Drug Price Trends for FLUOROMETHOLONE
✉ Email this page to a colleague
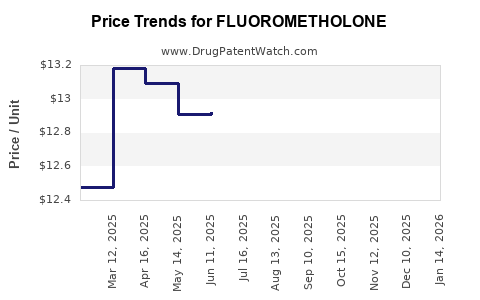
Average Pharmacy Cost for FLUOROMETHOLONE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| FLUOROMETHOLONE 0.1% EYE DROP | 60758-0880-05 | 12.92861 | ML | 2026-04-22 |
| FLUOROMETHOLONE 0.1% EYE DROP | 60219-1586-06 | 12.88236 | ML | 2026-04-22 |
| FLUOROMETHOLONE 0.1% EYE DROP | 60758-0880-10 | 12.88236 | ML | 2026-04-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Best Wholesale Price for FLUOROMETHOLONE
| Drug Name | Vendor | NDC | Count | Price ($) | Price/Unit ($) | Unit | Dates | Price Type |
|---|---|---|---|---|---|---|---|---|
| FLAREX 0.1% SUSP,OPH | Harrow Eye, LLC | 71776-0100-05 | 5ML | 93.59 | 18.71800 | ML | 2024-03-15 - 2029-03-14 | FSS |
| >Drug Name | >Vendor | >NDC | >Count | >Price ($) | >Price/Unit ($) | >Unit | >Dates | >Price Type |
