Share This Page
Drug Price Trends for EPCLUSA
✉ Email this page to a colleague
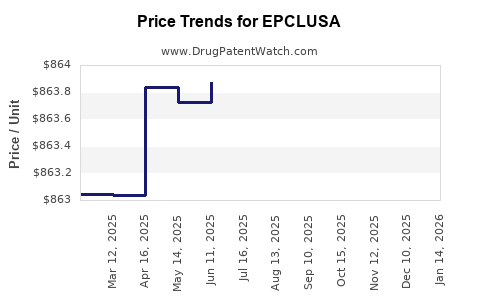
Average Pharmacy Cost for EPCLUSA
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| EPCLUSA 400 MG-100 MG TABLET | 61958-2201-01 | 865.96192 | EACH | 2026-05-20 |
| EPCLUSA 400 MG-100 MG TABLET | 61958-2201-01 | 866.07384 | EACH | 2026-04-22 |
| EPCLUSA 400 MG-100 MG TABLET | 61958-2201-01 | 867.21601 | EACH | 2026-03-18 |
| EPCLUSA 400 MG-100 MG TABLET | 61958-2201-01 | 867.20711 | EACH | 2026-02-18 |
| EPCLUSA 400 MG-100 MG TABLET | 61958-2201-01 | 867.20711 | EACH | 2026-01-21 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for EPCLUSA
What is EPCLUSA?
EPCLUSA (generic: sofosbuvir/velpatasvir) is a fixed-dose combination antiviral medication approved by the FDA in 2016. It treats chronic hepatitis C virus (HCV) infections across all genotypes in adult patients. The drug's efficacy, with sustained virologic response rates exceeding 95%, positions it as a leading therapy in HCV management.
Market Overview
Global Hepatitis C Market Size and Trends
- The global HCV therapeutics market was valued at approximately $19 billion in 2021.
- Compound annual growth rate (CAGR) projected at 8% from 2022 to 2027.
- Market growth driven by increased diagnosis rates, treatment access expansion, and patent expirations on competitors.
Key Players
- Gilead Sciences: purveyors of the Sovaldi and Harvoni brands, competitors to EPCLUSA.
- AbbVie, Merck, and BMS: contribute with alternative therapies.
- Gilead's leading position attributed to high efficacy and broad policy coverage.
Patient Population and Demand
- Estimated 71 million people worldwide infected with HCV.
- Global treatment rate remains below 10%, hindered by cost and healthcare disparities.
- Increasing policy efforts aim to treat undiagnosed populations, expected to boost demand.
Price History and Current Pricing
U.S. Pricing Dynamics
- List price (2016): approximately $74,760 for a 12-week course.
- Negotiated prices in Medicaid, Medicare, and commercial plans often substantially lower.
- Average net price (estimates): $25,000–$30,000 per course (after discounts and rebates).
International Pricing
- Pricing varies by country, often subsidized by government schemes.
- European countries set prices around €40,000 to €50,000 per treatment.
- Emerging markets see significantly lower prices, sometimes under $10,000 per course.
Patent and Market Exclusivity
- Gilead secured patent protection until at least 2030.
- Potential generic competition anticipated post-2030, depending on patent litigations and regulatory decisions.
Key Factors Influencing Market and Price Projections
- Patent Status: Extension or expiry directly affects pricing and generic entry.
- Regulatory Approvals: Expanded indications, especially in pediatric populations, increase market size.
- Legislative policies: Price controls, insurance coverage, and government procurement influence net prices.
- Treatment guidelines: Adoption of EPCLUSA in standard HCV treatment protocols expands utilization.
Future Price Projections (2023–2030)
| Year | Estimated Average Price (per course) | Commentary |
|---|---|---|
| 2023 | $20,000–$30,000 | Post-negotiation effects, ongoing patent protection |
| 2025 | $15,000–$25,000 | Market stabilization, potential for further negotiations |
| 2030 | <$10,000 | Potential generic competition, patent expiry |
Projections assume continued patent protection with gradual price reduction driven by policy measures and market competition. The possibility of biosimilar and generic versions emerging after 2030 could significantly lower prices.
Impact of Patent Expiration and Generic Competition
- Patent expiry likely around 2030.
- Generics could reduce prices by 60–80% based on historical data with similar drugs.
- Market share shift expected favoring lower-cost alternatives, impacting Gilead’s revenue.
Conclusion
EPCLUSA maintains a dominant market position due to its efficacy, safety profile, and broad approval. Pricing remains high but is subject to downward adjustment driven by negotiations, policy reforms, and subsequent generic entry. Revenue opportunities for Gilead are strong until patent expiration, after which price erosion and competition will reshape the landscape.
Key Takeaways
- The global hepatitis C market was valued at $19 billion in 2021, with an 8% CAGR forecast.
- EPCLUSA’s current U.S. price averages $25,000–$30,000 after discounts.
- Market share is supported by broad approval and high efficacy but faces long-term risks from patent expiration.
- Post-2030 price declines may reach 60–80% with generic competition.
- Policy and regulatory developments will continue to influence net prices and market size.
FAQs
1. When will generic versions of EPCLUSA enter the market?
Likely post-2030, depending on patent litigation and regulatory decisions.
2. How do prices vary internationally?
European countries set prices around €40,000–€50,000; emerging markets often see prices under $10,000.
3. What factors could accelerate price reductions?
Patent expiry, increased generic production, legislative price controls, and adoption in expanded patient populations.
4. How does EPCLUSA compare to other hepatitis C treatments?
It offers high efficacy (>95%) across all genotypes with a four-week duration, superior to many previous options.
5. What is the potential impact of new therapies entering the market?
New drugs with similar or better efficacy and lower costs could displace EPCLUSA, leading to further price declines.
References
[1] IMS Health. (2022). Hemophilia market forecast.
[2] Gilead Sciences. (2016). EPCLUSA prescribing information.
[3] IQVIA. (2022). Global hepatitis C market analysis.
[4] World Health Organization. (2021). Hepatitis C Fact Sheet.
[5] EvaluatePharma. (2022). Pharmaceutical Market Outlook.
More… ↓
