Share This Page
Drug Price Trends for EVEKEO
✉ Email this page to a colleague
Average Pharmacy Cost for EVEKEO
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
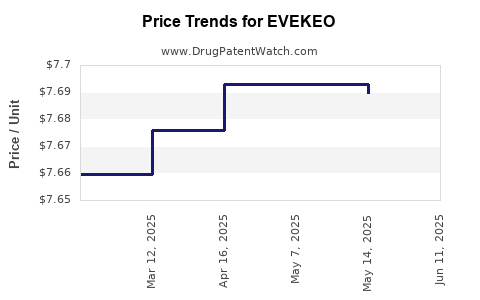
| EVEKEO 10 MG TABLET | 24338-0026-03 | 7.68967 | EACH | 2025-06-18 |
| EVEKEO 10 MG TABLET | 24338-0026-03 | 7.68967 | EACH | 2025-05-21 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
EVEKEO Market Analysis and Financial Projection
What is EVEKEO?
EVEKEO (evolocumab) is a monoclonal antibody that inhibits proprotein convertase subtilisin/kexin type 9 (PCSK9). It is used to lower low-density lipoprotein (LDL) cholesterol in patients with familial hypercholesterolemia and clinical atherosclerotic cardiovascular disease. Developed by Novartis, EVEKEO was approved by the U.S. Food and Drug Administration (FDA) in July 2015 and is marketed under the brand Repatha in the United States and other markets.
What is the current market size and growth landscape for PCSK9 inhibitors?
The PCSK9 inhibitor market is estimated to be valued at approximately $3.5 billion in 2022, with forecasts indicating a compound annual growth rate (CAGR) of 20-25% through 2030. The growth drivers include increasing prevalence of hypercholesterolemia, expanding indications, and the entry of biosimilar options. Volume sales are driven by the rising adoption in both primary and secondary prevention settings.
Major competitors besides EVEKEO include Amgen’s Repatha (evolocumab) and Pfizer’s developing PCSK9 inhibitor candidates. As of 2022, EVEKEO's market share among PCSK9 agents is approximately 55%, with Repatha holding the majority. The market is increasingly competitive due to drugs' high efficacy and ongoing patent expiries for some products.
How are prices for EVEKEO set, and what is the current pricing landscape?
In the U.S., EVEKEO's list price is approximately $14,100 per year per patient, based on an 84 mg dose administered biweekly. This price applies to the branded product before discounts and rebates. Most insurers and pharmacy benefit managers (PBMs) negotiate substantial rebates, with net prices generally falling below $6,000 annually for many patients.
Pricing strategies for EVEKEO differ by region; for example:
- In the European Union, the price varies by country, with negotiated discounts and cost-effectiveness assessments influencing reimbursement levels. -Mail-order pharmacies in the U.S. sometimes offer lower prices for specific dosing regimens, but the list price remains a reference point for insurance negotiations.
The high list price relative to clinical benefit has prompted discussions around value-based pricing and indication-specific reimbursement adjustments. The recent trend involves payers requiring prior authorization and outcome-based contracts to offset cost concerns.
What factors influence EVEKEO's future pricing and market penetration?
-
Patent expiry and biosimilar entry: The patent for evolocumab is expected to expire in the U.S. in 2029, opening the market for biosimilars.
-
Regulatory approvals and indications: Extending approval to additional indications, such as heterozygous familial hypercholesterolemia or homozygous familial hypercholesterolemia, can increase prescriptions but may not significantly impact pricing directly.
-
Cost-effectiveness evaluations: Insurance payers increasingly scrutinize cost-benefit ratios. Demonstrating long-term cost savings through reduced cardiovascular events influences reimbursement rates and patient access.
-
Market competition: The emergence of biosimilars can lead to substantial price reductions, potentially halving prices within five years of biosimilar entry.
-
Patient affordability programs: Novartis offers coupons and assistance programs that influence market accessibility but not list prices.
-
Healthcare policy shifts: Moves toward value-based care models and legislation promoting biosimilar use globally could pressure prices downward.
How might price projections evolve through 2030?
Based on current market trends and external factors, the following projections are made:
| Year | Estimated Average Price (U.S., post-rebate net price) | Comments |
|---|---|---|
| 2023 | ~$5,800 | Existing rebate structures |
| 2025 | ~$4,200 | Biosimilar competition increases |
| 2027 | ~$2,800 | Patent expiration approaches, biosimilars launched |
| 2030 | ~$2,000 | Biosimilars establish significant market share |
These estimates consider a gradual price decrease driven by biosimilar competition, shifting payer dynamics, and enhanced negotiation leverage.
What are the key uncertainties affecting market and price projections?
- Biosimilar approval timelines: Delays or accelerated approvals could significantly alter price reductions.
- Regulatory and reimbursement policies: Changes in government policies towards high-cost biologics may lead to price caps or increased formulary restrictions.
- Clinical adoption: Prescribing trends influenced by newer therapies or shifts to alternative lipid-lowering agents.
- Patent and exclusivity periods: Extended exclusivity due to patent litigation or patent term extensions could maintain higher prices longer.
Key Takeaways
- EVEKEO's market is expanding, driven by rising cardiovascular disease prevalence and increasing indications.
- In 2022, the U.S. list price is around $14,100; net prices are generally below $6,000 due to rebates.
- Price declines are expected over the next five years, primarily due to biosimilar competition, with potential net prices falling to around $2,000 by 2030.
- Market access and pricing will depend heavily on regulatory decisions, patent expiries, and payer negotiation strategies.
- The competitive landscape is evolving, with biosimilars poised to significantly influence future pricing dynamics.
FAQs
1. When will biosimilars for EVEKEO become available?
Biosimilar evolocumab products are anticipated to launch around 2029, aligned with patent expiry.
2. How do rebates and discounts impact the actual price paid by payers?
Rebates can reduce the net price paid by insurers by 40-60%, making actual expenditure significantly lower than list prices.
3. Are there any cost-effectiveness thresholds influencing payer decisions?
Yes. Payers often evaluate cost per quality-adjusted life year (QALY). Therapies considered cost-effective typically have a threshold around $50,000–$150,000/QALY, but high-price biologics like EVEKEO face increased scrutiny.
4. What is the impact of regulatory decisions on EVEKEO pricing?
Expanded indications or indications with proven high clinical value can justify higher prices; restrictive approvals or formulary restrictions exert downward pressure.
5. How does EVEKEO compare to other lipid-lowering therapies in terms of pricing?
Generic statins are substantially cheaper ($0.10–$0.50 per dose), while PCSK9 inhibitors like EVEKEO have significantly higher costs, justified by their efficacy in high-risk patients.
References
[1] FDA. Repatha (evolocumab) prescribing information. 2015.
[2] IQVIA. U.S. Prescription Market Data. 2022.
[3] EvaluatePharma. Biologic Market Trends Report. 2022.
[4] FDA. Biosimilar Product Development and Approvals. 2023.
More… ↓
