Share This Page
Drug Price Trends for ESTRADIOL-NORETH
✉ Email this page to a colleague
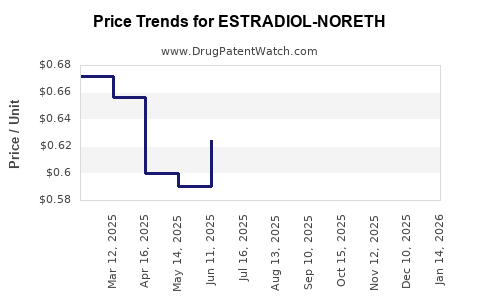
Average Pharmacy Cost for ESTRADIOL-NORETH
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| ESTRADIOL-NORETH 0.5-0.1 MG TB | 00378-7294-85 | 0.75878 | EACH | 2026-03-18 |
| ESTRADIOL-NORETH 0.5-0.1 MG TB | 50742-0658-84 | 0.75878 | EACH | 2026-03-18 |
| ESTRADIOL-NORETH 0.5-0.1 MG TB | 51991-0623-28 | 0.75878 | EACH | 2026-03-18 |
| ESTRADIOL-NORETH 0.5-0.1 MG TB | 50742-0658-28 | 0.75878 | EACH | 2026-03-18 |
| ESTRADIOL-NORETH 1-0.5 MG TAB | 51991-0474-28 | 0.65878 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
ESTRADIOL-NORETH Price Outlook and Market Dynamics
Estradiol-noreth, a combination hormone therapy, faces evolving market dynamics driven by patent expiries, generic competition, and shifts in therapeutic guidelines. This analysis forecasts price trajectories and examines key factors influencing market demand and supply.
What is Estradiol-Noreth and Its Current Market Position?
Estradiol-noreth is a fixed-dose combination of estradiol, a synthetic estrogen, and norethindrone acetate, a synthetic progestin. It is primarily used in hormone replacement therapy (HRT) for the management of menopausal symptoms. These symptoms include hot flashes, vaginal dryness, and the prevention of osteoporosis in postmenopausal women. The drug is administered orally.
The current market for estradiol-noreth is characterized by the presence of both branded and generic versions. The branded product, often marketed under specific trade names, typically commands a premium price. However, as patents expire, generic manufacturers enter the market, leading to significant price erosion. The therapeutic area of HRT has seen fluctuations in demand due to evolving safety concerns and updated clinical recommendations from regulatory bodies and medical associations. Despite some historical controversies surrounding HRT, current understanding and formulations have improved safety profiles, leading to a resurgence in its use for appropriately selected patient populations.
The global market for HRT products, including estradiol-noreth, is substantial. Market size is influenced by the prevalence of menopause, women's access to healthcare, and physician prescribing habits. Key geographical markets include North America, Europe, and parts of Asia. The competitive landscape involves several pharmaceutical companies, both large and small, producing and marketing various HRT formulations.
What is the Patent Status of Estradiol-Noreth?
The patent landscape for estradiol-noreth is critical to understanding future market dynamics and pricing. For many established estradiol-noreth formulations, the primary patents protecting the original composition of matter have long since expired. This allows for the introduction of generic alternatives.
- Original Composition of Matter Patents: These patents, which protected the initial discovery and formulation of estradiol-noreth, expired decades ago for many foundational products. For instance, patents related to early fixed-dose combinations of estradiol and norethindrone have been off-patent for many years.
- Formulation and Manufacturing Patents: While composition patents expire, pharmaceutical companies may hold patents on specific improved formulations, novel delivery systems, or advanced manufacturing processes. These patents can extend market exclusivity for specific product variations. For example, patents might cover extended-release formulations or specific polymorphic forms of the active pharmaceutical ingredients (APIs).
- Method of Use Patents: Patents may also exist for specific indications or methods of treating certain conditions using estradiol-noreth. However, the primary use in menopausal symptom management is well-established and generally not subject to broad, active method of use patents that would significantly restrict generic entry for this indication.
- Pediatric Exclusivity: In the U.S., for drugs approved for pediatric use, an additional six months of market exclusivity can be granted. However, estradiol-noreth is not typically indicated for pediatric use.
As of the most recent patent analyses, there are no broad, blocking patents that would prevent generic manufacturers from producing and marketing standard oral estradiol-noreth formulations for HRT in major markets like the United States and the European Union. The key period of patent exclusivity for most basic estradiol-noreth products has concluded. This allows for the continuous introduction of new generic competitors.
What are the Projected Price Trends for Estradiol-Noreth?
The projected price trends for estradiol-noreth are largely dictated by the forces of generic competition and supply-side economics.
- Current Generic Pricing: The price of generic estradiol-noreth has already seen significant declines from the peak prices of branded products. Prices for a typical monthly supply of generic estradiol-noreth tablets can range from $10 to $30 in the United States, depending on the dosage strength, manufacturer, and pharmacy. This is a substantial reduction compared to branded HRT products, which could historically cost upwards of $50 to $100 per month.
- Downward Price Pressure: The continued presence and potential entry of additional generic manufacturers will exert persistent downward pressure on prices. As more companies compete, price wars are likely to occur, especially for high-volume generic products. This trend is expected to continue for the foreseeable future.
- Price Stabilization: While prices will likely continue to decrease, the rate of decline may stabilize as the market reaches a point where manufacturers operate on thinner margins but can still achieve profitability due to volume. The lowest prices will likely be achieved by manufacturers with efficient production processes and established distribution networks.
- Potential for Price Increases (Niche/Specialty): In very specific niche formulations or if a novel delivery system for estradiol-noreth were to emerge with significant patent protection and demonstrable clinical advantage, prices for such specialized products could be higher. However, for the standard oral dosage forms, this is unlikely.
- Impact of Reimbursement Policies: Payer policies, including Medicare, Medicaid, and private insurance formularies, play a significant role. Increased formulary restrictions or higher co-pays for branded products can accelerate the shift to generics, further driving down the average selling price. Government-backed health programs often negotiate aggressively for lower generic prices.
- Forecasting Model: Based on current market data and typical generic drug lifecycle curves, prices for standard oral estradiol-noreth are projected to decrease by an additional 5-15% annually over the next three to five years, before potentially stabilizing. This assumes no major supply disruptions or significant changes in regulatory landscape that would favor branded products. By 2028, the average price for a month's supply of generic estradiol-noreth could fall to between $8 and $25.
Who are the Key Manufacturers and Competitors?
The competitive landscape for estradiol-noreth includes both originators of branded products and a multitude of generic manufacturers.
- Branded Products (Historical & Current): While specific trade names vary by region, historically, major pharmaceutical companies have marketed branded estradiol-noreth. Examples include products that were once dominant players. The market share for these branded products has diminished significantly due to generic erosion.
- Major Generic Manufacturers: The production of generic estradiol-noreth is dominated by companies with robust generic portfolios and large-scale manufacturing capabilities. Key players in the U.S. and European generic markets include:
- Teva Pharmaceuticals
- Viatris (formed from the merger of Mylan and Pfizer's Upjohn)
- Aurobindo Pharma
- Lupin
- Cipla
- Sun Pharmaceutical Industries
- Dr. Reddy's Laboratories
- Mayne Pharma
- Emerging Competitors: The relatively low barrier to entry for established generic drugs means that smaller or regional generic manufacturers can also enter the market, further intensifying competition.
- Competition Dynamics: Competition is primarily based on price, product availability, and supply chain reliability. Pharmaceutical wholesalers and pharmacy benefit managers (PBMs) exert considerable purchasing power, favoring manufacturers who can offer the lowest prices. Quality and compliance with regulatory standards (e.g., FDA, EMA) are prerequisites for market participation.
What Factors Influence Estradiol-Noreth Market Demand?
Demand for estradiol-noreth is influenced by a complex interplay of demographic, medical, and societal factors.
- Menopause Prevalence: The primary driver of demand is the menopausal population. As global populations age, the number of women experiencing menopause is increasing. This demographic trend provides a baseline demand for HRT.
- Therapeutic Guidelines and Clinical Recommendations: Changes in medical guidelines regarding HRT significantly impact demand.
- Past Concerns: Historical studies, such as the Women's Health Initiative (WHI) in the early 2000s, raised concerns about the risks associated with HRT (e.g., increased risk of breast cancer, stroke, and heart disease). This led to a sharp decline in HRT prescriptions.
- Current Understanding: Subsequent re-analysis of WHI data and newer research have refined the understanding of HRT risks and benefits, suggesting that HRT is safe and effective for many women when initiated at the time of menopause, with individualized risk assessments. Current guidelines from organizations like the North American Menopause Society (NAMS) and the Endocrine Society advocate for HRT as a first-line treatment for moderate to severe menopausal symptoms in healthy women. This has led to a cautious but growing resurgence in HRT use.
- Physician Prescribing Patterns: Physician comfort level and familiarity with current HRT guidelines directly influence prescribing habits. Doctors who are well-versed in updated evidence are more likely to prescribe HRT.
- Patient Awareness and Preferences: Patient education and awareness about the benefits and risks of HRT play a crucial role. Women seeking relief from bothersome menopausal symptoms may actively request HRT from their physicians.
- Availability of Alternatives: The market for menopausal symptom management includes various alternatives, such as non-hormonal prescription medications (e.g., SSRIs, SNRIs), herbal supplements, and lifestyle interventions. The efficacy and side effect profiles of these alternatives influence patient and physician choices.
- Healthcare Access and Insurance Coverage: Access to healthcare services and comprehensive insurance coverage for HRT are critical. In regions with limited healthcare infrastructure or restrictive insurance policies, demand can be suppressed.
- Emerging Research: Ongoing research into novel HRT formulations, personalized medicine approaches for HRT, and the long-term effects of HRT can shape future demand.
What are the Regulatory Considerations for Estradiol-Noreth?
Regulatory bodies worldwide play a pivotal role in the approval, marketing, and post-market surveillance of estradiol-noreth.
- Drug Approval Process: Estradiol-noreth products, both branded and generic, must undergo rigorous review by regulatory agencies before they can be marketed. In the United States, this is the Food and Drug Administration (FDA). In Europe, it is the European Medicines Agency (EMA).
- Generic Drug Approval (ANDA/ aMA): Generic versions of estradiol-noreth are approved through Abbreviated New Drug Applications (ANDAs) in the U.S. and Marketing Authorisation Applications (aMA) in Europe. To gain approval, generic manufacturers must demonstrate bioequivalence to the reference listed drug (RLD), meaning their product is absorbed into the bloodstream at the same rate and extent as the brand-name drug. They also must demonstrate that the product meets quality and manufacturing standards.
- Labeling and Prescribing Information: Regulatory agencies scrutinize product labeling and prescribing information. This includes details on indications, contraindications, warnings, precautions, adverse reactions, and drug interactions. Labeling must be consistent with current medical knowledge and regulatory requirements, particularly concerning the risks and benefits of HRT.
- Post-Market Surveillance (Pharmacovigilance): Manufacturers are required to monitor and report adverse events associated with their products. Regulatory bodies also conduct post-market surveillance to detect safety signals and ensure ongoing product safety.
- Good Manufacturing Practices (GMP): All manufacturing facilities producing estradiol-noreth must adhere to strict GMP regulations to ensure product quality, consistency, and safety. Regular inspections by regulatory authorities verify compliance.
- Marketing and Advertising Regulations: The marketing and advertising of prescription drugs are heavily regulated. Claims made in promotional materials must be accurate, balanced, and supported by scientific evidence. Off-label promotion (promoting a drug for an unapproved use) is prohibited.
- Therapeutic Equivalence and Interchangeability: Regulatory agencies assess therapeutic equivalence. In many jurisdictions, generic drugs deemed therapeutically equivalent can be substituted for their brand-name counterparts.
Key Takeaways
Estradiol-noreth is experiencing sustained price erosion due to the widespread expiry of foundational patents, enabling robust generic competition. Projected price declines of 5-15% annually are anticipated over the next three to five years. Demand is driven by an aging female population and a more favorable, risk-benefit-adjusted clinical consensus on hormone replacement therapy, counterbalancing historical safety concerns. Key generic manufacturers are focused on cost-efficient production and supply chain reliability to compete in this price-sensitive market. Regulatory oversight ensures bioequivalence and quality standards for generic approvals, with ongoing pharmacovigilance critical for post-market safety.
Frequently Asked Questions
-
Will branded estradiol-noreth products be discontinued? Branded estradiol-noreth products are unlikely to be fully discontinued in the short term but will continue to lose market share to generics. Manufacturers may maintain these products for specific patient preferences or legacy market segments, but their economic viability will be increasingly challenged.
-
Are there new formulations of estradiol-noreth in development that could impact pricing? While innovation in HRT continues, significant patent-protected novel oral formulations of estradiol-noreth that would drastically alter the generic market dynamics are not widely anticipated in the near future. The focus has shifted towards other delivery methods or combination therapies for specific indications, which may have different pricing structures and patent landscapes.
-
How do international pricing differences for estradiol-noreth compare? International pricing varies significantly due to differences in healthcare systems, national drug pricing regulations, and market competition. Generally, prices in countries with strong price negotiation powers or centralized healthcare systems (e.g., Canada, UK) tend to be lower than in less regulated markets like the U.S.
-
What is the impact of the Women's Health Initiative (WHI) study on current demand for estradiol-noreth? The WHI study significantly reduced demand for HRT, including estradiol-noreth, due to safety concerns. However, subsequent analyses and evolving clinical guidelines have re-evaluated the risks and benefits, leading to a more nuanced approach. Current demand reflects a balance between acknowledging risks and utilizing HRT for appropriate patients experiencing bothersome menopausal symptoms.
-
Can a generic estradiol-noreth product be substituted for any brand-name combination hormone therapy? Generic estradiol-noreth products are approved as substitutes for specific reference listed drugs (RLDs) with identical active ingredients, dosage forms, and strengths. Substitution for other combination hormone therapies with different progestins or dosages would not be permissible without a new prescription.
Citations
[1] North American Menopause Society (NAMS). (2022). The 2022 Hormone Therapy Position Statement of The North American Menopause Society. Menopause, 29(7), 767-794. [2] The Endocrine Society. (2017). Endocrine Society Clinical Practice Guideline: Treatment of Menopausal Symptoms. [3] U.S. Food and Drug Administration. (n.d.). Abbreviated New Drug Applications (ANDAs). Retrieved from [FDA website on ANDAs] [4] European Medicines Agency. (n.d.). Guidance on the content of Marketing Authorisation Applications. Retrieved from [EMA website on MAAs] [5] IMS Health Market Track data (Proprietary market data summaries accessible via subscription services). [6] Various generic drug pricing databases and pharmacy benefit manager (PBM) formularies.
More… ↓
