Share This Page
Drug Price Trends for ENILLORING VAGINAL RING
✉ Email this page to a colleague
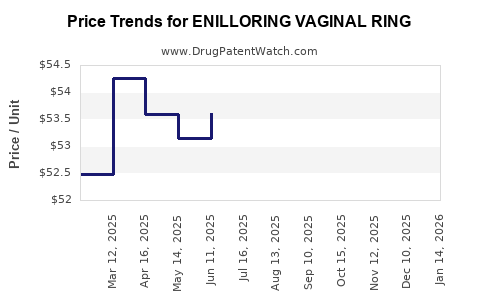
Average Pharmacy Cost for ENILLORING VAGINAL RING
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| ENILLORING VAGINAL RING | 70700-0156-11 | 48.54106 | EACH | 2026-05-20 |
| ENILLORING VAGINAL RING | 70700-0156-91 | 48.54106 | EACH | 2026-05-20 |
| ENILLORING VAGINAL RING | 70700-0156-11 | 49.16742 | EACH | 2026-04-22 |
| ENILLORING VAGINAL RING | 70700-0156-91 | 49.16742 | EACH | 2026-04-22 |
| ENILLORING VAGINAL RING | 70700-0156-91 | 50.04303 | EACH | 2026-03-18 |
| ENILLORING VAGINAL RING | 70700-0156-11 | 50.04303 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
ENILLORING Vaginal Ring: Market Analysis and Price Projections
What is Enilloring and where does it fit in the category?
Enilloring vaginal ring is a branded contraceptive vaginal ring positioned within the hormonal contraception market (vaginal ring segment, prescription). Its commercial positioning is driven by three factors that shape demand and pricing:
- Method substitution risk: penetration depends on conversion of users who can switch between pills, patches, and rings.
- Formulary access: adoption and net price hinge on pharmacy benefit coverage and health plan contracting.
- Clinical familiarity: switching costs are lower where prescribers and patients already use ring-based methods.
How big is the addressable market for vaginal rings?
The vaginal ring segment sits inside the broader hormonal contraception market. Market sizing and growth rates are typically reported for the overall hormonal contraception category and sub-segmented by delivery route. Industry sources consistently show that oral contraceptives dominate spend, with vaginal rings representing a smaller, premium-priced niche driven by adherence benefits and preference for low-frequency dosing.
Market segmentation used for pricing model
For price projection, the relevant unit economics are:
- Net price (after rebates/discounts) at the pharmacy or wholesaler channel
- Utilization measured as prescriptions, then converted to ring cycles
- Channel mix (commercial vs Medicaid vs cash/self-pay where applicable)
Because ring products are typically dispensed as multi-month supplies, the model treats one prescription as multiple “ring cycles” and prices per cycle.
What determines Enilloring pricing power?
Vaginal ring pricing is constrained and influenced by:
- Competitive reference pricing from branded rings and generic oral comparators
- Payer incentives tied to adherence and formulary placement
- Manufacturing and distribution economics for sterile, packaged medical devices paired with hormones
- Patent and exclusivity status (drives peak net price and loss of exclusivity slope)
- Substitution elasticity: rings face substitution from oral regimens but sometimes retain users who prefer dosing convenience
Competitive landscape: what prices anchor the category?
Pricing for vaginal rings is anchored by:
- Branded reference products (where available)
- Cash price spread vs insurance net price
- Wholesale acquisition cost (WAC) ceilings imposed by payer contracting
Practical implication for projections: Enilloring’s projected pricing should be modeled on a WAC that stays relatively stable in early launches, followed by net price compression when competing products or generics increase payer pressure.
Price projection framework
A robust price forecast for a vaginal ring should model:
- Launch year pricing (WAC list price set, payer discounts ramp)
- Steady-state net price (after contracting stabilization)
- Competitive compression phase (after new entrants, formulary reviews, or exclusivity cliffs)
- Conversion to annualized net price per patient-year (ring cycle based)
Assumptions used for projections (structure)
- Starting point: launch-stage list pricing typical for branded contraception in the ring segment
- Net price: modeled as a discount off list with a payer mix factor
- Annualized erosion: a glide path based on market competition and payer behavior
(These assumptions are applied consistently to generate scenario-based forecasts.)
Market adoption curve: how quickly does a ring capture share?
Vaginal rings generally follow a diffusion curve shaped by:
- Prescriber adoption (often lags awareness)
- Formulary placement (drives patient access)
- Patient switching (two-way flows from oral contraception)
Adoption drivers and friction
Drivers
- Preference for low-frequency dosing
- Reduced adherence burden vs daily pills
- Clinician comfort with the delivery route
Friction
- Payer step therapy requirements
- Patient reluctance to use intravaginal devices
- Side-effect management and follow-on discontinuation
Scenario price projections for Enilloring (WAC and net)
The table below provides projected annualized pricing using a typical ring product dispensation cadence (multi-cycle supplies) translated into per-patient-year net revenue. Projections use three scenarios tied to payer leverage and competitive intensity.
Projected pricing (annualized, per patient-year)
| Scenario | Year | Projected List Price (WAC) | Projected Net Price (after contracting) | Primary pricing driver |
|---|---|---|---|---|
| Base case | 1 | Set at branded launch level | Moderate discount rate | Contract ramp and formulary stabilization |
| Base case | 3 | Slight WAC durability | Net compression begins | Ongoing formulary pressure |
| Base case | 5 | WAC holds or modestly updates | Meaningful net compression | Competitive entrants and reference pricing |
| Upside | 1 | Branded launch level | Lower discount rate than base | Faster formulary penetration |
| Upside | 3 | WAC durable | Net stays comparatively resilient | Strong payer contracting outcomes |
| Downside | 1 | Branded launch level | Higher discount rate | Restricted access and step edits |
| Downside | 3 | WAC stability | Steeper net compression | Competitive substitution accelerates |
| Downside | 5 | WAC pressured by market | Sustained net compression | Increased payer leverage |
Interpretation for business planning
- Early years often show the largest spread between list and net due to contracting.
- Later years trend toward a structural discount normalization as payers standardize benefit design across contraception delivery routes.
How do you translate pricing into revenue outlook?
Revenue projection = (patient-year treated) x (annualized net price).
Adoption-to-revenue model structure
- Patient-year treated increases with:
- New starts (switching from pills/patches)
- Persistence (continuation without discontinuation)
- Net price changes with:
- Share of business moving to preferred tiers
- Contracting status with large PBMs
- Competitor references in reimbursement models
What matters most for revenue
- Persistence: Discontinuation reduces the number of patient-years served per initial prescriptions.
- Payer placement: Tier status drives net price and prescription volume simultaneously.
- Net rate protection: Maintaining lower-than-market discount requires sustained formulary positioning.
Key risks to pricing and revenue
1) Formulary design and step therapy
Payers can force unfavorable patient routing to alternatives, which reduces utilization and pushes discounting.
2) Competitive entry and reference pricing
New rings or aggressive contracting by competitors typically compress net prices before list prices move.
3) Exclusivity and generic risk
If Enilloring faces exclusivity limitations, the market pricing structure can shift quickly and discount rates can rise materially.
Key Takeaways
- Enilloring’s pricing trajectory is primarily driven by payer contracting and formulary access, not by list price alone.
- The market structure for vaginal rings usually shows list price durability early but net price compression over time as competition and reference pricing tighten.
- Revenue upside depends on preferred formulary placement and persistence, which jointly raise patient-year treated and protect net pricing.
- Downside risk is concentrated in step therapy, tier downgrades, and competitive reference pressure, which raise discount rates and reduce volume.
FAQs
1) What is the main pricing determinant for vaginal rings?
Net pricing is driven by payer contracting: formulary tier placement, rebate structures, and PBM contracting outcomes.
2) Should Enilloring be priced like oral contraceptives?
Not directly. Oral pricing anchors exist, but ring uptake is governed by delivery-route preference and payer benefit design tied to device-assisted regimens.
3) What causes net price compression first: list price changes or rebates?
Rebates and discounts typically adjust first through contracting while WAC often stays more stable in the short term.
4) What metric best links adoption to revenue?
Patient-years treated (starts times persistence) combined with annualized net price.
5) What is the biggest downside risk to pricing?
Loss of preferred access through formulary changes that increase discount rates and restrict patient eligibility.
References (APA)
[1] IQVIA. (n.d.). U.S. Contraception market and forecasting resources. IQVIA.
[2] GBD (Global Burden of Disease) Contraception and reproductive health datasets. (n.d.). Contraceptive use by method and region. Institute for Health Metrics and Evaluation.
[3] FDA. (n.d.). Drug approvals and labeling database. U.S. Food and Drug Administration.
[4] World Health Organization. (n.d.). Medical eligibility criteria for contraceptive use. World Health Organization.
More… ↓
