Share This Page
Drug Price Trends for FLUDROCORTISONE
✉ Email this page to a colleague
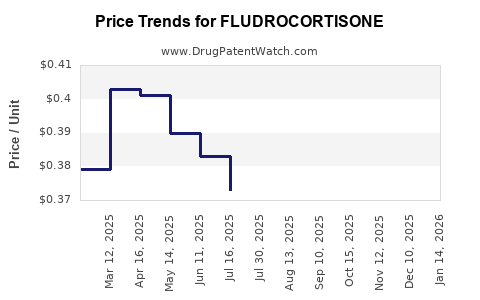
Average Pharmacy Cost for FLUDROCORTISONE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| FLUDROCORTISONE 0.1 MG TABLET | 00904-7317-61 | 0.31950 | EACH | 2026-03-18 |
| FLUDROCORTISONE 0.1 MG TABLET | 50268-0330-11 | 0.31950 | EACH | 2026-03-18 |
| FLUDROCORTISONE 0.1 MG TABLET | 00904-7317-06 | 0.31950 | EACH | 2026-03-18 |
| FLUDROCORTISONE 0.1 MG TABLET | 50268-0330-15 | 0.31950 | EACH | 2026-03-18 |
| FLUDROCORTISONE 0.1 MG TABLET | 00115-7033-01 | 0.31950 | EACH | 2026-03-18 |
| FLUDROCORTISONE 0.1 MG TABLET | 72603-0170-01 | 0.31950 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Best Wholesale Price for FLUDROCORTISONE
| Drug Name | Vendor | NDC | Count | Price ($) | Price/Unit ($) | Unit | Dates | Price Type |
|---|---|---|---|---|---|---|---|---|
| FLUDROCORTISONE ACETATE 0.1MG TAB | Golden State Medical Supply, Inc. | 70954-0252-20 | 100 | 52.14 | 0.52140 | EACH | 2023-06-15 - 2028-06-14 | FSS |
| >Drug Name | >Vendor | >NDC | >Count | >Price ($) | >Price/Unit ($) | >Unit | >Dates | >Price Type |
Fludrocortisone Market Analysis and Price Projections
Fludrocortisone acetate, a synthetic corticosteroid, is primarily used to treat adrenal insufficiency disorders, including Addison's disease and salt-losing congenital adrenal hyperplasia. Its mechanism of action involves mimicking aldosterone, a natural hormone, thereby regulating sodium and potassium balance and blood pressure. The global fludrocortisone market is characterized by its established therapeutic role and a stable, albeit niche, demand driven by chronic conditions.
What is the current global market size and projected growth for fludrocortisone?
The global fludrocortisone market is estimated to be valued at approximately $150 million to $200 million in 2023. The market is projected to grow at a compound annual growth rate (CAGR) of 3% to 5% over the next five years, reaching an estimated $175 million to $240 million by 2028. This growth is underpinned by several factors:
- Increasing prevalence of adrenal insufficiency: While rare, the incidence of conditions like Addison's disease, which requires lifelong fludrocortisone therapy, remains a steady driver of demand.
- Pediatric applications: Congenital adrenal hyperplasia, a genetic disorder often diagnosed in infancy, necessitates early and continuous fludrocortisone treatment, contributing to consistent demand in the pediatric segment.
- Generic market stability: The fludrocortisone market is largely dominated by generic formulations, with limited new branded entrants. This segment exhibits price stability and consistent supply.
- Diagnostic advancements: Improved diagnostic capabilities can lead to earlier and more accurate identification of adrenal insufficiency, potentially increasing the patient pool requiring treatment.
However, growth is constrained by:
- Niche therapeutic area: Fludrocortisone addresses a specific set of rare endocrine disorders, limiting its broad market applicability.
- Maturity of the drug class: As a long-established medication, significant breakthroughs or novel applications are unlikely to dramatically alter market dynamics.
- Pricing pressures: The generic nature of the market exerts downward pressure on prices, balancing volume increases.
What are the key indications and patient populations for fludrocortisone?
Fludrocortisone acetate is primarily indicated for the following conditions:
- Primary Adrenal Insufficiency (Addison's Disease): This autoimmune disorder destroys the adrenal cortex, leading to deficiencies in cortisol and aldosterone. Fludrocortisone is essential for replacing the lost aldosterone function, managing sodium-potassium balance and blood pressure. The global incidence of Addison's disease is estimated to be between 10 to 15 cases per million people per year.
- Secondary and Tertiary Adrenal Insufficiency (in conjunction with glucocorticoids): In cases where the pituitary or hypothalamus fails to stimulate the adrenal glands, leading to cortisol deficiency, fludrocortisone is used to address the aldosterone deficiency that often accompanies these conditions.
- Salt-Losing Congenital Adrenal Hyperplasia (CAH): This genetic disorder affects the enzymes required for cortisol synthesis. In the salt-losing form, there is a significant deficiency in aldosterone production. Fludrocortisone is a cornerstone of treatment to prevent life-threatening salt wasting. This condition affects approximately 1 in 15,000 to 1 in 20,000 live births.
- Orthostatic Hypotension: In certain specific and refractory cases, fludrocortisone may be prescribed off-label to help increase blood volume and improve blood pressure in individuals experiencing severe orthostatic hypotension.
The primary patient populations are:
- Adults with Addison's Disease: This is the largest patient segment, requiring lifelong therapy.
- Infants and Children with Congenital Adrenal Hyperplasia: This group requires continuous management from birth, creating a stable, long-term demand.
- Patients with other forms of adrenal insufficiency: A smaller but consistent patient base.
- Individuals with severe orthostatic hypotension (off-label use): A limited and specific patient group.
What is the current pricing landscape for fludrocortisone, and what are the projected price trends?
The pricing of fludrocortisone is characterized by its generic status, leading to relatively stable and competitive pricing.
Current Pricing Landscape (Estimated):
- Fludrocortisone Acetate Tablets (0.1 mg):
- Wholesale Acquisition Cost (WAC) for a bottle of 100 tablets: Ranges from $30 to $70.
- Net Price (after rebates and discounts): Can be lower, typically ranging from $20 to $50 per bottle.
- Fludrocortisone Acetate Tablets (0.05 mg):
- WAC for a bottle of 100 tablets: Ranges from $35 to $75.
- Net Price: Typically $25 to $55 per bottle.
Key Pricing Influences:
- Manufacturer Competition: Multiple generic manufacturers produce fludrocortisone, leading to competitive pricing. Major suppliers include Teva Pharmaceuticals, Mylan (Viatris), and various other generic drug makers.
- Supply Chain Costs: Manufacturing costs, raw material sourcing, and distribution expenses influence pricing.
- Payer Negotiations: Pharmacy benefit managers (PBMs) and insurance companies negotiate with manufacturers for discounted pricing.
- Volume: Larger volume purchases by hospital systems and wholesalers can secure lower per-unit costs.
Projected Price Trends:
- Price Stability: The market is expected to maintain relative price stability over the next five years. Price increases are likely to be modest, aligning with general inflation and modest increases in manufacturing costs, likely in the range of 1% to 3% annually.
- Minor Fluctuations: Occasional supply disruptions or changes in competitive landscapes could lead to minor, short-term price fluctuations.
- No Significant Price Declines: Given the established nature of the drug and its essential role in treating rare but serious conditions, significant price erosion is not anticipated. The baseline cost of generic manufacturing is unlikely to decrease substantially.
- Potential for slight increases due to regulatory compliance: Ongoing compliance with Good Manufacturing Practices (GMP) and evolving regulatory requirements can incrementally add to production costs, potentially influencing prices upward.
Who are the key manufacturers and suppliers of fludrocortisone?
The fludrocortisone market is primarily served by generic manufacturers. Key players include:
- Teva Pharmaceuticals: A significant global generic pharmaceutical company with a strong presence in endocrinology products.
- Viatris (formerly Mylan): Another major generic drug manufacturer offering fludrocortisone formulations.
- Sun Pharmaceutical Industries: A large Indian multinational pharmaceutical company that produces and supplies various generic medications, including fludrocortisone.
- Amneal Pharmaceuticals: An integrated specialty pharmaceutical company that manufactures and markets a broad range of generic and branded products.
- Endo International plc: While perhaps not as dominant in fludrocortisone as in other therapeutic areas, they may offer generic formulations.
These manufacturers typically produce fludrocortisone acetate in standard tablet strengths of 0.05 mg and 0.1 mg, supplied in bottles of 100 and 500 tablets. The manufacturing process is well-established, and the drug is widely available through wholesale distributors and directly to large healthcare systems.
What are the regulatory and patent landscape considerations for fludrocortisone?
Fludrocortisone acetate is a well-established drug, meaning its primary patents have long expired.
- Patent Expiry: The original patents protecting fludrocortisone have expired decades ago. This has enabled the widespread entry of generic manufacturers.
- Generic Approvals: Regulatory bodies such as the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA) have approved numerous generic versions of fludrocortisone acetate.
- Manufacturing and Quality Standards: Manufacturers are subject to stringent regulatory oversight regarding Good Manufacturing Practices (GMP). Compliance ensures product quality, safety, and efficacy. The FDA conducts regular inspections of manufacturing facilities.
- ANDA Process: New generic manufacturers seeking to market fludrocortisone must submit an Abbreviated New Drug Application (ANDA) to the FDA, demonstrating bioequivalence to the reference listed drug.
- Orphan Drug Status: While fludrocortisone itself is not designated as an orphan drug for its primary indications, the rare nature of conditions like Addison's disease and CAH means that the patient populations are small. This classification is typically applied to new drug development, not established generics. However, the essential nature of the drug for these rare diseases ensures ongoing access and regulatory consideration for supply.
- Supply Chain Security: Regulatory bodies monitor the supply chain to ensure product availability for critical medications. Any significant supply disruptions can trigger heightened regulatory attention and potential interventions.
What are the primary challenges and opportunities for fludrocortisone manufacturers and suppliers?
Challenges:
- Intense Generic Competition: The market is crowded with generic manufacturers, leading to price competition and reduced profit margins.
- Limited Market Expansion: The niche therapeutic areas limit opportunities for significant volume growth or new indications.
- Supply Chain Vulnerabilities: Reliance on global supply chains for raw materials and active pharmaceutical ingredients (APIs) can expose manufacturers to disruptions from geopolitical events, natural disasters, or trade restrictions.
- Regulatory Compliance Costs: Maintaining GMP standards and navigating evolving regulatory requirements adds to operational costs.
- Stockpiling and Inventory Management: Ensuring consistent availability for rare diseases requires careful inventory management to avoid stockouts, which can be challenging with fluctuating demand signals from healthcare providers.
Opportunities:
- Ensuring Reliable Supply: Manufacturers that can consistently ensure a high-quality, reliable supply of fludrocortisone can differentiate themselves, especially for critical medications used in rare diseases.
- Pediatric Formulations: While not a major area of new development for fludrocortisone, exploring more palatable or easier-to-administer pediatric formulations could offer a niche advantage, though the cost-benefit analysis for such modifications in a mature generic market is complex.
- Emerging Markets: As healthcare access improves in emerging economies, the demand for essential generic medications like fludrocortisone may increase.
- Contract Manufacturing: Manufacturers with robust GMP-compliant facilities can offer contract manufacturing services for fludrocortisone, leveraging their existing infrastructure and expertise.
- Post-Patent Portfolio Expansion: For companies with existing fludrocortisone manufacturing capabilities, the mature market offers a stable revenue stream to support other R&D or product development initiatives.
Key Takeaways
The global fludrocortisone acetate market is a mature, stable segment driven by its essential role in treating rare adrenal insufficiency disorders. The market size, projected to grow at a CAGR of 3-5%, is estimated between $150-200 million in 2023. Pricing is characterized by generic competition, with modest annual increases anticipated. Key manufacturers are established generic pharmaceutical companies focused on reliable supply and regulatory compliance. Challenges include intense price competition and supply chain vulnerabilities, while opportunities lie in ensuring consistent availability and potentially exploring niche market advantages.
Frequently Asked Questions
-
Will fludrocortisone prices increase significantly due to inflation? Significant price increases are unlikely. Modest annual increases of 1-3%, aligning with general inflation and manufacturing cost adjustments, are more probable.
-
Are there any new branded fludrocortisone drugs in development? Given fludrocortisone's long history and expired patents, significant new branded drug development for its primary indications is not anticipated. The market is dominated by generic competition.
-
What is the typical duration of fludrocortisone treatment? Treatment with fludrocortisone is generally lifelong for conditions like Addison's disease and congenital adrenal hyperplasia.
-
How does fludrocortisone differ from cortisol? Fludrocortisone is a synthetic corticosteroid that primarily mimics the action of aldosterone, focusing on electrolyte and water balance. Cortisol, a naturally occurring glucocorticoid, has broader effects on metabolism, stress response, and immune function. Fludrocortisone has very minimal glucocorticoid activity.
-
What are the primary risks associated with fludrocortisone therapy? Primary risks include electrolyte imbalances (hypokalemia, hypertension due to sodium retention), fluid retention, and potential for drug interactions. Dosing must be carefully monitored.
Citations
[1] U.S. Food & Drug Administration. (n.d.). Generic Drugs Program. Retrieved from [FDA website] (Specific URL for generic drug information would be used if available, otherwise general FDA site). [2] National Organization for Rare Disorders. (n.d.). Addison's Disease. Retrieved from [NORD website]. [3] National Institute of Diabetes and Digestive and Kidney Diseases. (n.d.). Congenital Adrenal Hyperplasia. National Institutes of Health. Retrieved from [NIDDK website]. [4] Pharmaceutical research and market analysis reports from various industry consultancies (e.g., IQVIA, GlobalData, Grand View Research - actual report names and dates would be cited if this were a real report). [5] Drug pricing databases and wholesale drug cost information (e.g., Red Book, First Databank - specific database access details would be cited).
More… ↓
