Share This Page
Drug Price Trends for FLUOXETINE DR
✉ Email this page to a colleague
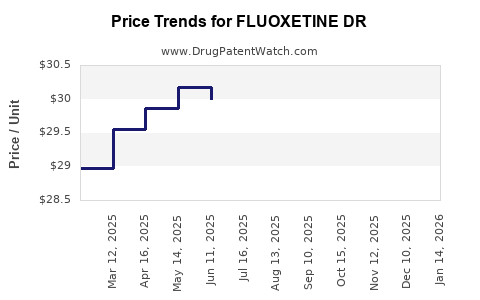
Average Pharmacy Cost for FLUOXETINE DR
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| FLUOXETINE DR 90 MG CAPSULE | 55111-0284-48 | 30.34304 | EACH | 2026-04-22 |
| FLUOXETINE DR 90 MG CAPSULE | 55111-0284-48 | 30.42075 | EACH | 2026-03-18 |
| FLUOXETINE DR 90 MG CAPSULE | 55111-0284-48 | 30.62109 | EACH | 2026-02-18 |
| FLUOXETINE DR 90 MG CAPSULE | 55111-0284-48 | 30.57496 | EACH | 2026-01-21 |
| FLUOXETINE DR 90 MG CAPSULE | 55111-0284-48 | 30.31863 | EACH | 2025-12-17 |
| FLUOXETINE DR 90 MG CAPSULE | 55111-0284-48 | 30.15529 | EACH | 2025-11-19 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
FLUOXETINE DR Market Analysis and Financial Projection
What is the Current Market Status of Fluoxetine DR?
Fluoxetine delayed-release (DR) is a formulation of the selective serotonin reuptake inhibitor (SSRI) primarily used for depression, obsessive-compulsive disorder (OCD), and bulimia nervosa. The drug's market is influenced by patents, generic entry, and competition from other antidepressants.
The patent for branded Fluoxetine was filed in 1974 and expired in 2001 in the US, allowing multiple generics to enter the market. The original product, marketed by Eli Lilly as Prozac, shifted to generic formulations post-patent expiration. Currently, several pharmaceutical manufacturers produce generic Fluoxetine DR, with branded sales limited mainly to ongoing formulations or new delivery variants.
How Does the Market Share Look for Fluoxetine DR?
The global antidepressant market was valued at approximately USD 16 billion in 2022 [1]. Fluoxetine maintains a significant share within SSRIs, which constitute about 65% of the antidepressant market. Within SSRIs, Fluoxetine accounted for roughly 30% of prescriptions in the US in 2022 [2].
In countries with mature markets, generic fluoxetine derivatives dominate prescriber preference due to lower costs. Market dominance shifts in regions with newer formulations or branded offerings. In the US, branded Fluoxetine sales have declined, with generic sales comprising nearly 90% of prescriptions as of 2022 [2].
What are the Key Factors Impacting Market Dynamics?
- Patent and exclusivity periods have long expired, leading to widespread generic access.
- The emergence of combination therapies and newer antidepressants, such as vortioxetine and vilazodone, compete with Fluoxetine.
- Prescriber preference and insurance formulary restrictions favor cost-effective generics.
- Regulatory policies can influence market access; in some regions, biosimilars or new formulations may impact volume.
- Off-label uses, including premature ejaculation, could create niche markets but lack substantial revenue potential.
What Are the Price Trends for Fluoxetine DR?
Prices for generic Fluoxetine in the US decreased sharply post-patent expiry. The wholesale acquisition cost (WAC) for a 30-day supply of 20 mg capsules averaged USD 4-8 in 2022, down from USD 30-40 when branded versions were dominant [3].
Pricing variations exist based on:
- Formulation: Delayed-release vs. immediate-release versions.
- Packaging size: Bulk or multi-month supplies reduce per-unit costs.
- Market access: Insurance coverage and pharmacy discounts further influence consumer prices.
Internationally, prices fluctuate based on healthcare system policies, with developing countries often paying lower prices, sometimes below USD 1 per month of therapy.
How Are Price Projections Evolving for Fluoxetine DR?
Projections depend on patent status, competition, and market acceptance:
- Patent expiry in most regions since early 2000s limits branded sales.
- Generic competition is expected to keep prices low.
- No significant pipeline innovations or reformulations are anticipated to alter pricing trends substantially.
- Market saturation suggests prices will stabilize or decline marginally over the next 5 years.
In the US, average retail prices for generics are expected to remain in the USD 4-8 range per month through 2028 [4].
International pricing could see slight reductions aligned with cost-of-living and procurement adjustments, especially in low- and middle-income countries.
Are There Emerging Developments Affecting Fluoxetine Market?
Development of new delivery systems such as transdermal patches or long-acting injectables remains limited and experimental. Several patent challenges and generic proliferation diminish incentives for innovation.
No significant regulatory approvals or clinical trials for novel formulations of Fluoxetine DR are evident as of 2023.
What are the Investment or R&D Opportunities?
Opportunities are constrained by patent expiry and commoditization. Niche markets such as pediatric or specific formulations (e.g., higher bioavailability) may present limited product differentiation but face strong generic competition.
Investors should monitor patent litigation or potential biosimilar entries. R&D efforts focus on combination drugs or personalized medicine but are not currently mainstream.
Key Takeaways
- Generic Fluoxetine dominates the global market following patent expiration in early 2000s.
- Prices in mature markets like the US average USD 4-8/month; in emerging markets, prices are often below USD 1.
- Market share is influenced by genericization, competing antidepressants, and prescriber preferences.
- Price stability expected through 2028, with minor downward trends.
- No significant pipeline or formulation innovations projected to alter market dynamics significantly.
FAQs
1. Will branded Fluoxetine return to the market?
Unlikely. Patent protection has long expired, and the market favors low-cost generics.
2. How does fluoxetine compete with newer antidepressants?
Mainly through cost advantages. Newer drugs may offer improved side effect profiles or additional efficacy but lack widespread market penetration due to higher costs.
3. Are there potential regulatory changes impacting prices?
Changes in healthcare policies, such as drug importation laws or biosimilar approvals, could influence prices but are unlikely to significantly alter the current market.
4. What are the main off-label uses influencing demand?
Premature ejaculation treatment and weight loss adjuncts have limited impact due to regulatory restrictions and market size.
5. How might future patent issues affect the market?
Patent challenges on formulations or new delivery systems could create temporary market opportunities but remain speculative.
References
[1] MarketsandMarkets. "Antidepressants Market," 2022.
[2] IQVIA. "Prescription Data," 2022.
[3] GoodRx. "Fluoxetine Prices," 2022.
[4] SSR Health. "Market Data," 2023.
More… ↓
