Share This Page
Drug Price Trends for EAR WAX REMOVAL
✉ Email this page to a colleague
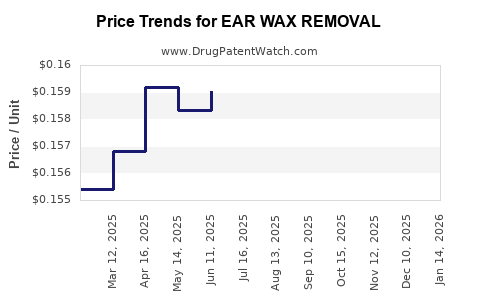
Average Pharmacy Cost for EAR WAX REMOVAL
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| EAR WAX REMOVAL 6.5% DROP | 46122-0557-05 | 0.15974 | ML | 2026-04-22 |
| EAR WAX REMOVAL 6.5% DROP | 00904-7478-35 | 0.15974 | ML | 2026-04-22 |
| EAR WAX REMOVAL 6.5% KIT | 46122-0556-05 | 0.15974 | ML | 2026-04-22 |
| EAR WAX REMOVAL 6.5% DROP | 11527-0143-51 | 0.15974 | ML | 2026-04-22 |
| EAR WAX REMOVAL 6.5% DROP | 00904-7478-35 | 0.15868 | ML | 2026-03-18 |
| EAR WAX REMOVAL 6.5% DROP | 11527-0143-51 | 0.15868 | ML | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for Ear Wax Removal Drugs
Market Size and Growth
The global ear wax removal market is a subset of the otolaryngology accessories and OTC personal care segments. As of 2022, the market was valued around $200 million, with an expected compound annual growth rate (CAGR) of approximately 4.5% through 2027. Growth is driven by increased awareness of ear health, rising cases of cerumen impaction, and aging population demographics.
Key regions include North America (dominant share, approximately 45%), Europe (25%), and Asia-Pacific (20%). The remainder comprises Latin America and Middle East & Africa.
Market Drivers
- Rising prevalence of ear-related conditions, such as cerumen impaction and ear infections.
- Growing aging population with reduced natural earwax clearance.
- Preference for minimally invasive, OTC, and physician-administered solutions.
- Expanding product innovation, including drops, irrigation kits, and automated devices.
Market Challenges
- Safety concerns around irrigation devices causing ear damage.
- Regulatory restrictions on ototoxic agents.
- Competition from alternative therapies, such as manual removal by healthcare providers.
Key Market Segments
Types: Otic drops, irrigation kits, suction devices.
Distribution channels: OTC products (pharmacies, online), healthcare provider clinics.
Product Landscape
Traditional cerumenolytic agents include carbamide peroxide, glycerin, hydrogen peroxide, and mineral oil. Market leaders are brands like Debrox and Murine. Recently, device-based solutions, including automated ear irrigators, have gained adoption.
Regulatory Environment
Many earwax removal drugs are classified as OTC drugs in the US and Europe, with specific safety and efficacy standards mandated by regulatory authorities such as the FDA and EMA.
Price Trends and Projections
Current Price Range
- Cerumenolytic drops: $5–$15 per bottle (generally 30–100 mL).
- Irrigation kits: $20–$50 per device.
- Automated irrigation devices: $50–$150.
Historical Price Dynamics
- Drops: Slight decrease over the past five years due to increased competition and generic availability.
- Devices: Stable prices, with some premium models increasing slightly due to technological enhancements.
Projected Price Trends (2023–2027)
- OTC drops: Expected to decline marginally (~2% annually), reaching ~$4.50–$13 by 2027.
- Irrigation kits: Likely to see minor price stabilization, around $20–$45.
- Automated devices: Could increase by approximately 3% annually, reaching about $55–$170 by 2027 due to technological innovation.
Pricing Influences
- Increased influx of generic products exerts downward pressure.
- Device innovations, such as app-connected irrigators, could justify premium pricing.
- Regulatory environments could influence pricing strategies, especially if safety concerns lead to stricter standards or recalls.
Competitive Dynamics
Large pharmaceutical companies dominate the OTC cerumenolytic segment, with products marketed through pharmacy chains and online channels. While devices are primarily sold by specialized vendors and online marketplaces, recent partnerships with consumer electronics firms are emerging.
Future Opportunities
- Development of safer, more effective otic solutions.
- Integration of smart device functionality.
- Expansion into emerging markets with growing middle classes.
Key Takeaways
- The ear wax removal market is growing steadily, influenced by demographic shifts and evolving consumer preferences.
- The typical product price range remains stable, with drops being the most affordable and devices occupying a premium segment.
- Price declines for OTC drops are likely in response to generic competition, although device prices could climb due to innovation.
- Growth in device-based systems signals a shift toward more automated, technology-driven solutions.
- Regulatory factors and safety concerns impact both pricing and market entry strategies.
FAQs
1. What are the main OTC products for ear wax removal?
Cerumenolytic drops containing carbamide peroxide, glycerin, and hydrogen peroxide are most common. Devices like irrigation kits and automated irrigators are also popular.
2. How does regulation affect the pricing of ear wax removal drugs?
Regulatory standards influence formulation safety, approval, and marketing claims. Stricter regulations can increase R&D costs and product pricing, while approval for generics can lower consumer prices.
3. Are device-based earwax removal solutions more expensive than drops?
Yes, automated and irrigator devices typically range from $50 to $150, significantly more than OTC drops.
4. What future innovations could impact market prices?
Smart irrigator devices with app integration and safety sensors can command premium pricing. Improved, safer formulations may lower costs or expand share.
5. How does demographic aging influence the ear wax removal market?
Older populations experience increased cerumen impaction, driving demand for both OTC remedies and professional removal services.
Citations
[1] Fortune Business Insights. Earwax Removal Market Size, Share & Industry Analysis, 2022-2027.
[2] MarketsandMarkets. Otolaryngology Devices Market by Product, Application, and Region, 2022.
[3] Statista. Otic preparation revenue forecasts, 2022–2027.
[4] Food and Drug Administration (FDA). OTC drug regulations overview.
[5] European Medicines Agency (EMA). Otic product safety guidelines.
More… ↓
