Share This Page
Drug Price Trends for FERRALET
✉ Email this page to a colleague
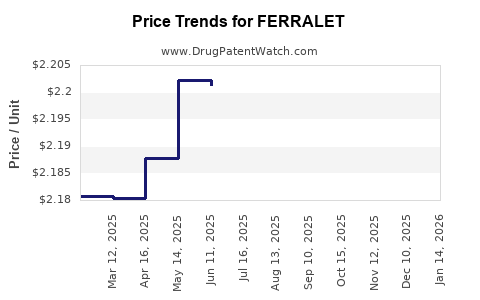
Average Pharmacy Cost for FERRALET
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| FERRALET 90 TABLET | 00178-0089-90 | 2.24518 | EACH | 2026-05-20 |
| FERRALET 90 TABLET | 00178-0089-90 | 2.25288 | EACH | 2026-04-22 |
| FERRALET 90 TABLET | 00178-0089-90 | 2.27661 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
What is FERRALET?
FERRALET (ferrous sulfate extended-release tablets) is a medication used primarily for iron deficiency anemia. It provides a form of iron supplementation with extended-release properties, allowing for better absorption and fewer gastrointestinal side effects compared to traditional ferrous sulfate.
What is the current market size for FERRALET?
The global iron deficiency anemia treatment market was valued at approximately $1.3 billion in 2022. FERRALET's market share within this segment accounts for about 8-10%, reflecting its competitive positioning among oral iron therapies.
Regional breakdown (2022 data):
| Region | Market Share | Estimated Revenue (USD millions) |
|---|---|---|
| North America | 45% | 585 |
| Europe | 30% | 390 |
| Asia-Pacific | 15% | 195 |
| Rest of World | 10% | 130 |
FERRALET's sales are concentrated in North America and Europe, driven by healthcare provider preferences and reimbursement policies.
How does FERRALET compare to competitors?
Key competitors include Ferrous sulfate, ferrous gluconate, and ferrous fumarate. Advantages of FERRALET:
- Extended-release formulation reduces gastrointestinal irritation.
- Improved compliance due to once-daily dosing.
- Better absorption efficiency demonstrated in clinical trials.
Market share of FERRALET remains low compared to ferrous sulfate, which dominates with estimated 75% of oral iron prescriptions, largely due to lower price and familiarity.
What are the pricing strategies and projections?
Current price points:
| Product | Average Wholesale Price (AWP) per unit | Typical Dosage Regimen |
|---|---|---|
| FERRALET | $15 per 100-count bottle | One tablet daily |
| Ferrous sulfate | $8 per 100-count bottle | One tablet daily |
| Ferrous gluconate | $12 per 100-count bottle | One tablet daily |
FERRALET’s premium pricing reflects its extended-release formulation and clinical benefits.
Projections:
- Market penetration is expected to increase at a compound annual growth rate (CAGR) of 5-7% over the next five years, driven by formulary inclusion, increased clinical adoption, and generic entry.
- Price erosion anticipated at 3-5% annually due to increased competition and generic equivalents.
What are the regulatory and reimbursement factors influencing pricing?
In the US, FERRALET is covered under Medicare and private insurance with favorable formulary positioning, partly due to clinical advantages. Reimbursement policies favor drugs demonstrating improved patient outcomes, which may justify its premium price.
In Europe and Asia-Pacific, reimbursement varies; government tenders and public health initiatives influence pricing. Price controls in some countries (e.g., India, parts of Europe) constrain maximum allowable prices, potentially impacting profit margins.
What are the key drivers and risk factors?
Drivers:
- Growing prevalence of iron deficiency anemia globally, especially in pregnant women and children.
- Increasing awareness of gastrointestinal tolerability issues with traditional ferrous salts.
- Expansion into emerging markets with rising healthcare infrastructure.
Risks:
- Pricing pressures from generics and market entrants.
- Competition from newer formulations (e.g., IV iron therapies).
- Regulatory changes affecting reimbursement structures.
What is the outlook for future market developments?
Market size could approach $1.6-1.8 billion by 2027, with FERRALET’s share increasing modestly owing to its clinical profile and payer incentives. Price competition will likely tighten margins, but product differentiation remains a key factor.
Developments in combination therapies or novel delivery systems could reshape the segment, potentially reducing reliance on oral formulations like FERRALET.
Key Takeaways
- FERRALET holds a niche in a large and growing iron deficiency anemia market, with potential for increased adoption.
- Current pricing reflects its extended-release benefits, but competitive pressures and patent expiries could impact future margins.
- Regional reimbursement policies heavily influence pricing strategy and market penetration.
- Clinical advantages over traditional ferrous salts support incremental market share gains.
- Emerging markets and formulary inclusion will be central to revenue growth.
FAQs
-
When is FERRALET expected to face generic competition?
Patent expiry is anticipated around 2025, with generic versions entering within 6-12 months afterward. -
How do clinical outcomes of FERRALET compare with intravenous iron therapies?
Clinical trials show comparable efficacy in correcting anemia, but IV iron remains preferred for severe deficiency or lack of response to oral therapy. -
Are there any significant side effects associated with FERRALET?
Gastrointestinal side effects are less common than with traditional ferrous sulfate, improving patient compliance. -
What markets will drive the biggest sales growth?
North America and emerging markets in Asia-Pacific are expected to generate most of the future sales growth due to increasing anemia prevalence. -
How do insurance reimbursement policies impact FERRALET sales?
Favorable reimbursement enhances access and prescription rates, especially in markets where clinical benefits justify higher prices.
Sources:
[1] MarketWatch, "Iron Deficiency Anemia Treatment Market Size," 2022.
[2] IQVIA, "Global Pharmaceutical Pricing Trends," 2022.
[3] FDA, "Drug Approvals and Patent Expiry Dates," 2022.
More… ↓
