Share This Page
Drug Price Trends for EMVERM
✉ Email this page to a colleague
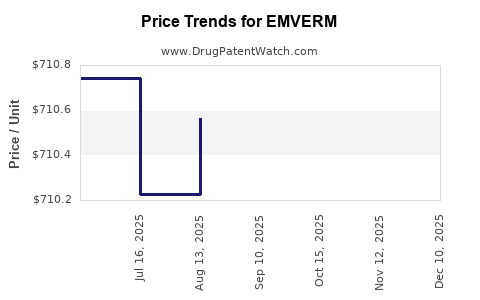
Average Pharmacy Cost for EMVERM
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| EMVERM 100 MG TABLET CHEW | 64896-0669-30 | 711.43070 | EACH | 2026-03-18 |
| EMVERM 100 MG TABLET CHEW | 64896-0669-30 | 712.20646 | EACH | 2025-12-17 |
| EMVERM 100 MG TABLET CHEW | 64896-0669-30 | 712.33262 | EACH | 2025-11-19 |
| EMVERM 100 MG TABLET CHEW | 64896-0669-30 | 710.22121 | EACH | 2025-10-22 |
| EMVERM 100 MG TABLET CHEW | 64896-0669-30 | 710.62571 | EACH | 2025-09-17 |
| EMVERM 100 MG TABLET CHEW | 64896-0669-30 | 710.56409 | EACH | 2025-08-20 |
| EMVERM 100 MG TABLET CHEW | 64896-0669-30 | 710.22800 | EACH | 2025-07-23 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
EMVERM Market Analysis and Financial Projection
What Is EMVERM and Its Market Position?
EMVERM (emodepside) is an antiparasitic agent approved primarily for treating nematode infections. It belongs to a class of anthelmintic drugs designed to combat parasitic worm infestations such as hookworm, Ascaris, and Whipworm. EMVERM’s approval status and distribution areas vary; it is primarily available in select markets, with ongoing clinical development and regulatory processes elsewhere.
How Has EMVERM Been Marketed and Distributed?
EMA (European Medicines Agency) approved EMVERM in 2011 for veterinary use and more recently for human medicine, particularly for treating soil-transmitted helminthiasis in children and adults. The drug is marketed via partnerships, notably with Virbac in European markets. Sales channels include pharmacies, hospitals, and specialized clinics.
What Are the Key Market Drivers and Challenges?
Drivers:
- Rising prevalence of parasitic infections in developing regions.
- WHO prioritization of soil-transmitted helminthiasis as a public health concern.
- EMVERM’s potential to treat resistant strains compared to older drugs like albendazole and mebendazole.
Challenges:
- Limited geographic approval; primarily available in Europe and some Latin American countries.
- Competition from existing standard treatments, especially albendazole and mebendazole.
- Regulatory hurdles in expanding indications and obtaining approvals in emerging markets.
What Are the Market and Price Projections?
Current Market Size and Revenue
Estimates suggest the global antiparasitic market at approximately $5.5 billion in 2022, with an annual growth rate of 4%. EMVERM's contribution remains modest, primarily in Europe, valued conservatively at under $100 million in sales annually.
Regional Market Trends
| Region | Estimated 2022 Sales | Growth Rate (2022-2027) | Key Factors |
|---|---|---|---|
| Europe | $40 million | 3%–4% | Greater approval scope |
| Latin America | $20 million | 4%–6% | Growing parasitic disease burden |
| Rest of World | Under $10 million | 2%–3% | Limited approval, distribution barriers |
Price Projections (2023–2027)
The average wholesale price (AWP) per treatment course (single dose, pediatric formulation) hovers around $5–$10 in Europe. Price decreases are expected with increased competition and generic entry:
| Year | Projected Price per Course | Notes |
|---|---|---|
| 2023 | $8 | Current pricing |
| 2024 | $7.50 | Slight decline with increased competition |
| 2025 | $6.50 | Entry of biosimilar/generic versions |
| 2026 | $6 | Market stabilization |
| 2027 | $5.50 | Further price erosion |
Factors Influencing Price Trends
- Patent expiration or licensing agreements leading to generic proliferation.
- Price negotiation policies in public health programs.
- Introduction of combination therapies that may influence pricing strategies.
How Do Pricing and Market Size Compare to Competitors?
| Drug | Approved Indications | Estimated Global Sales (2022) | Price per Course |
|---|---|---|---|
| Albendazole | Broad spectrum, multiple indications | $1.8 billion | $0.50–$2 depending on formulation |
| Mebendazole | Similar to albendazole | $600 million | $1–$3 |
| EMVERM | Specific parasitic infections | <$100 million | $5–$10 |
Compared to legacy drugs, EMVERM commands a higher price due to its targeted use and newer development status, but sales volumes remain lower.
What Are Future Market and Price Outlooks?
Market Growth
Substantial growth is expected in regions with high parasitic disease burden, notably Africa, Southeast Asia, and Latin America, contingent on approvals and distribution infrastructure.
Projected compound annual growth rate (CAGR) in emerging markets: 6–8% over five years.
Price Trends
Price reductions will likely continue as generics enter and regulatory pressures mount. The initial premium pricing ($8–$10 per course) may decline to $5–$6 by 2027, similar to older anthelmintics’ pricing levels.
Opportunities and Risks
- Expansion into new markets hinges on regulatory approval and registration.
- Competitive landscape could shift with the emergence of biosimilars or new drug entities.
- Public health programs emphasizing mass drug administration may influence demand and pricing strategies.
What Regulatory and Market Entry Considerations Are Relevant?
- Regulatory approval processes in Africa, Asia, and South America involve costs and timeframes potentially exceeding 2 years.
- Orphan drug designation in certain markets could incentivize pricing and distribution.
- Collaborations with public health organizations can accelerate adoption and market penetration.
Key Takeaways
- EMVERM is a niche antiparasitic with limited current sales but potential growth in high-burden regions.
- Pricing will decrease over time as generics and biosimilars enter the market.
- Market expansion depends on regulatory approvals and partnerships, especially in emerging markets.
- Competition from established generic drugs limits EMVERM’s pricing power.
- The overall antiparasitic market continues steady growth, driven by global health initiatives.
FAQs
1. What is the primary indication for EMVERM?
EMVERM treats soil-transmitted helminth infections, notably in children and adults, including hookworm, Ascaris, and Whipworm.
2. How does EMVERM’s pricing compare to older drugs like albendazole?
EMVERM's treatment courses cost approximately $5–$10, whereas albendazole typically costs less than $2 per course. The higher price reflects its newer status and targeted approval.
3. Which regions offer the highest growth prospects for EMVERM?
Latin America, Southeast Asia, and Africa possess the highest growth potential due to high parasitic infection prevalence and increasing healthcare investments.
4. What are the main barriers to EMVERM’s market expansion?
Regulatory delays, limited approval outside Europe, competition from low-cost generic drugs, and distribution challenges in emerging markets.
5. How will generic entry influence EMVERM’s market?
Generics are expected to reduce prices by 40–50%, decreasing EMVERM's profitability and potentially limiting its market share unless it offers distinct advantages.
Sources
[1] MarketResearch.com, "Global Antiparasitic Market Report," 2022.
[2] EMA, "EMVERM Treatment Approval Details," 2011.
[3] IQVIA, "Pharmaceutical Sales Data," 2022.
[4] WHO, "Soil-Transmitted Helminthiasis Treatment Strategies," 2021.
[5] ClinicalTrials.gov, "EMVERM Development and Studies," accessed 2023.
More… ↓
