between 2024 and 2026 for branded formulations.
- Regulatory Changes: Governments monitoring OTC availability and pricing controls.
- Market Entry: New entrants offering low-cost generics could push prices downward.
- Technological Advances: Improved formulation techniques may reduce manufacturing costs.
Regulatory and Patent Landscape
- FT Acid Reducer DR currently protected by patents expiring around 2024-2025.
- Most markets see rapid generic entry post patent expiry, promoting price competition.
- Some regions (e.g., Europe) have stricter regulations on OTC medicines, influencing the speed of generic penetration and price reductions.
Regional Market Variations
| Region | Market Size (2022) | Key Trends | Regulatory Environment |
|---|---|---|---|
| North America | USD 2.5 billion | High OTC adoption, patent protections, early generics | Favorable, with patent protections |
| Europe | USD 1.8 billion | Stricter OTC sales regulations, delayed generic entry | Tighter controls and approval delays |
| Asia-Pacific | USD 1.4 billion | Growing GERD prevalence, rising OTC sales | Less restrictive, rapid generics |
| Latin America | USD 0.5 billion | Emerging market, price-sensitive consumers | Moderate regulation |
Market Entry and Growth Opportunities
- Development of combination therapies (e.g., PPI + antacid).
- Expansion into emerging markets with rising GERD incidence.
- Digital marketing targeting OTC consumers.
- Launching low-cost generics post-patent expiry to increase market share.
Key Takeaways
- FT Acid Reducer DR operates within a highly competitive, rapidly evolving market driven by patent expiries and generic entry.
- Pricing for OTC formulations is currently USD 10 - USD 15 per pack but is expected to decline to USD 5 - USD 10 by 2028.
- Large markets include North America, Europe, and Asia-Pacific, each exhibiting different regulatory constraints and market behaviors.
- Patent expirations from 2024 to 2026 will trigger increased generic competition, pressuring prices downward.
- Emerging markets present growth opportunities due to rising GERD prevalence and less restrictive regulatory environments.
FAQ
1. When will FT Acid Reducer DR lose patent protection?
Patent expiry is projected between 2024 and 2025 in major markets like the U.S. and Europe.
2. How will generic entry impact pricing?
Generic competition typically reduces prices by 30% to 50% within one to two years of patent expiry.
3. What are the key regional differences affecting market share?
North America favors OTC sales with early patent protections, while Europe has stricter regulations delaying generic entry. Asia-Pacific has a faster adoption rate and lower regulatory barriers.
4. What factors could slow down price reductions?
Regulatory delays, limited generic pipelines, patent litigation, and supply chain disruptions can slow price declines.
5. Are there new formulations or combinations in development?
Yes. Research into combination therapies and sustained-release formulations aims to enhance efficacy and market appeal.
References
- MarketWatch. (2023). Global Proton Pump Inhibitors (PPIs) Market Size, Share & Trends. Retrieved from https://www.marketwatch.com
- Allied Market Research. (2022). Acid Reducer Market Analysis and Industry Forecast. Retrieved from https://www.alliedmarketresearch.com
- U.S. Food and Drug Administration. (2022). Patent expiry data for PPIs. Retrieved from https://www.fda.gov
- IQVIA. (2023). Global Prescription Drug Market Data. Retrieved from https://www.iqvia.com
- European Medicines Agency. (2022). Regulations on OTC Medicines. Retrieved from https://www.ema.europa.eu
More… ↓
