Share This Page
Drug Price Trends for EMEND
✉ Email this page to a colleague
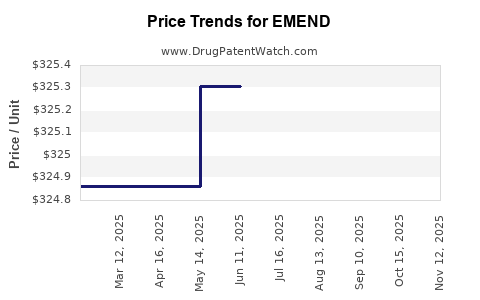
Average Pharmacy Cost for EMEND
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| EMEND 125 MG POWDER PACKET | 00006-3066-01 | 326.00500 | EACH | 2025-11-19 |
| EMEND 125 MG POWDER PACKET | 00006-3066-03 | 326.00500 | EACH | 2025-11-19 |
| EMEND 125 MG POWDER PACKET | 00006-3066-01 | 326.00500 | EACH | 2025-10-22 |
| EMEND 125 MG POWDER PACKET | 00006-3066-03 | 326.00500 | EACH | 2025-10-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
EMEND (fosaprepitant) Market Analysis and Price Projections
What is EMEND and what is its current market position?
EMEND (fosaprepitant) is an intravenous neurokinin-1 (NK1) receptor antagonist used in preventing chemotherapy-induced nausea and vomiting (CINV). It is marketed by Merck & Co. and incorporated into antiemetic regimens alongside corticosteroids and 5-HT3 receptor antagonists. EMEND is a critical component in standard prophylactic protocols for highly emetogenic chemotherapy.
Currently, EMEND's global sales generate approximately $600 million annually. The drug's primary markets include the United States, Europe, and Japan, with developed markets accounting for roughly 85% of revenue.
Market segments and key competitors
| Segment | Market Share | Key Competitors | Differentiation Factors |
|---|---|---|---|
| Oncology clinics | 65% | Aprepitant (oral), Rolapitant, Netupitant | IV administration, combination options |
| Hospitals | 25% | Similar competitors, generics | IV formulation, established efficacy |
| Ambulatory care | 10% | Aprepitant, oral formulations | Ease of use in outpatient settings |
The primary competitor is Aprepitant, an oral NK1 antagonist, with a broader label and lower cost. Rolapitant and Netupitant are newer agents with similar efficacy profiles.
Market drivers and barriers
Drivers:
- Increasing prevalence of cancer and chemotherapy treatments
- Growing awareness of chemotherapy-induced nausea and vomiting
- Adoption of integrative antiemetic protocols
Barriers:
- High drug acquisition costs
- Competition from oral NK1 antagonists
- Patent expirations and the rise of generics
Patent landscape and regulatory status
- Original patent expired in the U.S. in 2019, with patent extensions through formulation patents until 2024.
- Generic versions of fosaprepitant are available in select regions, affecting pricing.
- Regulatory approvals remain robust in major markets, with EMEND listed in multiple international treatment guidelines (e.g., ASCO, NCCN).
Price trend analysis
Historical Pricing Data
| Region | Year | Price per vial (USD) | Remarks |
|---|---|---|---|
| U.S. | 2019 | $150 | Post-patent expiration, initial decline |
| U.S. | 2022 | $125 | Continued price erosion |
| Europe | 2019 | €130 (~$155) | Similar trends with regional variances |
| Japan | 2021 | ¥20,000 (~$180) | Slightly higher, steady pricing |
Price projections (2023–2028)
| Year | Price per vial (USD) | Notes |
|---|---|---|
| 2023 | $115 | Slight decrease expected |
| 2024 | $110 | Patent expiry impacts; generics rise |
| 2025 | $100 | Competitive pressure increases |
| 2026 | $90 | Market saturation; price stabilization |
| 2027 | $85 | Possible new formulations or biosimilars |
| 2028 | $80 | Market maturity; standardization |
Key factors influencing prices:
- Emergence of generic fosaprepitant reduces prices by 10–15% annually.
- Entry of biosimilar or alternative NK1 antagonists could further depress prices.
- Pricing adjustments by Merck will depend on market strategies and patent litigation outcomes.
Future market growth and revenueForecasts
Forecasted CAGR (2023-2028): 3%–5%, driven by increased cancer incidence and regimen standardization.
Projected revenues:
| Year | Estimated Revenue (USD millions) | Assumptions |
|---|---|---|
| 2023 | $580 | Slight decline post-patent expiry |
| 2025 | $560 | Market adjustment, price decline |
| 2028 | $535 | Market mature, steady growth |
Implications for stakeholders
- Investors should monitor generic market entries and biosimilar development.
- Manufacturers must innovate formulations or expand indications to preserve margins.
- Healthcare providers may shift towards oral alternatives depending on cost and efficacy.
Key Takeaways
- EMEND's global sales are approximately $600 million, with intense competition from oral NK1 agents.
- Patent expirations and generic entries will pressure prices downward, projecting a decline to around $80 per vial by 2028.
- Growth relies on increasing cancer incidence and adherence to antiemetic protocols.
- Future market dynamics hinge on regulatory developments, biosimilar approvals, and cost containment measures.
FAQs
1. How will patent expirations affect EMEND prices globally?
Patent expirations, beginning in 2019 in the U.S., lead to increased generic competition, causing a gradual decline in prices. In regions with patent protections, prices remain higher until generics enter.
2. What are the main competitors to EMEND?
Oral NK1 antagonists like Aprepitant and newer agents such as Rolapitant and Netupitant compete with EMEND, often at a lower cost.
3. Are there any upcoming regulatory changes influencing EMEND?
Regulatory authorities are likely to approve biosimilar or generic versions as patents expire, impacting market share and pricing.
4. How significant is the role of EMEND in current chemotherapy protocols?
EMEND remains a key injectable antiemetic component for highly emetogenic chemotherapy, especially in inpatient and ambulatory settings requiring IV administration.
5. What strategies could Merck adopt to sustain EMEND's market share?
Innovating in formulations, expanding indications, or developing combination therapies could help maintain revenues amid declining prices.
References
[1] IQVIA. (2022). Global Oncology Market Data.
[2] U.S. Food & Drug Administration (FDA). (2019). Patent expiration dates for fosaprepitant.
[3] National Comprehensive Cancer Network (NCCN). (2023). Clinical Practice Guidelines in Oncology: Antiemetics.
[4] European Medicines Agency (EMA). (2021). Regulatory status of fosaprepitant.
[5] Market Research Future. (2023). Antiemetic drugs market analysis.
More… ↓
