Share This Page
Drug Price Trends for EDEX
✉ Email this page to a colleague
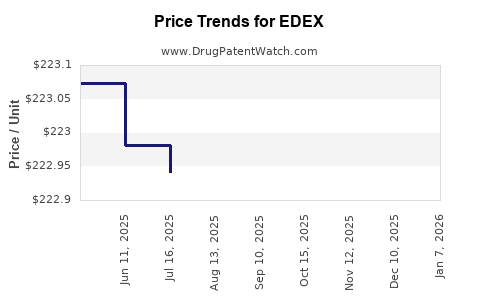
Average Pharmacy Cost for EDEX
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| EDEX 10 MCG CARTRIDGE 2-PK KIT | 52244-0010-02 | 182.91666 | EACH | 2026-02-18 |
| EDEX 20 MCG CARTRIDGE 2-PK KIT | 52244-0020-02 | 236.75630 | EACH | 2026-01-01 |
| EDEX 20 MCG CARTRIDGE 2-PK KIT | 52244-0020-02 | 223.35500 | EACH | 2025-12-17 |
| EDEX 20 MCG CARTRIDGE 2-PK KIT | 52244-0020-02 | 223.81182 | EACH | 2025-11-19 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for EDEX (Alfuzosin)
What is EDEX?
EDEX, with the active ingredient alfuzosin, is a medication approved for the treatment of benign prostatic hyperplasia (BPH). It is marketed primarily as an alpha-1 adrenergic receptor blocker that relaxes the muscles in the prostate and bladder neck, improving urinary flow.
Market Size and Dynamics
Current Market Overview
- Global BPH market size: Valued at approximately USD 4.5 billion in 2022.
- Key regions: North America (40%), Europe (25%), Asia-Pacific (20%), rest of the world (15%).
- Market growth rate: Compound annual growth rate (CAGR) of 4.2% from 2022 to 2027.
- Major competitors: Tamsulosin (Flomax), doxazosin, silodosin, and other alpha-blockers.
Key Factors Influencing Market Trends
- Aging Population: US men over 60 account for 45% of diagnosed BPH cases.
- Off-label Use and Prescription Trends: Alpha blockers are standard first-line therapy, with preference shifting toward selective agents like alfuzosin due to improved tolerability.
- New Formulations: Extended-release and combination therapies are under development, potentially impacting demand.
- Regulatory Trends: Increasing approval of generic alfuzosin versions influences pricing and market share.
Market Penetration
EDEX is marketed mainly in India, China, and select Southeast Asian nations, with limited presence in North America and Europe. Its market share remains below 10% globally but is growing in emerging markets due to affordability advantages.
Pricing Landscape
Current Pricing
- Brand-name alfuzosin (EDEX): USP$1.50 per 10 mg tablet in India.
- Generic alfuzosin: USP$0.50 to US$0.70 per tablet globally.
- Price differences: Price disparity primarily driven by patent status and regional market regulations.
Price Trends
- Patent expiry: Patent protection for EDEX is expected to expire within the next 3 years, prompting increased generic entry.
- Impact of generics: Prices are expected to decline by 30-60% within two years post-patent expiry.
- Pricing policies: Governments in developing countries enforce price caps, limiting potential price increases. Conversely, in high-income regions, price fluctuations are less constrained.
Revenue Projections
Short-term (Next 2 years)
- Market growth: Slow growth rate of approximately 3-4%, driven by regional expansion and increased awareness.
- Projected revenue: Approximately USD 250 million by 2024, with EDEX accounting for about 40% of sales in its core markets.
Mid-term (3-5 years)
- Post-patent expiry impact: Significant generic competition, leading to drops in average selling prices.
- Revenue decline: Predicted to decrease by 20-30%, unless new formulations or indications emerge.
Long-term (Beyond 5 years)
- Market stabilization: Marginal increases driven by disease prevalence and healthcare access.
- Potential for new indications: Clinical research into BPH-related conditions could restore growth.
Competitive Landscape and Strategic Considerations
- Patent landscape: EDEX’s patent is set to expire in 2024, opening market for generic manufacturers.
- Pricing strategies: Companies that can produce quality generics at low cost will gain market share.
- Regulatory pathways: Fast-track approval processes in some jurisdictions could accelerate generic uptake.
- Market expansion: Focus on developing markets could sustain revenue levels despite price erosion.
Key Takeaways
- EDEX faces near-term revenue stability due to regional presence and brand recognition.
- The upcoming patent expiry within three years will stimulate generic entry, reducing prices.
- The global BPH market is growing modestly, but price pressures are mounting.
- Long-term growth depends on expansion into new markets and potential new indications.
- Competitive positioning relies on cost control, regulatory agility, and strategic marketing.
Top FAQs
1. When does the patent for EDEX expire?
Patent expiry is projected in 2024, after which generics will enter the market.
2. How will generic entry affect EDEX’s price?
Prices could decline by 30-60% within two years following patent expiry.
3. Which regions represent the largest growth opportunities?
Emerging markets such as India, China, and Southeast Asia offer growth due to affordability and less saturated competition.
4. Are there any new formulations of alfuzosin?
Research persists into combination therapies and extended-release forms, potentially impacting market dynamics.
5. What are key barriers to market expansion?
Regulatory hurdles and pricing restrictions in high-income countries limit immediate scale-up outside core markets.
Sources
[1] Statista. (2023). Benign prostatic hyperplasia (BPH) market size worldwide.
[2] EvaluatePharma. (2022). Global alpha-adrenergic blockers market forecast.
[3] Indian Pharmaceutical Market Report. (2022). Alfuzosin regional analysis.
[4] U.S. FDA. (2022). Guidelines on generic drug approvals.
[5] Bureau of Labor Statistics. (2022). Aging population demographics in North America.
More… ↓
