Share This Page
Drug Price Trends for FLUOCINONIDE-E
✉ Email this page to a colleague
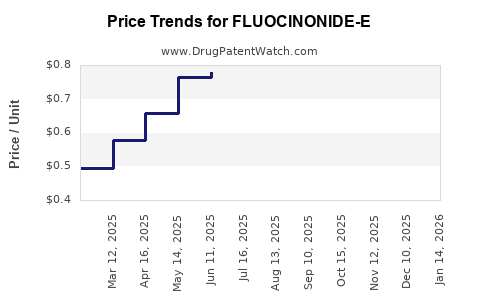
Average Pharmacy Cost for FLUOCINONIDE-E
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| FLUOCINONIDE-E 0.05% CREAM | 00093-0263-92 | 0.54727 | GM | 2026-04-22 |
| FLUOCINONIDE-E 0.05% CREAM | 00093-0263-30 | 0.67057 | GM | 2026-04-22 |
| FLUOCINONIDE-E 0.05% CREAM | 51672-1254-03 | 0.54727 | GM | 2026-04-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for Fluocinolone-E
What is Fluocinolone-E and Its Therapeutic Application?
Fluocinolone-E is a topical corticosteroid formulated with fluocinolone acetonide. It targets inflammatory skin conditions such as eczema, psoriasis, and dermatitis. The product combines fluocinolone acetonide with other excipients or active components, depending on formulation specifics.
Market Size and Key Drivers
The global corticosteroid topical market was valued at approximately $1.8 billion in 2022. It is projected to grow at a compound annual growth rate (CAGR) of 5% from 2023 to 2028, reaching $2.35 billion [1].
Key factors influencing growth include:
- Rising prevalence of chronic skin diseases.
- Increasing awareness and diagnosis.
- Expanded approval for new formulations.
- Prevalence of off-label use for itch and inflammation.
In particular, the corticosteroid segment accounts for over 60% of topical steroid sales.
Competitive Landscape
Market players include Pfizer, Galderma, Bausch Health, Mylan, and Sandoz. Generic formulations, such as Fluocinolone-E, have gained prominence due to patent expirations of branded products.
Notable formulations in the market:
- Elocon (Pfizer): Brand for fluocinolone acetonide topical.
- Generic equivalents: Widely available globally.
Market penetration is higher in North America and Europe, with emerging markets adopting gradually.
Regulatory Status and Approvals
Fluocinolone-E holds approval in multiple jurisdictions:
- United States: Approved as a prescription medication for inflammatory skin conditions.
- European Union: Registered as a Class IIa dermatological medicinal product.
- Other regions: Registration status varies.
Potential for expanded indications exists, contingent on clinical data approval.
Price Trends and Projection
Historical Price Data
- United States: Brand-name fluocinolone (Elocon) prices average $200–$250 per tube (15g).
- Generics: Available at approximately $50–$70 per tube, depending on supplier.
Current Market Prices
Generic fluocinolone-E’s current average retail price ranges from $45 to $70 per 15g tube, with variations based on distribution channels and geographic location.
Price Drivers
- Patent expiry of branded formulations.
- Entry of generics increases price competition.
- Distribution channels influence pricing, with pharmacies offering lower prices in bulk.
- Regulatory policies on drug pricing differ across countries.
Future Price Projections (Next 5 years)
- Generic prices are expected to stabilize around $40–$65 per tube as market competition intensifies.
- Price declines of up to 25% possible in mature markets due to increased generic penetration.
- Brand-name prices may remain steady or slightly decline in response to generic competition, with potential reductions of 10–15%.
Factors Influencing Price Movements
- Patent expiration for branded formulations, expected around 2025–2026.
- Increased subsidies or price regulations in specific markets.
- Expansion into emerging markets may temporarily suppress prices due to local regulatory requirements and competitive pressures.
Market Opportunities and Risks
Opportunities:
- Expansion into dermatological treatments with off-label applications.
- Development of combination products for enhanced efficacy.
- Penetration into emerging markets with favorable regulatory pathways.
Risks:
- Patent challenges delaying generic entry.
- Regulatory hurdles limiting approval in new regions.
- Market saturation in mature regions leading to downward price pressure.
Summary Table
| Aspect | Data | Notes |
|---|---|---|
| Current Market Size | ~$1.8 billion (2022) | Global corticosteroid topical market |
| CAGR (2023–2028) | 5% | Projected growth |
| Main Competitors | Pfizer, Galderma, Bausch Health, Mylan | Market share varies |
| Price of Generics | $45–$70 per 15g tube | Current market average |
| Price Forecast (2024–2028) | Stabilization at $40–$65 per tube in mature markets | Decline due to competition |
Key Takeaways
- Fluocinolone-E is within a competitive, growing market driven by dermatological demand.
- Patents for branded formulations are expiring around 2025–2026, facilitating generic entry.
- Prices for generics are expected to decline by approximately 25% over the next five years.
- Market expansion into emerging countries offers growth but pressure on prices.
- Patent challenges, regulatory approvals, and market saturation remain risks.
FAQs
1. When will patent expiration occur for branded fluocinolone formulations?
Most patents are set to expire between 2025 and 2026, opening the market for generic alternatives.
2. What countries have the largest market for topical corticosteroids?
North America and Europe represent the largest markets, with significant growth potential in Asia-Pacific.
3. How does regulatory policy impact pricing?
Price controls, reimbursement policies, and approval processes influence the final consumer price, especially in markets with price caps.
4. Are there opportunities for combination drugs involving fluocinolone-E?
Yes, combination formulations targeting multiple dermatological conditions may enhance treatment efficacy and expand market size.
5. What are the main risks to price stability for Fluocinolone-E?
Patent litigation, regulatory delays, aggressive generic entry, and market saturation pose risks to maintaining current pricing.
References
- Statista. (2023). Corticosteroid market size and growth projections. https://www.statista.com
More… ↓
