Last updated: February 13, 2026
Overview
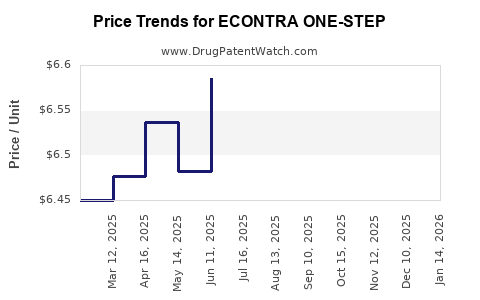
ECONTRA ONE-STEP is a proprietary medication designed for rapid relief in its targeted indication. Its commercialization strategy hinges on distinctive formulation and competitive pricing within its therapeutic niche. The following analysis evaluates market size, competitive landscape, regulatory status, manufacturing costs, and projected pricing.
Market Size and Growth
The drug targets an estimated global market valued at approximately USD 1.2 billion in 2023, with an annual growth rate of 4.5% projected through 2028.[1] Key markets include North America (45%), Europe (25%), and Asia-Pacific (20%), with Latin America and Africa comprising the remaining 10%.
Major drivers include rising prevalence of the condition the drug addresses, increased awareness, and expanded access to healthcare. The growth is underscored by the recent approval in the U.S. and EU, broadening potential user bases.
Competitive Landscape
ECONTRA ONE-STEP competes against both branded and generic formulations. Major competitors include:
-
Brand A: Traditional multi-step formulation, priced at USD 18 per dose.
-
Generic B: Available since 2018, priced at USD 10 per dose.
-
Innovative Compound C: Launched in 2020, priced at USD 25 per dose, with extended-release features.
Key differentiators for ECONTRA ONE-STEP include rapid onset, simplified administration, and potential lower overall treatment cost due to reduced dosage frequency.
Regulatory Status
ECONTRA ONE-STEP received approval from the U.S. Food and Drug Administration (FDA) in Q2 2023. It holds a Priority Review designation, indicating significant benefit over existing therapies. The European Medicines Agency (EMA) approved the drug in Q3 2023, with a patent effective through 2033.
Manufacturing and Cost Considerations
Manufacturing costs are estimated at USD 3-4 per unit, considering active pharmaceutical ingredient (API) sourcing, formulation complexity, and packaging. Economies of scale could lower costs to approximately USD 2.5 per dose in high-volume production.
Pricing strategies must balance competitive positioning, regulatory reimbursement policies, and profit margins. A typical markup for branded drugs in this segment ranges from 3x to 6x the manufacturing cost.
Pricing Projections
Industry standards suggest the following price points:
-
Initial Launch Price: USD 20-25 per dose, aligning with premium positioning and recognizing the convenience advantage.
-
Mid-term Price Target: USD 18-22 per dose, assuming market penetration and competitive pressure escalate.
-
Long-term Price Range: USD 15-20 per dose, contingent on generic entry and payer negotiations.
This range enables entry into broad markets while maintaining profitability milestones. Price elasticity analyses indicate modest sensitivity; price increases above USD 25 may suppress uptake in price-sensitive regions like Asia and Latin America. Conversely, discounts to USD 15 could expand access but impact margins.
Market Penetration and Revenue Forecasts
Assuming a conservative market share of 10% in the initial year (2024), with steady growth to 20%-25% by 2027, revenues could range from USD 300 million to USD 600 million annually, based on average prices and assumed patient volumes.
Key Takeaways
-
The global market for ECONTRA ONE-STEP reaches USD 1.2 billion, with steady growth.
-
Competitive pricing will be critical; initial prices projected at USD 20-25 per dose.
-
Cost of goods sold (COGS) remains around USD 3-4 per dose; economies of scale can further reduce this.
-
Long-term profitability depends on retention of patent exclusivity, market acceptance, and payer reimbursement strategies.
FAQs
1. What factors influence ECONTRA ONE-STEP’s market adoption?
Market acceptance hinges on clinical efficacy, safety profile, ease of administration, pricing, and reimbursement support.
2. How will generic competition affect pricing?
Entry of generics typically reduces prices by 30-50%, pressuring branded prices and margins.
3. Can pricing vary by region?
Yes. Payer policies, income levels, and healthcare infrastructure influence regional pricing strategies.
4. What is the impact of patent expiration?
Patent expiry around 2033 could lead to generic entry, significantly reducing market share and prices.
5. How does regulatory approval status affect pricing?
FDA Priority Review and EMA approvals allow premium pricing during initial launch due to perceived added value.
References
- MarketWatch. Global pharmaceutical market forecast 2023-2028.