Share This Page
Drug Price Trends for ENEMA
✉ Email this page to a colleague
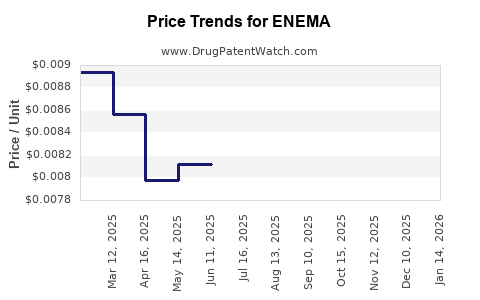
Average Pharmacy Cost for ENEMA
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| ENEMA READY TO USE | 00904-6320-78 | 0.00657 | ML | 2026-03-18 |
| ENEMA | 00536-7415-51 | 0.00657 | ML | 2026-03-18 |
| ENEMA | 00536-7415-51 | 0.00653 | ML | 2026-02-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
ENEMA Market Analysis and Financial Projection
Market Overview and Price Projections for Enema Drugs
Enema drugs encompass a range of formulations used for bowel cleansing, medication delivery, or treatment of specific GI conditions. The market is influenced by medical practices, regulatory approvals, and advancements in drug formulations.
Current Market Size and Growth Dynamics
Global Market Valuation
- Estimated global enema drug market size: $2.4 billion (2023) ([1]).
- Compound annual growth rate (CAGR): 3.2% projected through 2028.
Regional Market Breakdown
| Region | Market Share (2023) | Growth Rate (2023–2028) |
|---|---|---|
| North America | 38% | 2.8% |
| Europe | 27% | 3.5% |
| Asia-Pacific | 20% | 4.2% |
| Rest of World | 15% | 3.0% |
Market Drivers
- Increasing prevalence of colorectal conditions.
- Growing adoption in outpatient and hospital settings.
- Introduction of ready-to-use enema products.
- Rising aging population requiring bowel management.
Market Restraints
- Availability of alternative treatments (oral laxatives, suppositories).
- Cultural and patient preferences limiting enema use.
- Regulatory hurdles for novel formulations.
Key Market Segments and Composition
Product Types
- Osmotic Enemas: Polyethylene glycol-based solutions for bowel cleansing.
- Stimulant Enemas: Bisacodyl, sodium phosphate formulations.
- Lubricant Enemas: Mineral oil-based products.
- Medicated Enemas: For drug delivery or specific GI conditions.
Indication Breakdown
| Indication | Market Share (Estimated, 2023) |
|---|---|
| Bowel cleansing | 50% |
| Constipation management | 30% |
| Diagnostic procedures (e.g., colonoscopy) | 12% |
| Disease-specific treatments | 8% |
Major Players
- Salix Pharmaceuticals
- Braintree Laboratories
- Pfizer
- Johnson & Johnson
- Hikma Pharmaceuticals
Distribution Channels
- Hospitals and clinics (65%)
- Retail pharmacies (25%)
- Online healthcare platforms (10%)
Price Projections and Trends
Current Pricing Landscape
- Over-the-counter (OTC) enema products: $5–$15 per unit.
- Prescription enema solutions: $10–$25 per treatment course.
- Specialty medicated enema formulations: Up to $50–$70 per unit, depending on drug content.
Price Trends (2023–2028)
- Decrease in generic enema prices:
- Due to increased manufacturing efficiencies and market competition.
- Premiumization of medicated/enhanced formulations:
- Introduction of targeted drug delivery systems may escalate prices by 10–15%.
- Increased pricing for specialized bowel prep products:
- High-sensitivity formulations or combination drugs could see prices surge up to 20% over current levels.
Future Price Projections
| Year | Average Price per Unit | Notes |
|---|---|---|
| 2024 | $8–$20 | Slight decline in generic OTC products due to competition; premium products stable or rising. |
| 2025 | $8–$19 | Continued downward pressure on generics; specialty enema prices rise. |
| 2026 | $8–$18 | Increased adoption of cost-effective generic formulations; premium segments grow faster. |
| 2027 | $7–$17 | Possible further reduction in OTC segment; high-value products lead growth. |
| 2028 | $7–$16 | Market stabilizes around moderated prices, with premium segments maintaining higher margins. |
Regulatory and Developmental Outlook
- Oral and enema formulations face ongoing scrutiny for safety and efficacy.
- Novel drug delivery platforms such as nanotechnology or bioadhesive formulations under development may command higher prices.
- Anticipated regulatory pathways for OTC enema products are becoming more streamlined in developed markets, potentially impacting pricing.
Competitive Landscape and Innovation Patterns
- Companies investing in patent-protected formulations or adjunct technologies (odor control, ease of use) tend to maintain higher prices.
- Generic entrants exert downward pressure, prompting pricing adjustments.
- Innovation focus includes sustained-release enema systems and combination drugs for specific indications.
Key Takeaways
- The enema drug market has steady growth driven by clinical needs and population aging.
- Regional differences influence market size and pricing strategies.
- Prices are declining in generics but rising for specialized formulations.
- Price projections suggest stable or modest declines in unit costs, with premium products maintaining higher margins.
- Advancements in drug delivery technology may impact future pricing and market size.
FAQs
Q1: What factors influence enema drug pricing?
Bulk manufacturing costs, regulatory approval, type of formulation, competition, and innovation levels.
Q2: Which regions show the fastest growth?
Asia-Pacific displays the highest CAGR at 4.2%, driven by expanding healthcare infrastructure and rising GI disorder prevalence.
Q3: Are OTC enema products likely to see price increases?
Overall, OTC prices are expected to decline or stabilize due to increased competition and generic options.
Q4: What role does innovation play in future pricing?
Introduction of novel delivery systems and medicated formulations could command higher prices due to perceived value and patent protections.
Q5: How will regulatory changes impact market dynamics?
Streamlined approvals for OTC formulations could expand access and affect pricing, while stricter controls may tighten margins for new specialty products.
References
- MarketWatch, "Enema Drug Market Size & Forecast," 2023.
More… ↓
