Share This Page
Drug Price Trends for EFFER-K
✉ Email this page to a colleague
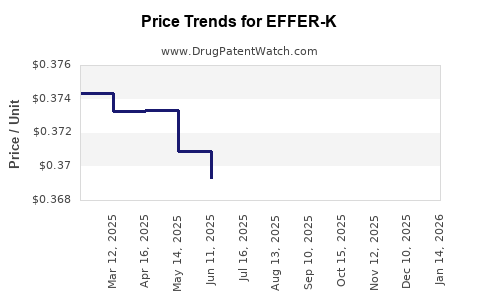
Average Pharmacy Cost for EFFER-K
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| EFFER-K 10 MEQ TABLET EFF | 51801-0014-30 | 0.45421 | EACH | 2026-03-18 |
| EFFER-K 20 MEQ TABLET EFF | 51801-0012-30 | 0.52231 | EACH | 2026-03-18 |
| EFFER-K 25 MEQ TABLET EFF | 51801-0001-01 | 0.36772 | EACH | 2026-03-18 |
| EFFER-K 20 MEQ TABLET EFF | 51801-0011-30 | 0.52231 | EACH | 2026-03-18 |
| EFFER-K 25 MEQ TABLET EFF | 51801-0007-30 | 0.36772 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market analysis and price projections for EFFER-K
Introduction
EFFER-K is a potassium supplement indicated primarily for treating hypokalemia. It is marketed under various brand names, including EFFER-K by Novartis. As a supplement, it often faces different pricing dynamics and regulatory considerations compared to prescription medications. This analysis evaluates the current market landscape, competitive environment, regulatory factors, and price projections.
Market Overview
- Indication and Usage: EFFER-K is used to restore potassium levels in patients with hypokalemia caused by diuretics, poor diet, or other medical conditions.
- Market Size: The global oral potassium supplements market was valued at approximately USD 1.35 billion in 2022 and is projected to reach USD 1.75 billion by 2030, growing at a CAGR of 3.5% (SMR, 2022). EFFER-K captures a significant share within this segment, especially in North America and Europe.
- Key Competitors:
- Generic potassium chloride formulations
- Brand-specific products such as Klor-Con, Micro-K, and Potassium Chloride ER.
- Non-oral forms, including intravenous potassium formulations.
Regulatory Environment
- FDA Regulations: As an over-the-counter (OTC) product in the U.S., EFFER-K maintains FDA approval for marketing. Prescription potassium formulations are separately regulated.
- Pricing Constraints: OTC supplements are subject to less strict pricing controls, yet reimbursement mechanisms influence retail pricing.
Pricing Dynamics
- Current Prices:
- Retail prices vary widely by region and packaging.
- In the U.S., a 30-count bottle of EFFER-K (15 mEq per tablet) retails between USD 10–15.
- Generics are priced similarly, with minor variations.
| Product Type | Typical U.S. Retail Price (USD) | Packaging Details |
|---|---|---|
| EFFER-K | 10–15 | 30 tablets, 15 mEq each |
| Micro-K (brand) | 12–18 | 30–40 tablets, 10–15 mEq each |
| Generic potassium chloride | 8–12 | Similar packaging, often lower-priced |
- Reimbursement & Insurance: OTC prices are not reimbursed; prescriptions are subject to insurance coverage, impacting patient access and overall market dynamics.
Market Drivers and Barriers
- Drivers:
- Rising prevalence of hypokalemia linked to diuretic use, chronic kidney disease, and gastrointestinal losses.
- Aging population increasing demand for electrolyte management.
- Increasing consumer awareness of supplement options.
- Barriers:
- Competition from lower-cost generics.
- Consumer preference shift toward intravenous formulations in clinical settings.
- Regulatory scrutiny on supplement labeling.
Pricing Projections (2023–2030)
- Short-term (2023–2025):
- Prices are expected to remain stable, around USD 10–15 per bottle in the U.S.
- Slight margin compression due to increasing generic competition.
- Medium-term (2025–2028):
- Marginal price declines anticipated as generics gain market share.
- Potential for price stabilization if brand innovation or formulation improvements occur.
- Long-term (2028–2030):
- Prices could decrease by 10–20% if market saturation occurs.
- Alternatively, premium formulations or combination products could command higher prices, offsetting generic pressure.
Factors Influencing Future Prices
- Market Entry of New Formulations: Development of slow-release or combination electrolyte products may increase prices.
- Regulatory Changes: Stricter OTC labeling or safety monitoring could impact pricing.
- Reimbursement Policies: Changes in insurance coverage or formularies could influence retail prices and consumer access.
- Supply Chain Dynamics: Raw material costs, manufacturing efficiencies, and distribution costs affect pricing margins.
Conclusion
EFFER-K operates within a mature market, with stable current retail prices and moderate growth prospects. Price trends are mainly influenced by generic competition, regulatory factors, and market demand driven by aging populations and chronic disease prevalence. Future pricing will depend on product innovation, market share shifts, and policy environments.
Key Takeaways
- EFFER-K’s retail pricing in the U.S. remains around USD 10–15 per bottle.
- Market growth is driven by hypokalemia prevalence and aging populations.
- Generic competition and regulatory factors will likely lead to modest price declines over time.
- Innovation in formulation or delivery methods could sustain higher price points.
- Reimbursement dynamics heavily influence market accessibility and consumer prices.
FAQs
1. How does EFFER-K’s pricing compare to other potassium supplements?
It is generally priced similarly to other branded OTC supplements, with retail prices around USD 10–15 for typical packaging. Generics are usually slightly cheaper.
2. What factors could cause EFFER-K’s price to increase in the future?
Product innovations, such as sustained-release formulations, or branding strategies could support higher prices. Regulatory changes and supply chain improvements also influence pricing.
3. Are insurance reimbursements common for EFFER-K?
No, EFFER-K is an OTC product, so reimbursement is uncommon unless prescribed and covered under insurance plans for specific medical conditions.
4. How does the global market outlook affect EFFER-K?
Increased demand for electrolyte replenishment globally supports consistent sales. Competitive pressures in emerging markets may lead to price reductions or generic product expansion.
5. What is the main competitive threat to EFFER-K?
Generic potassium chloride products, which are often priced lower, pose a significant competition in both retail and prescription markets.
Citations
[1] SMR, "Global Oral Potassium Supplements Market," 2022.
[2] U.S. FDA, "Over-the-Counter (OTC) Drug Review," 2022.
[3] MarketWatch, "Potassium Supplements Market Size, Share & Trends," 2022.
More… ↓
