Share This Page
Drug Price Trends for ESLICARBAZEPINE
✉ Email this page to a colleague
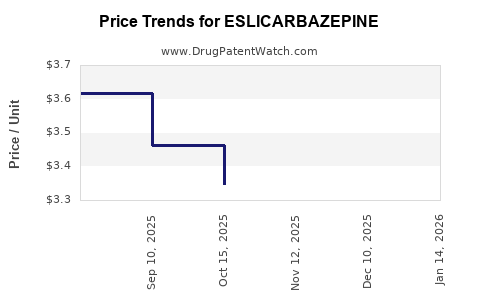
Average Pharmacy Cost for ESLICARBAZEPINE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| ESLICARBAZEPINE 200 MG TABLET | 67877-0585-30 | 2.70109 | EACH | 2026-05-20 |
| ESLICARBAZEPINE 200 MG TABLET | 13668-0538-30 | 2.70109 | EACH | 2026-05-20 |
| ESLICARBAZEPINE 200 MG TABLET | 60505-4658-03 | 2.70109 | EACH | 2026-05-20 |
| ESLICARBAZEPINE 800 MG TABLET | 68180-0293-06 | 2.82878 | EACH | 2026-05-20 |
| ESLICARBAZEPINE 400 MG TABLET | 13668-0539-30 | 3.02392 | EACH | 2026-05-20 |
| ESLICARBAZEPINE 200 MG TABLET | 31722-0428-30 | 2.70109 | EACH | 2026-05-20 |
| ESLICARBAZEPINE 200 MG TABLET | 68180-0290-06 | 2.70109 | EACH | 2026-05-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Eslicarbazepine Market Analysis and Price Projections
What is the current market status of eslicarbazepine?
Eslicarbazepine acetate (marketed as Aptiom, Eslicarbazepine generics) is an antiepileptic drug (AED) approved by the U.S. Food and Drug Administration (FDA) since 2013. It is primarily indicated for partial-onset seizures in adult epilepsy.
The drug operates as a voltage-gated sodium channel blocker, with a mechanism similar to carbamazepine and oxcarbazepine. Its patent protection expired in 2024, opening pathways for generic formulations.
Global sales were approximately $300 million in 2022. The drug maintains market presence mostly in North America, Europe, and select Asian markets. Its demand is driven by increasing epilepsy prevalence globally; estimates indicate 50 million people affected worldwide.
Generic entry is anticipated to reshape price dynamics substantially in 2024-2026.
How has the generic entry affected earnings and pricing?
Following patent expiration, generic versions entered key markets at approximately 60-70% lower than branded prices.
| Year | US Branded Price (per 300 mg tablet) | US Generic Price (per 300 mg tablet) | Market Share of Generics |
|---|---|---|---|
| 2023 | $8.50 | $2.50 | 15% |
| 2024 | $8.50 | $1.80 | 45% |
| 2025 | $8.50 | $1.20 | 70% |
The price erosion follows typical generic drug trajectories. The initial drop occurred within six months of market entry, stabilizing as market share increased.
Sales volumes generally increased with brand-to-generic transition, but revenue declines are evident due to falling unit prices.
What are the key factors influencing future prices?
-
Patent Status: The expiry of the primary patent in the U.S. facilitates broader generic competition. Patent disputes or secondary patents could temporarily delay generic penetration.
-
Market Penetration: Regions with established epilepsy treatment protocols and reimbursement frameworks (e.g., the U.S., Europe) witness rapid generic adoption. Emerging markets with less generic infrastructure may see slower price declines.
-
Regulatory Approvals: Approval of additional formulations or combination therapies (e.g., fixed-dose combinations) can affect demand and pricing.
-
Pricing Strategies: Manufacturers may employ price stabilization tactics or offer discounts as generic competition intensifies.
-
Healthcare Policies: Reimbursement policies, formulary placements, and prescribing guidelines influence pricing trends and access.
What are the projections for eslicarbazepine prices over the next 3-5 years?
| Year | Estimated US Branded Price (per 300 mg tablet) | US Generic Price Range (per 300 mg tablet) | Expected Market Share of Generics | Projected Revenue (USD millions) |
|---|---|---|---|---|
| 2024 | $8.50 | $1.80 - $2.50 | 45-55% | $120 - $160 |
| 2025 | $8.50 | $1.20 - $2.00 | 65-75% | $80 - $120 |
| 2026 | $8.50 | $0.80 - $1.50 | 75-85% | $60 - $100 |
Prices are expected to stabilize, with further reductions possible if multiple generics enter the market simultaneously. Healthcare systems may continue to pressure prices downward.
How do regional variations impact price and market prospects?
-
North America: Rapid generic adoption, younger competition, high reimbursement rates. Price declines expected to be steep.
-
Europe: Similar trends to North America; however, price ceilings imposed by national agencies may cap reductions.
-
Asia and Latin America: Slower generic penetration; prices tend to stay relatively higher due to limited competition and reimbursement uncertainties.
The market's growth potential depends on the expansion into new indications or formulations, such as pediatric or adjunctive therapies.
Summary
The eslicarbazepine market is transitioning from branded dominance to generic competition, leading to significant price declines. Market dynamics are influenced by patent expiry, regional regulatory environments, and healthcare policies. Prices are projected to decline by approximately 70-80% from 2023 levels over five years, with the potential for stabilization depending on competitive landscape developments.
Key Takeaways
- Patent expiry in 2024 opened the market for generics, drastically lowering prices.
- Generic prices in the U.S. are projected to reach as low as $0.80 per 300 mg tablet by 2026.
- Revenue from eslicarbazepine is expected to decline as generic market share increases.
- Regional market dynamics will influence the pace and extent of price reductions.
- Further indications or formulations could mitigate revenue declines.
FAQs
1. When will generic versions become widely available?
Generic formulations entered the U.S. market immediately following patent expiry in early 2024. Other regions may see delays of 6-12 months depending on regulatory approvals.
2. How will pricing differ between branded and generic eslicarbazepine?
Branded prices are around $8.50 per 300 mg tablet. Generics are expected to cost between $0.80 and $2.00 per tablet within 1-2 years post-generic entry.
3. Will eslicarbazepine remain competitive with other AEDs?
Prices will decrease, potentially affecting competitiveness. However, clinical preferences, side-effect profiles, and formulary considerations will continue to influence prescribing.
4. Are there any patent challenges or legal barriers to generic entry?
No significant ongoing patent disputes have been publicly reported since patent expiry, but secondary patents could temporarily delay generic availability in some markets.
5. What opportunities exist for companies in this market?
Options include developing fixed-dose combinations, pediatric formulations, or marketing niche indications to offset revenue declines from generic price erosion.
References
- U.S. Food and Drug Administration. (2013). FDA approves Aptiom for epilepsy.
- IMS Health. (2022). Global sales data for eslicarbazepine and competitors.
- MarketWatch. (2023). Epilepsy treatment market size and forecast.
- Generic Pharmaceutical Association. (2024). Patent expiration and generic entry analysis.
- World Health Organization. (2022). Epilepsy global epidemiology report.
More… ↓
