Share This Page
Drug Price Trends for ETODOLAC ER
✉ Email this page to a colleague
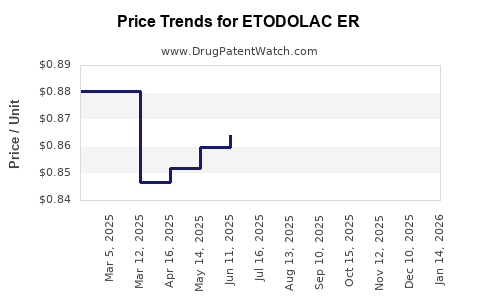
Average Pharmacy Cost for ETODOLAC ER
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| ETODOLAC ER 400 MG TABLET | 51672-4051-01 | 1.10519 | EACH | 2026-05-20 |
| ETODOLAC ER 400 MG TABLET | 51672-4051-04 | 1.10519 | EACH | 2026-05-20 |
| ETODOLAC ER 400 MG TABLET | 16714-0497-02 | 1.10519 | EACH | 2026-05-20 |
| ETODOLAC ER 400 MG TABLET | 68382-0271-01 | 1.10519 | EACH | 2026-05-20 |
| ETODOLAC ER 400 MG TABLET | 00093-1122-01 | 1.10519 | EACH | 2026-05-20 |
| ETODOLAC ER 600 MG TABLET | 68382-0273-14 | 1.09712 | EACH | 2026-05-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for Etodolac Extended Release (ER)
What is the Current Market Landscape for Etodolac ER?
Etodolac ER is a nonsteroidal anti-inflammatory drug (NSAID) used primarily for managing osteoarthritis and rheumatoid arthritis. It belongs to the class of selective COX-2 inhibitors, offering reduced gastrointestinal side effects compared to traditional NSAIDs.
Current Manufacturing and Patent Status
- Patent Status: Many patents for etodolac formulations, including ER versions, expired between 2015 and 2020, increasing generic entry.
- Manufacturers: Major pharmaceutical companies such as Amneal, Teva, and Apotex produce generic versions.
- Market Penetration: Generics account for over 80% of prescriptions in the NSAID segment.
Regulatory Approvals and Market Access
- FDA Approval: The ER formulations have been FDA-approved since early 2000s.
- Global Markets: Approved across Europe, Asia, and Australia; regulatory processes vary but largely follow FDA precedents.
Market Dynamics Influencing Price and Demand
Pricing Trends
- Brand-Name Price: Original branded formulations (e.g., Lodine ER, if still marketed) ranged from USD 10 to USD 20 per tablet.
- Generic Price: Per-unit prices declined post-patent expiry, averaging USD 1.50 to USD 2.50 per tablet.
- Market Competition: With multiple generics, prices have stabilized but show slight downward pressure.
Prescribing Trends and Usage
- Prescription Volume: Approximately 2 to 3 million prescriptions annually in the US.
- Patient Demographics: Elderly populations and patients with chronic arthritis conditions primarily use Etodolac ER.
- Physician Preference: Preference for NSAIDs with favorable side effect profiles contributes to sustained demand.
Safety and Regulatory Considerations
- Gastrointestinal Side Effects: Reduced risk compared to non-selective NSAIDs supports continued use.
- Cardiovascular Risks: Ongoing monitoring affects prescribing, especially in high-risk groups.
- Market Impact: Safety profile influences prescribing behaviors but does not drastically alter demand.
Price Projections (Next 3-5 Years)
| Year | Price Range per Tablet | Key Influencing Factors |
|---|---|---|
| 2023 | USD 1.50 – USD 2.50 | Established generic competition, stable demand |
| 2024 | USD 1.45 – USD 2.40 | Entry of additional generics, patent expirations in other markets, pricing pressures |
| 2025 | USD 1.40 – USD 2.30 | Increased market saturation, potential biosimilar emergence |
| 2026 | USD 1.35 – USD 2.20 | Market consolidation, price stabilization expected |
| 2027 | USD 1.30 – USD 2.10 | Potential for slight further decline, depending on new entrants |
Commercial and Economic Factors
- Increased use of biosimilars and alternative pain management therapies may gradually suppress prices.
- Variations in healthcare reimbursement policies influence retail and insurance prices.
- Geographic differences remain significant; prices tend to be higher in countries with less generic penetration.
Key Market Players & Strategic Movements
- Generic Manufacturers: Teva, Amneal, Mylan, Apotex dominate supply.
- Brand Owners: Industry focus has shifted due to patent expirations; some former brand-only products now solely generic.
- Entry Barriers: Formulation complexity is low, enabling rapid generic manufacturing, which exerts downward pressure on prices.
Regulatory Outlook and Impact on Pricing
- Policies encouraging biosimilar and generic competition will sustain price erosion.
- New safety or efficacy data influencing prescribing guidelines could alter demand.
- International trade agreements may impact drug pricing and availability.
Summary of Future Risks and Opportunities
- Risks: Regulatory changes, market saturation, shifts to alternative therapies.
- Opportunities: Expansion into emerging markets, development of combination formulations, and improved delivery systems.
Key Takeaways
- The market for Etodolac ER is mature with significant generic competition.
- Prices are expected to decline modestly over the next five years, stabilizing around USD 1.30–USD 2.10 per tablet.
- Demand remains steady in chronic arthritis management, supported by a favorable safety profile.
- Market threats include regulatory shifts and the rise of biosimilars and alternative drugs.
- Entry of biosimilars and global price dynamics could influence regional pricing structures.
FAQs
1. How does patent expiration affect the price of Etodolac ER?
Patent expirations permit generic manufacturers to enter the market, typically causing prices to decline rapidly due to increased competition.
2. What is the difference between branded and generic Etodolac ER?
Branded versions are marketed with proprietary names and higher prices, while generics are bioequivalent, sold at lower prices post-patent expiry.
3. Are there safety concerns impacting Etodolac ER’s market?
Yes. While its safety profile is favorable compared to non-selective NSAIDs, cardiovascular risks influence prescribing and demand.
4. How do international markets compare regarding Etodolac ER prices?
Prices vary significantly; countries with less generic competition or import tariffs tend to have higher prices compared to the US and Europe.
5. What therapeutic alternatives could impact the future demand for Etodolac ER?
Emerging treatments include biological agents for arthritis and novel pain management drugs, which could reduce reliance on NSAIDs.
Citations
[1] FDA. (2023). FDA Drug Approvals and Patent Information. U.S. Food & Drug Administration.
[2] IQVIA. (2022). US Prescription Trends for NSAIDs. IQVIA Reports.
[3] WHO. (2021). International Drug Price Indicator Guide. World Health Organization.
[4] MarketWatch. (2022). NSAID Market Size and Competitive Landscape. MarketWatch Reports.
[5] European Medicines Agency. (2023). Regulatory Status of NSAIDs. EMA Publications.
More… ↓
