Last updated: February 17, 2026
What is Frova?
Frova (frovatriptan) is a selective 5-HT1B/1D receptor agonist used primarily for acute treatment of migraine attacks with or without aura. Approved by the FDA in 2001, it is part of the triptan class, targeting vasoconstriction to alleviate migraine symptoms.
Market Landscape
Current Market Size and Trends
The global migraine drug market was valued at approximately $7.5 billion in 2022. The triptan segment accounts for roughly 55% of this figure, with Frova holding an estimated 4-6% market share within triptans.
Key Competitors
Major competitors include:
- Sumatriptan (Imitrex)
- Rizatriptan (Maxalt)
- Zolmitriptan (Zomig)
- Eletriptan (Relpax)
- Lasmiditan (Reyvow) — newer CGRP receptor antagonist class
Strengths and Limitations
Frova's advantages include a long half-life (26 hours), which permits sustained relief and potentially fewer doses per attack. However, its limited availability compared to other triptans and concerns about cardiovascular risks hinder broader adoption.
Patent Status
Frova's original patent expired around 2010. Since then, generic versions have entered the market, contributing to reduced pricing and increased competition.
Price Trends
Historical Pricing
- Brand-name Frova: Historically sold at approximately $20-$25 per tablet (30 mg dose).
- Generic Frova: Prices have declined to about $4-$10 per tablet due to market entry.
Current Pricing Data
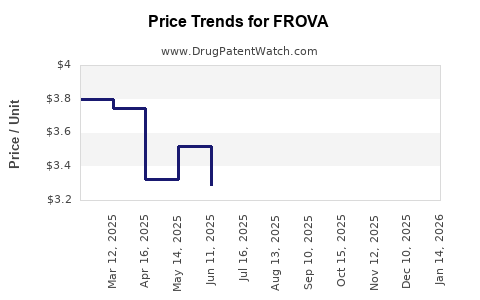
Based on databases like GoodRx and AAEM, average retail prices for generics in the U.S. range from $3.50 to $8.00 per tablet.
Reimbursement Environment
Insurance coverage varies; some payers favor generics, reducing patient co-payments.
Future Price Projections
Factors Influencing Prices
- Market Competition: Increased generic availability may sustain low prices.
- Regulatory Changes: Potential patent extension or new formulations could temporarily elevate prices.
- Manufacturing Costs: Stability in API prices and manufacturing may keep prices steady.
- Emergence of Alternatives: Newer drugs (e.g., CGRP antagonists) could erode Frova's market share, pressuring prices downward.
Projection Scenarios
| Scenario |
Timeline |
Price Trend |
Rationale |
| Base Case |
2023-2027 |
Stable to slight decline (~5%) |
Continued generic competition; steady demand |
| High Competition, Price Erosion |
2024-2026 |
Decline (~10-15%) |
Entry of new generics; increasing competition |
| Premium Segment Emerges |
2025-2027 |
Price stabilization or increase (~5%) |
Niche patient populations seeking longer half-life benefits |
Market Opportunities and Risks
Opportunities
- Positioning Frova as a long-acting triptan in markets with high migraine prevalence.
- Targeting historically underserved regions with low drug penetration.
- Developing combination formulations or new delivery systems.
Risks
- Patent expirations leading to increased generics.
- Market shift toward new drug classes like CGRP inhibitors.
- Pricing pressures from payers and healthcare systems.
Regulatory and Policy Impacts
- Patent landscapes show expiration of Frova's exclusivity.
- Generic approvals further support low-cost alternatives.
- Policy moves favoring biosimilars and generics could compress prices.
Conclusions
Frova's market remains stable primarily due to its long half-life and years of clinical use. However, high generic competition suppresses prices. Innovation or positioning within niche segments may sustain profitability. The overall trend suggests prices will remain low, with minor declines anticipated as generics dominate.
Key Takeaways
- Frova's market share is modest within the triptan class, mostly constrained by competition.
- Prices for generic Frova hover between $3.50 and $8.00 per tablet, trending downward.
- Market shifts toward CGRP therapies could erode Frova's segment share over the next five years.
- Future pricing likely to stabilize unless new formulations or indications drive niche markets.
- Manufacturers should consider differentiation strategies given the low-price environment.
FAQs
1. What is the primary therapeutic advantage of Frova?
Frova's long half-life (26 hours) allows sustained migraine relief with fewer doses, making it suitable for patients experiencing prolonged or recurrent migraines.
2. How has patent expiration affected Frova's market?
Patent expiration around 2010 led to the entry of generic formulations, significantly reducing prices and increasing accessibility, but also decreasing profitability for branded sales.
3. Are there opportunities for higher pricing in Frova?
Potential exists in niche markets where long-acting triptans are preferred, or through formulations with innovative delivery methods. Otherwise, generic competition constrains pricing.
4. How does Frova compare to other triptans in price?
Generic Frova is typically cheaper than brand-name triptans like Sumatriptan or Rizatriptan, making it a cost-effective choice for some patients and payers.
5. What future drug developments could impact Frova’s market?
Emergence of CGRP receptor antagonists and gepants offers alternative acute treatments, risking further market share erosion and influencing prices downward.
References
[1] IQVIA. "Global Migraine Market Analysis," 2022.
[2] GoodRx, "Frovatriptan Prices," 2023.
[3] FDA. "Frovatriptan Approval History," 2001.
[4] MarketWatch. "Migraine Drug Market Trends," 2022.