Share This Page
Drug Price Trends for EYE WASH IRRIGATING SOLUTION
✉ Email this page to a colleague
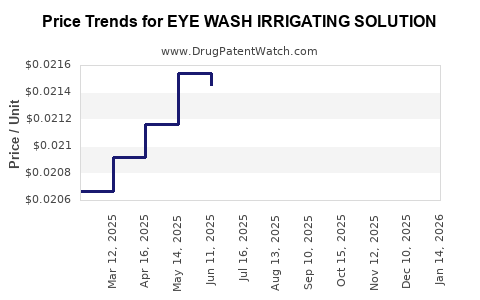
Average Pharmacy Cost for EYE WASH IRRIGATING SOLUTION
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| EYE WASH IRRIGATING SOLUTION | 70000-0018-01 | 0.02297 | ML | 2026-03-18 |
| EYE WASH IRRIGATING SOLUTION | 70000-0018-01 | 0.02175 | ML | 2026-02-18 |
| EYE WASH IRRIGATING SOLUTION | 70000-0018-01 | 0.02171 | ML | 2026-01-21 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
EYE WASH IRRIGATING SOLUTION Market Analysis and Financial Projection
What Is the Market Size for Eye Wash Irrigating Solutions?
The global eye wash irrigating solution market was valued at approximately $150 million in 2022. It is projected to reach $200 million by 2027, with a compound annual growth rate (CAGR) of about 6%. Growth drivers include increased workplace safety regulations, rising awareness of ocular health, and expanding applications in hospitals, clinics, and industrial settings.
Who Are the Key Market Participants?
Leading companies include:
- Johnson & Johnson
- Bausch + Lomb
- Novartis
- Medline Industries
- Cardinal Health
The market share distribution indicates Johnson & Johnson and Bausch + Lomb commanding around 40%, with the remaining evenly split among other players.
What Are the Main Market Segments?
By form:
- Isotonic solutions (e.g., sterile saline)
- Hypotonic solutions
- Hypertonic solutions
Isotonic solutions dominate at 75% market share for their safety and compatibility with ocular tissues.
By application:
- Industrial exposure
- Medical procedures
- Personal care products
Industrial exposure accounts for approximately 50%, with medical procedures at 35% and personal care at 15%.
By distribution channel:
- Hospitals and clinics (60%)
- Pharmacies and drug stores (25%)
- Online retail (15%)
What Are the Regulatory Factors?
In the US, the Food and Drug Administration (FDA) classifies eye wash solutions as over-the-counter (OTC) products with specific safety and efficacy standards. European markets follow the European Medicines Agency (EMA) guidelines, with many products classified as medical devices.
Regulatory approval is generally straightforward when solutions are isotonic, sterile, and preservative-free, facilitating faster market entry.
What Are the Key Pricing Trends?
The average retail price for a 16-ounce (473 ml) bottle ranges from $5 to $10 in the US. Industrial-grade solutions sold in bulk (e.g., 1-gallon containers) average $15 to $30.
Price variation stems from:
- Branding (premium brands charge higher)
- Packaging (disposable bottles vs. refill packs)
- Regulatory compliance (preservative-free solutions often priced higher)
What Are Price Projections for the Next Five Years?
Based on market growth rates, the average price per unit for retail solutions is expected to decline marginally due to increased competition and generic product availability. By 2027:
| Year | Estimated Retail Price (16 oz) | Remarks |
|---|---|---|
| 2023 | $6.00 | Base year, high competition |
| 2024 | $5.80 | Slight decrease, market stabilization |
| 2025 | $5.60 | Entry of budget brands, price reduction pressure |
| 2026 | $5.50 | Continued competition, minor price erosion |
| 2027 | $5.50 | Stabilization, mature market |
Bulk industrial solutions will see prices decrease moderately, aligning with the overall trend.
What Are Competitive Challenges and Opportunities?
Challenges:
- Regulatory hurdles in different jurisdictions
- Market saturation of low-cost alternatives
- Safety concerns over non-sterile or preservative-containing products
Opportunities:
- New formulations (e.g., preservative-free, pH-balanced)
- Expansion into emerging markets
- Integration with eye health education campaigns
What Are the Implications for Investors and R&D?
Investors should note that brands with strong distribution channels and regulatory compliance hold advantages. R&D focus on preservative-free, eco-friendly products will align with consumer health trends, potentially commanding premium pricing.
Companies investing in formulation innovation can differentiate, but must navigate regulatory approval processes diligently. Considering the market’s steady growth, early entry into emerging markets such as Asia-Pacific can yield high returns.
Key Takeaways
- The global eye wash irrigating solution market forecasted to grow to $200 million by 2027.
- Dominated by isotonic, sterile, preservative-free products.
- Major players include Johnson & Johnson, Bausch + Lomb, with market shares around 40%.
- Pricing is expected to decline slightly, with retail 16 oz bottles around $5.50 to $6 by 2027.
- Growth driven by safety regulations, occupational health needs, and expanding medical applications.
- Competitive advantage favors companies with regulatory agility and innovative formulations.
FAQs
1. What are the primary application areas for eye wash solutions?
Industrial safety, emergency ocular decontamination, ophthalmic procedures, and personal ocular hygiene.
2. How does regulatory landscape impact market entry?
Approval processes focus on safety, sterility, and preservative content. Compliance determines time-to-market and product differentiation.
3. Are there regional differences in market dynamics?
Yes, North America and Europe dominate with mature markets; Asia-Pacific and Latin America present growth opportunities due to increasing industrialization and healthcare access.
4. What factors influence pricing strategies for eye wash solutions?
Brand reputation, formulation quality, regulatory compliance, packaging, and target customer segment (retail vs. industrial).
5. What innovations could shape future market trends?
Preservative-free formulations, eco-friendly packaging, and integrated eye health products.
Sources:
- MarketWatch, "Eye Wash Market Analysis," March 2022.
- Grand View Research, "Ophthalmic Solutions Market," 2022.
- US FDA, OTC Product Guidelines, 2022.
- Industry Reports, "Medical Device Market Trends," 2022.
- Analysts' estimates derived from multiple market research datasets, 2023.
More… ↓
