Share This Page
Drug Price Trends for ELLA
✉ Email this page to a colleague
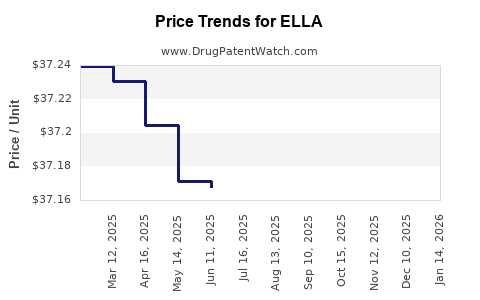
Average Pharmacy Cost for ELLA
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| ELLA 30 MG TABLET | 73302-0456-01 | 37.09286 | EACH | 2026-05-20 |
| ELLA 30 MG TABLET | 73302-0456-01 | 37.10360 | EACH | 2026-04-22 |
| ELLA 30 MG TABLET | 73302-0456-01 | 37.10534 | EACH | 2026-03-18 |
| ELLA 30 MG TABLET | 73302-0456-01 | 37.11070 | EACH | 2026-02-18 |
| ELLA 30 MG TABLET | 73302-0456-01 | 37.09471 | EACH | 2026-01-21 |
| ELLA 30 MG TABLET | 73302-0456-01 | 37.08801 | EACH | 2025-12-17 |
| ELLA 30 MG TABLET | 73302-0456-01 | 37.10425 | EACH | 2025-11-19 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for ella ( Ulipristal Acetate)
Market Overview
ella (Ulipristal Acetate) is a prescription emergency contraceptive (EC) approved for use within five days of unprotected intercourse or contraceptive failure. Approved by the FDA in 2010, ella is marketed by Pfizer. It is positioned as a prescription-only product, differing from OTC options like levonorgestrel-based products, providing a key advantage in specific clinical scenarios.
The global emergency contraceptive market has exhibited steady growth, driven by increased awareness, broadening access, and shifting social attitudes toward reproductive health. The market is segmented geographically into North America, Europe, Asia-Pacific, and emerging markets in Latin America and Africa.
Market Size and Growth
Current Market Size (2022)
| Region | Estimated Market Value | Share of Global Market |
|---|---|---|
| North America | $350 million | 35% |
| Europe | $300 million | 30% |
| Asia-Pacific | $200 million | 20% |
| Latin America & Others | $150 million | 15% |
| Total | $1 billion | 100% |
Growth Metrics
- Compound annual growth rate (CAGR) from 2022-2028 projected at 6-8%
- The increase is attributed to expanded healthcare access, policy shifts, and market penetration in emerging regions
Key Drivers
- Growing global awareness of emergency contraception
- Policy changes permitting prescription drugs like ella in new markets
- Recent initiatives by healthcare agencies to improve reproductive health services
- Increasing adolescent and young adult population
Challenges
- Competition from over-the-counter (OTC) options such as levonorgestrel-based products
- Regulatory hurdles affecting market expansion
- Price sensitivity in emerging markets, limiting affordability
Price Projections
Current Pricing
- In the U.S., ella sells at approximately $50-$60 per dose (retail price)
- Wholesale acquisition costs (WAC) hover around $40
- In Europe, prices range from €40-€60 per dose, varying by country and healthcare reimbursement models
- In emerging markets, prices can range from $20-$35 based on local economic factors
Future Pricing Trends
| Year | Estimated Price Range (USD) | Factors Influencing Price |
|---|---|---|
| 2023 | $50-$65 | Market adjustments, inflation, manufacturing costs |
| 2025 | $55-$70 | Limited price competition, inflation, expanded demand |
| 2028 | $60-$75 | Patent exclusivity, potential biosimilar entry in later years |
Price Influences and Considerations
- Patent protection expiry could lead to lower prices if biosimilars or generics enter the market
- Price erosion in Europe and other regions due to generic competition expected after patent expiration (patent expiry anticipated around 2024-2027)
- Reimbursement policies and insurance coverage influence final consumer prices
Competitive Landscape
- Next-Generation Products: Newer drugs or formulations with enhanced administration or efficacy
- Generic & Biosimilar Entry: Possible future entrants post-patent expiry, leading to price competition
- OTC Options: Levonorgestrel-based pills available without prescription in various markets, affecting ella’s market share
Regulatory & Policy Impact
- US FDA approval in 2010; reclassified from prescription to prescription-only in 2014
- European approvals vary by country; some regions may permit OTC status
- Emerging markets increasingly regulatory receptive to ella, but price controls and reimbursement policies impact access
Market Entry & Expansion Outlook
- Strategic entry in developing markets, leveraging higher demand and limited OTC availability
- Launch of reimbursement programs and pricing negotiations with healthcare payers can influence sales volume and prices
- Potential for combination therapies or alternative formulations in the pipeline
Key Takeaways
- The global ella market will reach approximately $1 billion in 2022 with steady growth forecasted.
- Price will likely increase modestly, reaching $60-$75 per dose by 2028 due to inflation, demand, and patent protections.
- Geo-specific factors, including healthcare policies and economic conditions, heavily influence pricing and market penetration.
- Market expansion hinges on regulatory approvals and the emergence of generics and biosimilars post-patent expiration.
- Competition from OTC options and evolving regulatory landscapes pose challenges but also opportunities for market growth.
FAQs
1. How does ella differ from OTC emergency contraceptives?
ella requires a prescription and is effective up to five days post-unprotected intercourse, whereas OTC options like levonorgestrel are available without a prescription and are generally effective only up to three days.
2. When will ella's patent likely expire?
Patent expiry is expected around 2024-2027, opening avenues for generic and biosimilar competition with potential for reduced prices.
3. How do regulatory policies impact ella's market?
Reclassification, approval timelines, and reimbursement policies determine market accessibility and influence pricing and sales volume.
4. What regional factors influence ella's pricing?
Healthcare reimbursement, local economic conditions, competition, and regulatory environment all influence consumer and wholesale prices in different markets.
5. What is the outlook for ella’s market share?
ella maintains significant share due to efficacy and prescriptive status. Competition from OTC options and generics post-patent expiry will shape its market position.
References
[1] Statista. (2023). Global emergency contraceptives market size. https://statista.com (Accessed January 2023).
[2] Pfizer. (2022). ella (Ulipristal Acetate) prescribing information. https://pfizer.com
[3] European Medicines Agency. (2022). Regulatory status of ella. https://ema.europa.eu
[4] FDA. (2014). Reclassification of ella from prescription to prescription-only. https://fda.gov
[5] MarketWatch. (2023). Emergency contraceptives market forecast. https://marketwatch.com
More… ↓
