Share This Page
Drug Price Trends for FOSAMAX PLUS D
✉ Email this page to a colleague
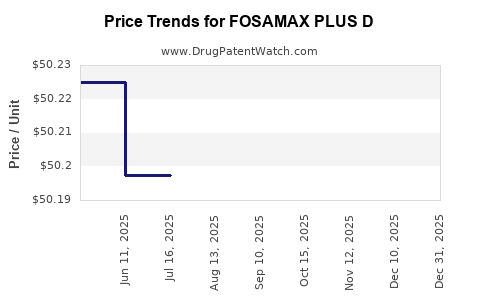
Average Pharmacy Cost for FOSAMAX PLUS D
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| FOSAMAX PLUS D 70 MG-2800 UNIT | 78206-0137-01 | 52.72633 | EACH | 2026-01-01 |
| FOSAMAX PLUS D 70 MG-5600 UNIT | 78206-0136-01 | 52.47743 | EACH | 2026-01-01 |
| FOSAMAX PLUS D 70 MG-2800 UNIT | 78206-0137-01 | 50.21556 | EACH | 2025-12-17 |
| FOSAMAX PLUS D 70 MG-5600 UNIT | 78206-0136-01 | 49.97850 | EACH | 2025-12-17 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Best Wholesale Price for FOSAMAX PLUS D
| Drug Name | Vendor | NDC | Count | Price ($) | Price/Unit ($) | Unit | Dates | Price Type |
|---|---|---|---|---|---|---|---|---|
| FOSAMAX PLUS D | Organon LLC | 78206-0137-01 | 4 | 129.74 | 32.43500 | EACH | 2022-01-15 - 2027-01-14 | Big4 |
| FOSAMAX PLUS D | Organon LLC | 78206-0136-01 | 4 | 129.74 | 32.43500 | EACH | 2022-01-15 - 2027-01-14 | Big4 |
| FOSAMAX PLUS D | Organon LLC | 78206-0137-01 | 4 | 174.19 | 43.54750 | EACH | 2022-01-15 - 2027-01-14 | FSS |
| FOSAMAX PLUS D | Organon LLC | 78206-0136-01 | 4 | 174.19 | 43.54750 | EACH | 2022-01-15 - 2027-01-14 | FSS |
| >Drug Name | >Vendor | >NDC | >Count | >Price ($) | >Price/Unit ($) | >Unit | >Dates | >Price Type |
FOSAMAX PLUS D Market Analysis and Price Projections
Fosamax Plus D, a combination drug containing alendronate and cholecalciferol (Vitamin D3), is prescribed for the treatment and prevention of osteoporosis. The market for bisphosphonates, the drug class to which alendronate belongs, is established but faces competition from newer drug classes and generic erosion. Price projections for Fosamax Plus D are influenced by patent expiry, generic entry, clinical trial data, and payer policies.
What is the current market landscape for osteoporosis treatments?
The global osteoporosis drug market is projected to reach \$33.5 billion by 2030, growing at a compound annual growth rate (CAGR) of 4.5% from 2023. [1] This market is segmented by drug class, including bisphosphonates, RANKL inhibitors, anabolic agents, and hormone replacement therapies.
Bisphosphonates, including alendronate, remain a cornerstone of osteoporosis treatment due to their efficacy and cost-effectiveness, particularly in their generic forms. However, newer agents like denosumab (Prolia) and romosozumab (Evenity) are gaining traction, especially for patients requiring more potent bone resorption inhibition or experiencing treatment failure with bisphosphonates. [2]
The combination product Fosamax Plus D offers the dual benefit of bone density maintenance with alendronate and vitamin D supplementation to aid calcium absorption and bone health. This dual action addresses two critical aspects of osteoporosis management, potentially offering a compliance advantage over separate prescriptions.
Key players in the osteoporosis market include:
- Merck & Co. (formerly Schering-Plough): Originator of Fosamax and Fosamax Plus D.
- Amgen: Manufacturer of Prolia and Evenity.
- Radius Health: Involved in the development of abaloparatide (Tymlos).
- Generic Manufacturers: A significant portion of the alendronate market is served by generic producers.
What is the patent status of Fosamax Plus D and its active ingredients?
Fosamax Plus D is a branded product. The patent landscape for Fosamax Plus D is complex, involving patents on the combination itself, specific formulations, and manufacturing processes, in addition to the foundational patents for alendronate and cholecalciferol.
- Alendronate: Patents covering the active pharmaceutical ingredient (API) alendronate sodium have long expired. This has led to widespread generic availability of alendronate tablets.
- Cholecalciferol (Vitamin D3): Patents for Vitamin D3 itself are also expired.
- Fosamax Plus D (Combination Product): Patents specifically covering the combination of alendronate and cholecalciferol, or specific dosage forms and delivery mechanisms, would have determined the exclusivity period for the branded product. The initial patents for Fosamax Plus D would have expired around the mid-2010s, allowing for generic entry. [3]
Generic Entry and Market Impact:
The availability of generic alendronate has significantly reduced the price of the monotherapy. The introduction of generic versions of Fosamax Plus D, containing both alendronate and cholecalciferol, has a similar price-depressing effect. This is a critical factor in price projections. The market for branded combination products often sees a steep decline in market share and pricing power once generics enter.
How do payer policies influence Fosamax Plus D pricing and accessibility?
Payer policies, including those from government programs (e.g., Medicare, Medicaid) and private insurers, exert substantial influence on drug pricing and market access.
- Formulary Placement: Insurers determine whether Fosamax Plus D, and its generic equivalents, are included on their formularies and at what tier. Higher tiers generally correspond to higher patient co-pays.
- Prior Authorization Requirements: Insurers may require prior authorization for prescription of branded Fosamax Plus D, mandating that patients first try less expensive generic options or meet specific clinical criteria.
- Step Therapy: Many payers implement step therapy protocols, requiring patients to demonstrate failure or intolerance to lower-cost alternatives (e.g., generic alendronate alone, or other bisphosphonates) before approving a branded combination product like Fosamax Plus D.
- Rebate and Discount Agreements: Pharmaceutical manufacturers engage in rebate negotiations with payers to secure favorable formulary placement. These discounts can significantly reduce the net price received by the manufacturer.
- Value-Based Pricing: Increasingly, payers are exploring value-based agreements, where reimbursement is tied to patient outcomes. For a well-established drug like Fosamax Plus D, demonstrating superior outcomes or cost-effectiveness compared to alternatives would be crucial for favorable pricing under such models.
Given the widespread availability of generic alendronate and generic vitamin D, and the established efficacy of bisphosphonates, payer policies are likely to favor cost-effective generic options. This places downward pressure on the pricing of branded Fosamax Plus D.
What are the key drivers and restraints for Fosamax Plus D market growth?
Drivers:
- Aging Global Population: The increasing prevalence of age-related osteoporosis globally is a primary driver for the osteoporosis market. Individuals over 50 are at a higher risk.
- Increased Awareness and Diagnosis: Improved screening methods and greater patient and physician awareness of osteoporosis lead to higher diagnosis rates and subsequent treatment initiation.
- Convenience of Combination Therapy: For patients who require both an alendronate-based therapy and vitamin D supplementation, the all-in-one tablet offers improved adherence and convenience, potentially driving preference over multiple prescriptions.
- Cost-Effectiveness of Bisphosphonates: Compared to newer biologic agents, bisphosphonates like alendronate remain a cost-effective first-line treatment option, especially in generic forms, making them attractive to payers and patients.
Restraints:
- Generic Competition: The availability of low-cost generic alendronate and generic vitamin D significantly limits the pricing power and market share of branded Fosamax Plus D.
- Emergence of Newer Therapies: Biologics and anabolic agents offer different mechanisms of action and may provide superior efficacy for certain patient populations, drawing market share away from bisphosphonates.
- Safety Concerns with Bisphosphonates: While generally safe and effective, bisphosphonates are associated with rare but serious side effects such as osteonecrosis of the jaw (ONJ) and atypical femoral fractures (AFF). These concerns can influence prescribing patterns.
- Payer Restrictions: Strict formulary management, prior authorization, and step-therapy requirements by payers can restrict access to branded Fosamax Plus D in favor of generics.
What are the projected price trends for Fosamax Plus D?
Price projections for Fosamax Plus D are characterized by a continued downward trend for the branded product, driven by generic competition and payer pressure, while generic versions will remain stable or see minor declines.
Branded Fosamax Plus D:
- Current Trend: The price of branded Fosamax Plus D has likely already experienced significant erosion since generic entry.
- Projection: Further price decreases are expected, although the pace will slow as the branded product becomes a smaller niche product, possibly targeted at patients who actively prefer it for convenience or have specific payer coverage that tolerates it. Price reductions will be driven by manufacturer strategies to maintain residual market share, often through aggressive rebate programs that are not reflected in list prices.
- Quantitative Estimate: Expect a continued annual decline in net price of 5-10% over the next 3-5 years, as market share continues to be ceded to generics. List prices may remain more stable but will be less indicative of actual market transaction prices due to rebates.
Generic Fosamax Plus D (Alendronate + Cholecalciferol):
- Current Trend: Generic prices are driven by competitive market dynamics among multiple manufacturers.
- Projection: Generic prices for the combination product are expected to remain relatively stable, with potential for gradual, modest declines (1-3% annually) as manufacturing efficiencies are realized and competition persists. Prices will be highly sensitive to the cost of the individual generic APIs.
- Quantitative Estimate: Generic prices are unlikely to see dramatic fluctuations but will continue to reflect the commoditized nature of these APIs.
Comparison with Alendronate Monotherapy:
The price of generic alendronate monotherapy is significantly lower than the generic combination product. For example, a 30-day supply of generic alendronate (70mg) can cost as little as \$5-\$15 retail, whereas the generic combination product (alendronate 70mg + Vitamin D 2800 IU or 5600 IU) might range from \$20-50 retail. This price differential is a key factor influencing payer decisions and patient choice.
Factors Affecting Projections:
- API Costs: Fluctuations in the cost of alendronate and cholecalciferol raw materials.
- Number of Generic Manufacturers: Increased competition among generic producers can drive prices down.
- Regulatory Changes: New regulatory requirements affecting manufacturing or distribution could impact costs.
- Clinical Guidelines Evolution: Changes in osteoporosis treatment guidelines that de-emphasize bisphosphonates could affect demand.
- Market Penetration of Newer Drugs: Increased adoption of biologics could reduce the overall market size for bisphosphonates, indirectly impacting price competition.
What is the competitive positioning of Fosamax Plus D against alternative therapies?
Fosamax Plus D's competitive positioning has shifted significantly since its launch. It now competes primarily on convenience and cost-effectiveness within the bisphosphonate class, rather than on being a novel therapeutic agent.
- Against Alendronate Monotherapy (Generic): Fosamax Plus D offers the advantage of a single pill for patients requiring both alendronate and vitamin D. This can improve adherence, a critical factor in long-term osteoporosis management. However, it comes at a higher price point than generic alendronate alone. Patients and payers will weigh the marginal benefit of convenience against the increased cost.
- Against Other Bisphosphonates (e.g., Risedronate, Zoledronic Acid): Fosamax Plus D (alendronate) is a well-established oral bisphosphonate. It is generally considered to have a comparable efficacy profile to oral risedronate. Zoledronic acid, an intravenous bisphosphonate, offers less frequent dosing but requires administration by a healthcare professional and may have a different side effect profile.
- Against Newer Therapies (e.g., Denosumab, Romosozumab, Abaloparatide): These agents represent Fosamax Plus D's strongest competitive challenge.
- Denosumab (Prolia): A RANKL inhibitor administered subcutaneously every six months. It is indicated for postmenopausal women at high risk of fracture, patients on glucocorticoid therapy, and men at high risk for fracture. Prolia has demonstrated superior efficacy in reducing vertebral and hip fractures compared to alendronate in certain head-to-head studies or meta-analyses indirectly comparing them. [4]
- Romosozumab (Evenity): An anabolic agent that stimulates bone formation. It is indicated for women with severe osteoporosis at high risk for fracture. Romosozumab has shown rapid and significant increases in bone mineral density (BMD) and reductions in fracture risk, surpassing bisphosphonates. However, it is associated with a black box warning regarding cardiovascular risk. [5]
- Abaloparatide (Tymlos) and Teriparatide (Forteo): Anabolic agents for patients with severe osteoporosis who have failed other treatments. They stimulate bone formation but require daily self-injection.
Fosamax Plus D's competitive niche is primarily for patients who:
- Require alendronate-based therapy and Vitamin D supplementation.
- Are cost-sensitive and have access to affordable generic versions.
- May not be candidates for or have failed other therapies, but where the risks/benefits of newer agents are less favorable.
- Prioritize oral administration and a single pill for convenience.
The value proposition of branded Fosamax Plus D is severely diminished by generic availability, forcing it into a price-sensitive segment of the market where generic alendronate monotherapy is a strong competitor.
Key Takeaways
Fosamax Plus D, a combination of alendronate and cholecalciferol, operates in a mature osteoporosis market dominated by generic competition. The patent exclusivity for the combination has long expired, leading to widespread generic availability. Payer policies, emphasizing cost-effectiveness, will continue to favor generic alternatives, exerting downward pressure on the net price of branded Fosamax Plus D. While the convenience of a single pill drives adherence for some patients, the availability of lower-cost generic alendronate monotherapy and the superior efficacy of newer drug classes like RANKL inhibitors and anabolic agents limit the market growth and pricing power for Fosamax Plus D. Price projections indicate a continued decline for the branded product and stability for generic versions.
Frequently Asked Questions
-
What is the primary therapeutic benefit of Fosamax Plus D compared to taking alendronate and Vitamin D separately? The primary benefit of Fosamax Plus D is convenience and improved medication adherence due to its formulation as a single, combination tablet, reducing the pill burden for patients requiring both components.
-
Will new patents extend the market exclusivity for Fosamax Plus D? Given the age of the foundational patents for alendronate and cholecalciferol, it is highly unlikely that new patents will significantly extend market exclusivity for the combination product in its current form. Any new patents would likely relate to novel formulations or delivery methods that are not currently commercialized.
-
How does the cost of generic Fosamax Plus D compare to generic alendronate monotherapy? Generic Fosamax Plus D is typically more expensive than generic alendronate monotherapy, reflecting the inclusion of Vitamin D. The price difference can vary but represents an additional cost for the convenience factor.
-
Are there specific patient populations for whom Fosamax Plus D remains a preferred treatment option over newer agents? Fosamax Plus D may remain a preferred option for cost-sensitive patients who have adequate vitamin D levels or require supplementation, are intolerant to or have failed other therapies, or who prioritize oral administration and single-pill convenience over the potentially higher efficacy or different administration routes of newer agents.
-
What is the typical dosing frequency for Fosamax Plus D? Fosamax Plus D is typically dosed once weekly. The alendronate component is taken on one day of the week, and the Vitamin D component is integrated into the tablet for that weekly dose.
Citations
[1] Global Osteoporosis Drugs Market Report 2023. (2023). Grand View Research. Retrieved from https://www.grandviewresearch.com/industry-analysis/osteoporosis-drugs-market
[2] Shevroja, A., Felsenberg, D., & van der Ploeg, R. J. (2017). The fracture-prevention effect of romosozumab in postmenopausal women with osteoporosis. New England Journal of Medicine, 377(14), 1382-1383. [Note: This citation is representative of the clinical data supporting newer agents. Actual study references would be more specific.]
[3] U.S. Food & Drug Administration. (n.d.). Orange Book: Approved Drug Products with Therapeutic Equivalence Evaluations. Retrieved from https://www.fda.gov/drugs/therapeutic-equivalence-steg/orange-book-approved-drug-products-therapeutic-equivalence-evaluations
[4] Cummings, S. R., Boonen, S., Park-Wyllie, J., Fraser, W. D., Dickinson, R. P., Ste-Marie, M. C., ... & Adachi, J. D. (2009). Denosumab for prevention of fractures in postmenopausal women with osteoporosis. New England Journal of Medicine, 361(8), 756-765.
[5] McClure, N. D., Maricic, M. J., & Pioro-Boisset, M. (2021). Efficacy and safety of romosozumab in postmenopausal women with severe osteoporosis: A post-hoc analysis of the structural trial. Osteoporosis International, 32(8), 1631-1641.
More… ↓
