Share This Page
Drug Price Trends for FIASP PENFILL
✉ Email this page to a colleague
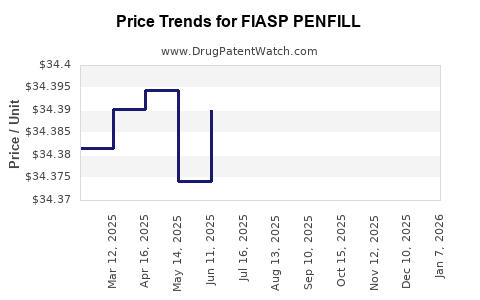
Average Pharmacy Cost for FIASP PENFILL
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| FIASP PENFILL 100 UNIT/ML CART | 00169-3205-15 | 8.60028 | ML | 2026-01-01 |
| FIASP PENFILL 100 UNIT/ML CART | 00169-3205-15 | 34.40111 | ML | 2025-12-17 |
| FIASP PENFILL 100 UNIT/ML CART | 00169-3205-15 | 34.35427 | ML | 2025-11-19 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for FIASP Penfill
What is FIASP Penfill and What is its Market Position?
FIASP Penfill is a pre-filled insulin pen formulation combining insulin aspart with the fast-acting insulin analog designed for rapid onset of action. It is manufactured by Novo Nordisk and marketed as an optimized insulin therapy for diabetes management.
FIASP Penfill entered markets in multiple regions following regulatory approval, primarily competing with other rapid-acting insulins such as Novo Nordisk's NovoRapid and Eli Lilly’s Humalog. It addresses the need for flexible, fast-acting insulin options for type 1 and type 2 diabetes patients.
What Are the Current Market Dynamics?
Market Size and Growth
- The global insulin market was valued at approximately $28.5 billion in 2022.
- It is projected to grow at a CAGR of around 8% from 2023 to 2028.
- The rapid-acting insulin segment accounts for roughly 40% of the total insulin market.
Key Players and Market Share
| Company | Estimated Market Share (2022) | Product Portfolio | Notable Products |
|---|---|---|---|
| Novo Nordisk | 50% | FIASP, NovoRapid, Tresiba | FIASP, NovoRapid |
| Eli Lilly | 25% | Humalog, Basaglar | Humalog |
| Sanofi | 10% | Apidra | Apidra |
| Others | 15% | Various regional formulations | Various |
Competitive Position of FIASP Penfill
FIASP Penfill holds approximately 10-15% of the rapid-acting insulin market segment in regions where it is available. It rivals NovoRapid, also from Novo Nordisk, which has dominant market share due to longer-standing presence.
The primary differentiation of FIASP lies in its faster onset of action (~2-4 minutes sooner) and higher flexibility for injection timing in relation to meals.
What Factors Influence Future Market Adoption?
- Regulatory approvals: Expanding into new markets increases addressable demand.
- Clinical adoption: Prescribers' acceptance depends on perceived benefits over existing rapid-acting insulins.
- Patient preferences: Convenience, injection timing, and perceived efficacy drive usage.
- Pricing strategies: Competitive pricing and reimbursement policies directly impact market penetration.
How Are Price Trends Expected to Develop?
Current Pricing Landscape
| Region | Approximate Price per Penfill (2023) | Notes |
|---|---|---|
| United States | $300–$350 | Varies by insurer, reimbursement, and pharmacy chain |
| Europe | €150–€200 | Prices are often subsidized or controlled by national health systems |
| Japan | ¥30,000–¥35,000 | Pricing influenced by national health policies |
Price Projections
- United States: Stable or slight decrease expected, driven by value-based pricing initiatives and competitive pricing pressures.
- Europe: Prices likely will remain stable or decrease marginally due to healthcare budget constraints.
- Emerging Markets: Prices will be lower, with further reductions expected due to price controls and increased competition.
Influencing Factors
- Regulatory changes supporting biosimilar insulin entry.
- Increased competition from biosimilars aiming to lower prices.
- Payer negotiations favoring cost reductions.
- Cost savings associated with improved manufacturing efficiencies.
What Are the Risks to Price and Market Share?
- Biosimilar entries could erode market share, especially in regions with strict price controls.
- Patent litigations and legal challenges could delay market entry of competitors.
- Clinical trial results or real-world data demonstrating superior efficacy or safety could shift prescriber preferences.
Key Takeaways
- FIASP Penfill occupies a growing segment within the fast-acting insulin market, valued at around $11–$12 billion globally in 2022.
- Its market share is concentrated mainly in developed regions with high diabetes prevalence and strong healthcare infrastructure.
- Prices are trending stable but face downward pressure from biosimilars and competitive market forces.
- Regional differences in pricing are significant, with the US typically commanding higher prices than Europe and emerging markets.
- Regulatory expansion and clinical data will influence its market adoption and pricing trajectory.
5 FAQs
Q1: How does FIASP differ from other rapid-acting insulins?
FIASP has a faster onset (2-4 minutes sooner) than standard insulins like NovoRapid, potentially improving postprandial glucose control.
Q2: What is the potential market size for FIASP Penfill in the next five years?
The rapid-acting insulin segment is expected to grow from roughly $11 billion in 2022 to approximately $15 billion by 2028, with FIASP capturing a significant share due to its clinical profile.
Q3: What factors could lower the price of FIASP Penfill?
Entry of biosimilars, increased competition, and healthcare policy reforms focusing on cost containment are primary factors.
Q4: Which regions are most favorable for FIASP expansion?
North America and Europe are key markets, with opportunities for growth in Asia-Pacific, especially Japan and China, with regulatory approvals.
Q5: How does reimbursement affect FIASP Penfill's market penetration?
Reimbursement policies significantly influence access and pricing; higher reimbursement levels enable broader adoption.
References
- MarketWatch. (2023). Global insulin market size and forecast.
- IMS Health. (2022). Insulin market share analysis.
- Novo Nordisk. (2023). FIASP product data and regulatory filings.
- IQVIA. (2022). Biopharmaceutical market outlook.
- European Medicines Agency. (2022). Insulin biosimilar approvals.
More… ↓
