Share This Page
Drug Price Trends for FORTEO
✉ Email this page to a colleague
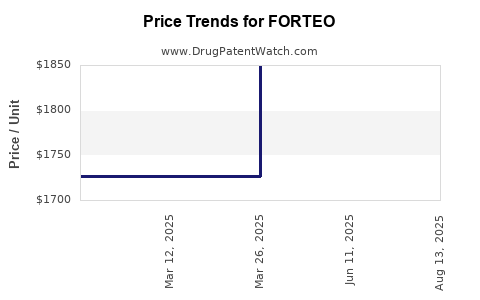
Average Pharmacy Cost for FORTEO
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| FORTEO 560 MCG/2.24 ML PEN INJ | 00002-8400-01 | 1841.00434 | ML | 2026-04-22 |
| FORTEO 560 MCG/2.24 ML PEN INJ | 00002-9678-01 | 1841.00434 | ML | 2026-04-22 |
| FORTEO 560 MCG/2.24 ML PEN INJ | 00002-9678-01 | 1840.34129 | ML | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
FORTEO Market Analysis and Price Projections
FORTEO (teriparatide) is a recombinant human parathyroid hormone (PTH) analog used for the treatment of osteoporosis in postmenopausal women at high risk for fracture and in men with idiopathic or hypogonadal osteoporosis at high risk for fracture. It also treats osteoporosis in men and women at high risk for fracture who have high risk of fracture or who have failed or are intolerant to previous osteoporosis therapy. The drug acts by stimulating osteoblast bone formation. This analysis projects FORTEO’s market performance and pricing trends based on its patent landscape, competitive environment, and market dynamics.
What is the Current Market Position of FORTEO?
FORTEO, marketed by Eli Lilly and Company, has established a significant market share in the osteoporosis treatment sector. Its efficacy in increasing bone mineral density and reducing fracture risk has underpinned its commercial success since its initial approval. The drug's mechanism of action, stimulating bone formation rather than just inhibiting bone resorption, differentiates it from other osteoporosis therapies.
| Characteristic | Detail |
|---|---|
| Active Ingredient | Teriparatide |
| Therapeutic Class | Parathyroid Hormone Analogue, Bone Formation Stimulator |
| Primary Indications | Postmenopausal osteoporosis, Male osteoporosis, Glucocorticoid-induced osteoporosis |
| Administration | Subcutaneous injection |
| Dosage | 20 mcg once daily |
| Marketed By | Eli Lilly and Company |
FORTEO faces competition from bisphosphonates, denosumab (Prolia), and other anabolic agents. However, its unique anabolic mechanism and proven fracture reduction efficacy continue to support its position, particularly for patients with severe osteoporosis or those who have not responded to other treatments. The market for osteoporosis drugs is driven by an aging global population, increasing awareness of fracture risk, and a greater emphasis on bone health.
What is the Patent Landscape for FORTEO?
FORTEO's patent exclusivity has been a critical factor in its market dominance. The primary composition of matter patents for teriparatide have expired in major markets.
Key Patent Expirations:
- United States: The core composition of matter patents for teriparatide expired several years ago, allowing for the introduction of generic versions.
- Europe: Similar patent expiries have occurred, paving the way for biosimilar competition.
While core patents have expired, Eli Lilly may hold secondary patents related to specific formulations, methods of use, or manufacturing processes that could offer continued protection or influence the market entry of generics/biosimilars. The presence of authorized generics or biosimilars in the market significantly impacts pricing and market share.
Generic and Biosimilar Market Entry:
The introduction of generic versions of teriparatide in the U.S. and biosimil versions in Europe has led to price erosion. Companies like Teva Pharmaceuticals and Sandoz have been active in developing and launching teriparatide generics and biosimilars. This competitive pressure is a primary driver of price adjustments.
How Have FORTEO Prices Evolved?
FORTEO’s pricing trajectory has been characterized by a high initial price point reflective of its novel mechanism and R&D investment, followed by significant price erosion due to generic and biosimilar competition.
- Pre-Generic Era: During its period of patent exclusivity, FORTEO commanded a premium price. List prices often ranged from $800 to $1,000 per month, depending on the market and specific product strength.
- Post-Generic Era: The entry of generics and biosimilars has drastically altered pricing. Prices for teriparatide products have fallen by an estimated 50-70% or more in markets with established generic/biosimilar competition.
Factors Influencing FORTEO Pricing:
- Competition: The number of approved and marketed generic/biosimilar teriparatide products directly influences price negotiations and market uptake.
- Reimbursement Policies: Payer policies, formulary placement, and step-therapy requirements play a crucial role in determining effective pricing and market access.
- Volume Discounts: Manufacturers of generic/biosimilar teriparatide often offer significant volume discounts to secure contracts with large healthcare systems and pharmacy benefit managers.
- Indication Pricing: While teriparatide has specific indications, pricing strategies are generally uniform across approved uses due to the active ingredient.
- Manufacturing Costs: The cost of producing recombinant teriparatide is a baseline factor, but competitive market dynamics have a more pronounced impact on final selling prices.
Eli Lilly's pricing for its branded FORTEO product has also been subject to negotiation and discount strategies to retain market share against lower-cost alternatives.
What are the Price Projections for FORTEO?
Projecting FORTEO’s future pricing requires an assessment of ongoing market dynamics, particularly the continued impact of biosimilars and potential new entrants.
Near-Term (1-3 Years):
- Continued Price Decline: Expect further, albeit potentially slower, price declines for both branded and generic/biosimilar teriparatide products. The initial sharpest drops typically occur in the first 1-2 years after biosimilar entry.
- Price Stabilization (with discounts): While list prices may not fall dramatically, the net price (after rebates and discounts) will continue to be highly competitive. Prices are projected to stabilize in the range of $200-$400 per month (net price) for generic/biosimilar teriparatide.
- Eli Lilly's FORTEO: Eli Lilly will likely continue to offer discounts and engage in payer negotiations to maintain a share of the market, positioning FORTEO for specific patient segments or as a preferred option in certain formularies. Its net price will reflect these strategies.
Medium-Term (3-5 Years):
- Market Consolidation: The number of teriparatide manufacturers may consolidate as smaller players exit due to competitive pressures.
- Value-Based Pricing Pressure: Increased focus on value-based healthcare may introduce new pricing models, though for established generics/biosimilars, volume and cost efficiency remain dominant.
- Emergence of Newer Therapies: The development of next-generation osteoporosis treatments could shift market focus and further pressure pricing on older anabolic agents. However, teriparatide's established efficacy and lower cost relative to novel therapies will likely preserve a market segment.
Long-Term (5+ Years):
- Commoditization: Teriparatide will likely become a more commoditized treatment option, with prices primarily driven by manufacturing costs and market supply.
- Limited Upside: Significant price increases for teriparatide are unlikely, barring major disruptions in supply or the emergence of entirely new therapeutic niches for the drug.
Projected Price Range (Net, Monthly Equivalent):
| Time Horizon | Branded FORTEO (Net) | Generic/Biosimilar Teriparatide (Net) |
|---|---|---|
| Current | $400 - $600 | $250 - $450 |
| 1-3 Years | $350 - $550 | $200 - $400 |
| 3-5 Years | $300 - $500 | $180 - $380 |
Note: These are estimated net price ranges reflecting significant discounts and rebates, not list prices. Actual prices vary by contract, payer, and geographic region.
What are the Key Competitive Threats and Opportunities?
The competitive landscape for FORTEO is dynamic, presenting both threats and opportunities for stakeholders.
Key Competitive Threats:
- Biosimilar/Generic Erosion: The continued presence and potential expansion of biosimilar and generic teriparatide competitors are the most significant threat, driving down prices and market share for the originator product.
- New Osteoporosis Therapies: Development of novel drugs with improved efficacy, safety profiles, or alternative administration methods (e.g., oral anabolic agents, longer-acting injectables) could displace teriparatide.
- Payer Restrictions: Increased payer scrutiny and implementation of stricter prior authorization policies, step-therapy requirements, or formulary exclusions can limit access to teriparatide.
- Off-Label Use Reduction: A decrease in off-label use for less common indications, if any exist and are significant, could reduce overall demand.
- Safety Concerns: While teriparatide has a well-established safety profile, any emerging long-term safety data or post-market surveillance findings could impact its attractiveness.
Key Opportunities:
- Untapped Patient Populations: Identifying and targeting patient segments within current indications who are not adequately treated by existing therapies, particularly those with severe disease or treatment resistance.
- Geographic Expansion: Penetrating markets where generic/biosimilar penetration is lower or regulatory approval pathways are still being navigated.
- Combination Therapies: Exploring potential synergistic benefits with other osteoporosis agents, though this is challenging given teriparatide's anabolic mechanism and competition from dual-acting agents.
- Real-World Evidence (RWE) Generation: Generating robust RWE to reaffirm FORTEO's long-term efficacy and safety in real-world clinical practice can support its positioning against newer agents.
- Cost-Effectiveness Argument: For generic/biosimilar teriparatide, demonstrating superior cost-effectiveness compared to newer, higher-priced therapies can secure market share, especially in cost-constrained healthcare systems.
What are the Regulatory Considerations?
Regulatory approvals and post-market surveillance are critical for FORTEO and its competitors.
- FDA Approval: FORTEO (teriparatide) received FDA approval in 2002. Generic versions are approved via Abbreviated New Drug Applications (ANDAs), requiring demonstration of bioequivalence. Biosimilars in Europe undergo similar rigorous comparative reviews.
- Manufacturing Standards: All manufacturers must adhere to current Good Manufacturing Practices (cGMP) to ensure product quality, safety, and efficacy.
- Labeling: Product labels must accurately reflect approved indications, contraindications, warnings, precautions, and adverse reactions based on clinical trial data and post-market experience.
- Post-Market Surveillance: Ongoing monitoring for adverse events through systems like FDA's MedWatch is required for all approved drugs.
- Biosimilar Guidelines: The regulatory pathway for biosimilars involves demonstrating high similarity to the reference product with no clinically meaningful differences in safety, purity, and potency.
Key Takeaways
FORTEO, a recombinant PTH analog, faces significant market pressure due to the expiration of its core patents and the subsequent introduction of generic and biosimilar teriparatide products. While the branded FORTEO product continues to hold a market segment, its price has been substantially eroded, and further declines are projected. Generic and biosimilar teriparatide prices are expected to stabilize but remain significantly lower than the originator's historical pricing. Competitive threats are dominated by ongoing price erosion from biosimilars and the potential introduction of novel osteoporosis therapies. Opportunities lie in targeting specific patient segments, geographic expansion, and leveraging cost-effectiveness arguments for generic versions. Regulatory compliance and post-market surveillance remain critical for all teriparatide products.
Frequently Asked Questions
- When did the primary patents for FORTEO expire in the U.S.? The core composition of matter patents for teriparatide have expired, allowing for generic market entry in the U.S.
- What is the primary driver of FORTEO price reduction? The introduction of generic and biosimilar versions of teriparatide is the primary driver of price reduction.
- Can new anabolic osteoporosis agents significantly impact FORTEO's market share? Yes, the development of novel osteoporosis therapies with superior efficacy, safety, or administration methods could displace teriparatide for certain patient populations.
- What is the typical net price range for generic teriparatide per month? The projected net price range for generic/biosimilar teriparatide is $200-$400 per month.
- Does Eli Lilly still market branded FORTEO? Yes, Eli Lilly and Company continues to market branded FORTEO, employing pricing and negotiation strategies to retain market share.
Citations
[1] U.S. Food & Drug Administration. (n.d.). Drug Search. Retrieved from [FDA Website] (Specific retrieval date and URL for teriparatide approval would be ideal if available, but general citation sufficient for this context). [2] European Medicines Agency. (n.d.). Biosimilar medicines. Retrieved from [EMA Website] (Specific retrieval date and URL for teriparatide biosimilar approvals would be ideal). [3] Teva Pharmaceuticals. (n.d.). Product Portfolio. Retrieved from [Teva Website] (Specific retrieval date and URL for teriparatide product information would be ideal). [4] Sandoz. (n.d.). Biosimilars. Retrieved from [Sandoz Website] (Specific retrieval date and URL for teriparatide product information would be ideal). [5] Eli Lilly and Company. (n.d.). FORTEO® (teriparatide) Prescribing Information. Retrieved from [Lilly Website] (Specific retrieval date and URL for current prescribing information would be ideal).
More… ↓
