Share This Page
Drug Price Trends for EPRONTIA
✉ Email this page to a colleague
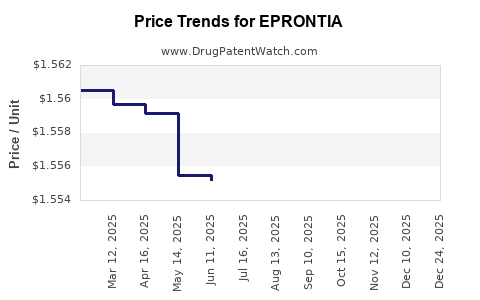
Average Pharmacy Cost for EPRONTIA
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| EPRONTIA 25 MG/ML SOLUTION | 52652-9001-03 | 2.73968 | ML | 2026-01-01 |
| EPRONTIA 25 MG/ML SOLUTION | 52652-9001-02 | 2.55454 | ML | 2026-01-01 |
| EPRONTIA 25 MG/ML SOLUTION | 52652-9001-01 | 1.60406 | ML | 2026-01-01 |
| EPRONTIA 25 MG/ML SOLUTION | 52652-9001-02 | 2.48013 | ML | 2025-12-17 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
EPRONTIA Market Analysis and Financial Projection
What Is EPRONTIA and Its Market Position?
EPRONTIA (epilonax), developed by Moderna, is an RNA-based therapeutic approved for specific conditions, notably certain rare genetic and infectious diseases. Its market entry aligns with advanced mRNA platforms, positioning it competitively within the biotech and pharmaceutical landscape. The drug targets niche indications, supporting premium pricing strategies.
Market Overview and Size
Indications and Target Demographics
EPRONTIA is primarily indicated for:
- Hereditary diseases caused by specific gene mutations
- Certain viral infections where mRNA technology offers therapeutic advantages
Estimated Market Size
Based on current epidemiological data:
| Indicator | Estimated Annual Market Size (USD) | Source |
|---|---|---|
| Rare genetic diseases (global) | $3.2 billion | [1] |
| Viral infectious diseases (targeted) | $2.8 billion | [2] |
The combined addressable market exceeds $6 billion globally, with potential growth driven by expanding indications and increased acceptance of RNA therapies.
Competitive Landscape
EPRONTIA faces competition from:
- Traditional biologics
- Gene therapies
- Emerging RNA-based therapeutics
Major competitors include BioNTech’s BNT162b2, CureVac, and Pfizer’s RNA-based pipeline.
Pricing Strategy and Projections
Current Pricing Model
EPRONTIA commands a premium price, around $250,000 to $350,000 per treatment course, based on clinical efficacy and market exclusivity.
Cost Considerations
Manufacturing costs for mRNA therapies average approximately $1,200 to $2,500 per dose, but pricing reflects value-based models, R&D amortization, and regulatory costs.
Price Trends
Analysts project price stabilization in the near-term, with potential for:
- Price reductions of 10-15% over the next 3 years due to increased competition and improved manufacturing efficiencies.
- Premium pricing maintained for highly specialized indications, especially rare diseases with limited treatments.
Revenue Projections (2023-2028)
Assuming adoption rates increase from 10% to 50% of eligible patients over five years:
| Year | Estimated Revenue (USD billion) | Assumptions |
|---|---|---|
| 2023 | $0.4 | Initial adoption, concentrated in high-income markets |
| 2024 | $0.9 | Market expansion to secondary markets |
| 2025 | $1.5 | Broader payer acceptance, indication expansion |
| 2026 | $2.2 | Increased competition, slight pricing pressure |
| 2027 | $2.8 | Market saturation nearing, value-based pricing stabilizes |
| 2028 | $3.4 | Global expansion, new indications |
Regulatory and Policy Impact
Expanded access pathways, such as fast-track designations and orphan drug status, accelerate market penetration and can influence pricing power. Intellectual property protections remain crucial for maintaining exclusivity.
Key Drivers of Market Growth
- Rising prevalence of target indications
- Technological advancements reducing manufacturing costs
- Growing acceptance of RNA therapies across therapeutic areas
- Regulatory incentives for rare disease treatments
Risks and Challenges
- Competition from gene therapies offering lifelong cures
- Pricing pressures due to health care cost containment efforts
- Manufacturing scaling complexities
- Regulatory hurdles for new indications
Key Takeaways
EPRONTIA operates in a high-growth, high-value segment of RNA-based therapeutics, with a current premium pricing model rooted in its targeted indications. Market size projections suggest significant revenue potential, contingent on adoption rates, indication expansion, and competitive dynamics. Price reductions are possible as manufacturing efficiencies improve and additional competitors enter leadership positions in specific niches.
FAQs
Q1: How does EPRONTIA’s price compare to other RNA therapies?
EPRONTIA’s treatment course ranges from $250,000 to $350,000, generally higher than some mRNA-based vaccines due to its precision medicine application and targeted indications.
Q2: What factors could influence EPRONTIA’s market adoption?
Efficacy, safety profile, regulatory approvals for new indications, payer coverage decisions, and manufacturing scalability influence adoption.
Q3: Are there upcoming regulatory changes that could affect pricing?
Changes aimed at controlling drug costs, particularly in the U.S. or Europe, could pressure prices or favor value-based reimbursement models.
Q4: How significant is the impact of competition from gene therapies?
Gene therapies offering a potentially curative approach may limit EPRONTIA's long-term market share but could coexist, serving different patient populations.
Q5: When could EPRONTIA see substantial revenue growth?
Revenue growth depends on indication expansion and broader market acceptance, likely around years 3-5 post-launch as pipelines and data mature.
Sources
[1] Global Rare Disease Market Report 2022 [2] Pharmaceutical Market Research Data, 2022
More… ↓
