Last updated: February 14, 2026
What is felbamate and how is it currently positioned in the market?
Felbamate is an antiepileptic drug approved for use in the United States and Europe for treatment-resistant partial seizures and Lennox-Gastaut syndrome. It was introduced in the late 1980s. Its market presence diminished after reports of serious adverse effects, including aplastic anemia and hepatic failure, led to restricted use and a decline in prescriptions. The drug's market has historically been limited to specialist neurologists and epilepsy centers, with low overall sales volume.
How is the regulatory landscape evolving for felbamate?
Recent developments include renewed interest in safer derivatives or formulations that mitigate serious side effects. Regulatory agencies like the FDA have maintained warnings but are open to new formulations or combination therapies. New clinical trials focusing on safety could influence future approvals or expanded indications, potentially reshaping its market positioning.
What are the current demand drivers for felbamate?
Demand remains stable primarily in niche markets: patients with treatment-resistant epilepsy unable to tolerate other therapies. Off-label use is virtually nonexistent. The drug's toxicology profile limits widespread adoption, constraining demand growth. However, ongoing research into safer derivatives or combination therapies could catalyze demand expansion.
What are the key competitive factors influencing felbamate's market?
Safety profile and efficacy define its market niche. Alternatives include other antiepileptic drugs such as valproate, lamotrigine, and topiramate, which offer similar efficacy with improved safety profiles. The high toxicity risks associated with felbamate limit its use, giving it a minimal share among broader epilepsy treatments. Development of new drugs with improved safety could further erode its market share.
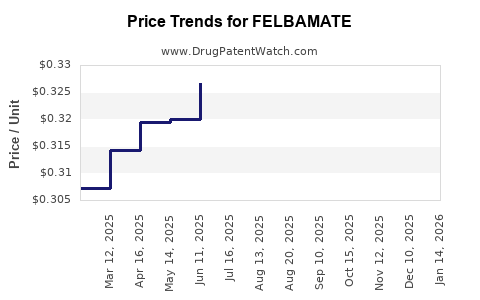
What are the prevailing price points for felbamate?
In the United States, the generic form of felbamate typically costs between $10 and $30 per 100 mg tablet, translating to approximately $200 to $600 per month for a standard dose (e.g., 1,200 mg/day). Brand-name options are not available; the drug is primarily marketed as a generic. Prices in Europe are comparable, though regional pricing variations exist due to regulatory and distribution factors.
What are the potential future price projections for felbamate?
Given the drug's safety concerns, market size is expected to remain limited, with low volatility. Price stability may persist unless new formulations or indications gain approval. If new safety profiles are introduced or approved, prices could increase slightly due to the value of improved safety, but widespread adoption is unlikely unless safety improves substantially.
How might patent or regulatory changes influence the outlook?
Felbamate's original patent expired decades ago, leaving it open for generic manufacturing. No new patents are in place. Regulatory relaxations or re-approvals for newer formulations could generate a brief price increase or trial use, but existing safety issues are a primary barrier. Market entry of rival drugs with better safety profiles will continue to pressure felbamate's market share and pricing.
What are the key market risks for felbamate?
The primary risks include adverse safety reports, regulatory restrictions, and dominance of newer, safer antiepilestics. The limited market size constrains revenue growth potential. Pricing pressure from generics and emerging therapies will likely continue. Clinical advances targeting safer options may render felbamate obsolete in many indications.
What is the outlook for segment-specific growth?
Niche expansion depends on clinical trial success for safer derivatives or combination therapies. If such drugs demonstrate improved safety with comparable efficacy, felbamate’s share would decline further. No significant segment-specific growth is anticipated unless safety concerns are substantially addressed or special regulatory approvals are granted.
Key Takeaways
- Felbamate remains a niche drug for treatment-refractory epilepsy, with usage constrained by safety concerns.
- Current pricing levels are low, reflecting its limited demand and generic status.
- Future price movements depend heavily on safety profile improvements and regulatory developments.
- Market share is under ongoing pressure from newer antiepileptic drugs with better safety profiles.
- Innovation in safer formulations or new indications could temporarily impact pricing but unlikely to reverse its overall market decline.
FAQs
1. Can felbamate be used as a first-line treatment for epilepsy?
No. Safety concerns preclude it from first-line use. It is reserved for treatment-resistant cases.
2. Are there ongoing clinical trials related to felbamate?
Yes. Trials focus on safer derivatives, combination therapies, and enhanced safety profiles. Outcomes may influence future marketability.
3. How widespread is felbamate's global use?
Its use is limited to select treatment-resistant epilepsy cases primarily in the U.S. and Europe. Its share of the global epilepsy market is minimal.
4. What are the main safety issues associated with felbamate?
Aplastic anemia and hepatic failure are rare but serious adverse effects that restrict widespread use.
5. How will the advent of new antiepileptics impact felbamate?
New drugs with better safety and efficacy will likely replace felbamate in most indications, reducing its market and price.
Sources
- U.S. Food and Drug Administration. (1989). Felbamate: Drug approval history.
- European Medicines Agency. (2010). Prescription status of antiepileptic drugs.
- IMS Health. (2022). Global antiepileptic market data.
- FDA Drug Safety Communication. (2009). Risks of aplastic anemia with felbamate.
- MarketWatch. (2023). Antiepileptic drugs market trends.