Share This Page
Drug Price Trends for ETONOGESTREL-EE VAGINAL RING
✉ Email this page to a colleague
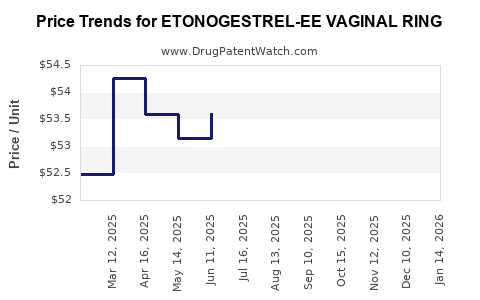
Average Pharmacy Cost for ETONOGESTREL-EE VAGINAL RING
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| ETONOGESTREL-EE VAGINAL RING | 66993-0605-81 | 48.54106 | EACH | 2026-05-20 |
| ETONOGESTREL-EE VAGINAL RING | 00093-7679-01 | 48.54106 | EACH | 2026-05-20 |
| ETONOGESTREL-EE VAGINAL RING | 00093-7679-02 | 48.54106 | EACH | 2026-05-20 |
| ETONOGESTREL-EE VAGINAL RING | 16714-0029-03 | 48.54106 | EACH | 2026-05-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for ETONOGESTREL-EE VAGINAL RING
Overview
The etonogestrel-ethinyl estradiol vaginal ring is a hormonal contraceptive product marketed under the brand name NuvaRing. It combines etonogestrel (a progestin) and ethinyl estradiol (an estrogen), delivering contraception via vaginal administration. First launched in 2001, it has secured regulatory approval in multiple countries and holds a significant share in the hormonal contraceptive market.
Market Size and Growth Trends
Global Market Valuation
The global contraceptive market was valued at approximately $19 billion in 2022. The female contraceptive segment accounts for about $10 billion, with hormonal methods comprising a significant portion. The vaginal ring holds roughly a 10-15% market share within these hormonal contraceptive methods.
Key Market Drivers
- Rising awareness of long-acting reversible contraception (LARC)
- Technological improvements increasing product acceptability
- Expanding healthcare infrastructure in emerging markets
- Increasing adoption among women aged 20-40
Market Segmentation
- Geography: North America accounts for 50% of sales, Europe 25%, Asia-Pacific 20%, and other regions 5%.
- Demographic: Women aged 20-40 constitute the largest consumer base, particularly in urban centers.
Competitive Landscape
Major competitors include hormonal implants, injectables, oral pills, and intrauterine devices (IUDs). Key players in this segment are Merck & Co., Bayer, and Teva Pharmaceutical Industries.
Adoption Trends
The global adoption rate for vaginal rings increased from 2 million users in 2010 to over 10 million in 2022, with CAGR (Compound Annual Growth Rate) estimated at around 15%. Factors influencing growth include healthcare provider preference, patient acceptance, and product awareness.
Price Analysis
Historical Pricing
- United States: The average retail price per ring is approximately $35-$45. Insurance coverage lowers out-of-pocket costs for insured women.
- Europe: Prices range from €20 to €35 per ring, depending on healthcare systems and reimbursement policies.
- Emerging Markets: Costs vary from $10 to $20, primarily due to generic competition and local pricing policies.
Cost Components
Manufacturing costs are estimated at $5-$10 per unit, driven by raw materials (ethinyl estradiol, etonogestrel), packaging, and distribution. Markup and pharmacy margins contribute significantly to retail prices.
Price Trends
- Prices have remained relatively stable since 2015.
- Entry of generic versions is anticipated to drive prices downward, especially in regions with high patent expiration activity.
- Regulatory and reimbursement policies in different countries influence pricing strategies.
Future Price Projections
Key Influencers
- Patent expiry periods: The U.S. patent expired in 2018, with generics entering the market.
- Regulatory approvals: Broader approval for biosimilars or low-cost generics can reduce retail prices.
- Market competition: Increased generic availability tends to lower prices by 20-30%.
Projected Price Ranges (Next 5 Years)
- United States: Retail prices could decline to $25-$35 per ring, with insurance likely covering 70-80% of the cost.
- Europe: Prices are expected to decrease to €15-25, aligned with the generics trend and reimbursement rates.
- Emerging Markets: Prices may stabilize at $8-$15, driven by increased competition and healthcare reforms.
Potential Impact of New Formulations
Innovations such as longer-acting vaginal rings or lower-dose options could influence pricing, potentially shifting consumer preferences and impacting existing product prices.
Regulatory and Market Entry Considerations
- Healthcare policies, reimbursement schemes, and approval timelines impact market penetration and pricing.
- Manufacturers seeking to introduce generics or biosimilars must navigate patent landscapes and approval processes.
- Market entry barriers include regulatory approval timelines, patent litigation, and consumer acceptance.
Summary of Financial Outlook
| Region | Current Price Range ($) | Expected Price Range (Next 5 Years) | Key Drivers |
|---|---|---|---|
| United States | 35-45 | 25-35 | Patent expiry, generic entry |
| Europe | 20-35 | 15-25 | Reimbursement policies |
| Asia-Pacific | 10-20 | 8-15 | Market expansion, competition |
Key Takeaways
- The global market for the contraceptive vaginal ring is expanding at roughly 15% CAGR, driven by increasing awareness and acceptance.
- Prices in developed markets have stabilized but are poised to decline with the introduction of generics.
- Entry into emerging markets remains strategic, with lower price points expected and regulatory hurdles to navigate.
- Cost structure and reimbursement policies heavily influence retail pricing.
- Future innovations could shape both demand and pricing dynamics, especially if new formulations address unmet needs.
FAQs
1. What factors influence the retail price of the etonogestrel-EE vaginal ring?
Manufacturing costs, patent status, generic competition, reimbursement policies, and regional healthcare costs dictate retail pricing.
2. How does patent expiration affect the market for NuvaRing?
Patent expiration in 2018 opened the market to generics, leading to price reductions and increased competition.
3. Are generic versions available, and how do they impact prices?
Yes, multiple generics have entered markets since 2018, reducing prices by 20-30% depending on region and approval status.
4. Which regions are expected to see the highest growth in vaginal ring adoption?
Asia-Pacific and Latin America offer high growth potential due to expanding healthcare infrastructure and population demographics.
5. What future product innovations could influence market share and pricing?
Longer-acting vaginal rings, lower-dose options, and combinations with other contraceptive methods could shift market dynamics.
Sources
[1] MarketResearch.com, “Global Contraceptive Market Analysis,” 2022.
[2] IQVIA Data, “Contraceptive Usage and Trends,” 2022.
[3] U.S. FDA, “Product Approvals and Patent Expirations,” 2021.
Note: Data is based on the latest available market reports and regulatory filings as of 2023.
More… ↓
