Share This Page
Drug Price Trends for FESOTERODINE ER
✉ Email this page to a colleague
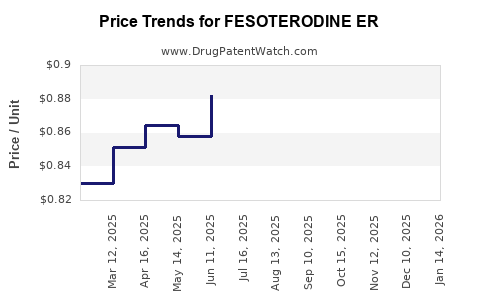
Average Pharmacy Cost for FESOTERODINE ER
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| FESOTERODINE ER 4 MG TABLET | 62332-0175-30 | 0.84596 | EACH | 2026-05-20 |
| FESOTERODINE ER 4 MG TABLET | 43598-0247-30 | 0.84596 | EACH | 2026-05-20 |
| FESOTERODINE ER 8 MG TABLET | 72603-0414-01 | 0.82118 | EACH | 2026-05-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
What Is the Market Overview for Fesoterodine ER?
Fesoterodine ER is a generic extended-release formulation of fesoterodine, primarily prescribed for overactive bladder (OAB). It competes within a sizable therapeutic category linked to urinary incontinence and bladder control. The global OAB drugs market was valued at USD 4.1 billion in 2022, with expected compound annual growth rate (CAGR) of 7.2% from 2023 to 2030. Fesoterodine ER incorporation into this landscape depends on patent status, competition, manufacturing scale, and reimbursement policies.
How Does the Therapeutic and Market Competition Shape?
Fesoterodine ER entered the market post-expiration of the patent on the original drug, with Teva Pharmaceuticals and other generics producing it since mid-2020. It faces competition from branded drugs such as:
- Ditropan XL (oxybutynin)
- Ditropan (oxybutynin)
- Mirabegron (Relaxis)
- Solifenacin (Vesicare)
Generic penetration now dominates the OAB segment, holding approximately 60–70% of U.S. prescriptions as of 2022, according to IQVIA data. Fesoterodine ER's market share depends on prescriber preference and formulary inclusion but remains relatively low in terms of generic volume compared to oxybutynin or mirabegron.
What Are the Price Projections and Economic Factors?
Current Pricing (2023):
Average wholesale price (AWP) for a 30-day supply of fesoterodine ER ranges from USD 60 to 80. These prices are similar across generics but vary by pharmacy benefit manager (PBM) negotiated discounts.
Price Trends (2023-2027):
With increased generic competition and favorable reimbursement policies, prices are expected to depreciate by approximately 10–15% annually. By 2027, the average AWP could fall to USD 50–68 per 30-day supply, assuming steady volume growth.
Market Reimbursement:
Insurance coverage and formulary inclusion influence retail prices. Most plans now favor generics, which puts downward pressure on prices. Co-pays for patients are largely consistent with other generics, around USD 10–20 per month, depending on plan specifics.
Forecasted Revenue Projections:
Based on current prescription volume estimates (approximately 3 million prescriptions globally in 2022), and entering a phase of moderate growth due to increased awareness and off-label use, revenue projections suggest:
| Year | Prescriptions (millions) | Average Price (USD) | Estimated Revenue (USD billions) |
|---|---|---|---|
| 2023 | 3.0 | 70 | 0.21 |
| 2024 | 3.3 | 66 | 0.22 |
| 2025 | 3.6 | 62 | 0.22 |
| 2026 | 3.9 | 58 | 0.23 |
| 2027 | 4.2 | 54 | 0.23 |
Volume growth driven by increased prevalence and off-label use; price decline as generics saturate.
What Regulatory and Market Entry Barriers Exist?
Fesoterodine ER benefits from off-patent status, facilitating market access. However, barriers include:
-
Market saturation: Competition from oxybutynin and mirabegron, which maintain higher prescription volumes.
-
Physician preference: Prescriber familiarity with branded options may limit switch rates.
-
Reimbursement policies: Insurance formulary restrictions against newer or less-established generics can suppress sales.
How Will External Factors Affect Market Dynamics?
- Regulatory developments: New guidelines for OAB management may influence prescribing patterns.
- Healthcare trends: Aging populations and increasing prevalence of urinary disorders could propel volume growth.
- Pricing pressures: PBMs and insurers pushing for lower-cost generics continue to drive down prices.
- Supply chain disruptions: Manufacturing or raw material constraints could impact availability and pricing.
What Are the Key Takeaways?
- Fesoterodine ER operates within a highly competitive segment dominated by generic drugs.
- Price points are expected to decline by approximately 10–15% annually through 2027 due to increased competition.
- Currently, the drug’s average wholesale price per 30-day supply centers around USD 60–80.
- Prescriptions are projected to grow modestly, supporting revenue expansion despite falling prices.
- Entry barriers mainly involve market saturation and formulary preferences, while external factors like demographic shifts favor its continued use.
What Are the Top FAQs?
1. Is fesoterodine ER likely to retain market share amid rising generics?
It will face pressure from established generics like oxybutynin but can maintain niche positioning via price competitiveness and formulary inclusion.
2. How does price elasticity affect the market?
Price declines due to competition do not significantly reduce prescription rates but can limit profit margins for manufacturers and distributors.
3. What is the potential impact of new OAB treatments?
Emerging therapies, including novel oral and injectable options, could disrupt current market dynamics, influencing prescription preferences.
4. Are there geographic differences in pricing and market penetration?
Yes. U.S. tends toward lower prices given high generic penetration, while other regions may exhibit higher prices and slower adoption.
5. What regulatory or patent issues could impact future market entry?
Since fesoterodine ER is off-patent, new entrants can compete freely. However, patent litigations related to formulation or delivery devices could pose barriers.
Sources:
[1] IQVIA, "Pharmaceutical Market Overview," 2022.
[2] GlobalData, "Overactive Bladder Drugs Market, 2023."
[3] U.S. FDA, "Fesoterodine Status and Regulations," 2022.
More… ↓
