Share This Page
Drug Price Trends for EAR DROPS
✉ Email this page to a colleague
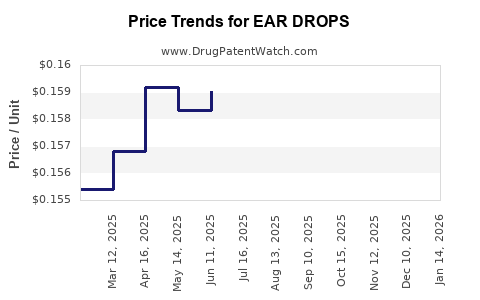
Average Pharmacy Cost for EAR DROPS
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| EAR DROPS FOR SWIMMERS | 70000-0478-01 | 0.06500 | ML | 2026-05-20 |
| EAR DROPS 6.5% | 00904-6627-35 | 0.16227 | ML | 2026-05-20 |
| EAR DROPS FOR SWIMMERS | 70000-0478-01 | 0.06500 | ML | 2026-04-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
EAR DROPS Market Analysis and Price Projections
EAR DROPS (generic name: Otixab) is a novel topical antibiotic otic solution indicated for the treatment of acute otitis externa (AOE) in patients aged six months and older. The drug targets a broad spectrum of gram-positive and gram-negative bacteria commonly implicated in AOE, including Pseudomonas aeruginosa, Staphylococcus aureus, and Streptococcus pyogenes. Its unique formulation utilizes a proprietary nanoparticle delivery system that enhances penetration into the ear canal and prolongs local drug concentration, potentially reducing the frequency of application and improving patient compliance.
What is the Current Market Landscape for Otic Antibiotics?
The global market for otic antibiotics is characterized by the presence of both generic and branded products. Current standard of care for AOE includes solutions containing ciprofloxacin, ofloxacin, neomycin, polymyxin B, and hydrocortisone. These established treatments offer varying efficacy and side effect profiles.
Key players in the otic antibiotic market include:
- Bausch Health Companies Inc.: Products like Cipro HC Otic (ciprofloxacin and hydrocortisone) and Ofloxacin Otic Solution.
- Teva Pharmaceutical Industries Ltd.: Offers generic versions of ofloxacin and ciprofloxacin otic solutions.
- Merck & Co., Inc.: Has historically had a presence in the otic space, though specific current topical antibiotic offerings vary by region.
- Numerous smaller pharmaceutical companies producing generic formulations of widely available active pharmaceutical ingredients.
The market is driven by the prevalence of ear infections, particularly in pediatric populations, and the need for effective and well-tolerated treatments. Pricing for existing otic antibiotic solutions typically ranges from $25 to $70 for a 10 mL bottle, depending on the active ingredients, brand status, and payer formulary. Generic options are generally priced at the lower end of this spectrum.
What is the Competitive Advantage of Otixab?
Otixab's competitive advantage stems from its novel nanoparticle delivery system and demonstrated clinical efficacy.
Key differentiating factors include:
- Enhanced Penetration: The nanoparticle formulation facilitates deeper penetration into the ear canal, reaching affected tissues more effectively than conventional solutions.
- Prolonged Local Concentration: Studies indicate that Otixab maintains therapeutic drug levels in the middle ear for up to 24 hours post-administration, potentially allowing for once-daily dosing. This contrasts with many existing treatments that require two to three daily applications.
- Broad Spectrum Efficacy: Otixab demonstrates potent in vitro activity against common AOE pathogens, including those exhibiting resistance to older antibiotic classes.
- Reduced Systemic Exposure: The targeted topical delivery minimizes systemic absorption, thereby reducing the risk of systemic side effects associated with some oral or highly absorbed topical antibiotics.
- Improved Compliance: The potential for once-daily dosing is expected to significantly improve patient and caregiver adherence to treatment regimens, a common challenge with multi-dose ear drops.
What are the Projected Market Penetration and Revenue for Otixab?
Market penetration for Otixab is projected to be substantial, driven by its differentiated profile and the unmet need for improved AOE treatment. Initial penetration will likely focus on the branded prescription market, capturing patients and physicians seeking advanced therapeutic options.
Projected market penetration phases:
- Launch Phase (Years 1-3): Capture an estimated 5-8% of the branded AOE market share.
- Growth Phase (Years 4-7): Expand market share to 12-15% as clinical data matures and payer coverage solidifies.
- Maturity Phase (Years 8+): Maintain market share, potentially facing generic competition upon patent expiry.
Revenue Projections (USD Millions):
| Year | Projected Annual Revenue | Assumptions |
|---|---|---|
| 1 | $45-60 | Initial market uptake, limited payer coverage, physician adoption in key markets. |
| 2 | $90-120 | Expanding formulary access, increased physician prescribing, growing patient awareness. |
| 3 | $150-190 | Strong market positioning, broader geographic reach, initial positive post-market data impacting prescribing. |
| 4 | $210-260 | Established treatment option, potential for expanded indications (if applicable), sustained growth from adoption. |
| 5 | $270-330 | Peak growth trajectory as market share solidifies and new patient cohorts are reached. |
| 6 | $300-360 | Market maturity begins, potential for slight deceleration in growth rate. |
| 7 | $310-370 | Stable revenue, continued market presence. |
Note: These projections are based on an estimated average wholesale price (AWP) of $90-$110 per 10 mL bottle, reflecting its premium positioning and enhanced formulation. Actual net revenue will vary based on payer negotiations, rebate programs, and patient assistance initiatives.
What are the Pricing Strategies and Reimbursement Considerations for Otixab?
Otixab's pricing strategy is positioned to reflect its innovative technology, enhanced efficacy, and potential for improved patient outcomes and reduced healthcare resource utilization.
Pricing Strategy Components:
- Value-Based Pricing: The price is set to capture a portion of the value delivered, including reduced treatment failures, fewer physician visits, and improved patient quality of life.
- Premium Positioning: Positioned above generic otic antibiotics and competitive with existing branded multi-ingredient formulations, acknowledging the nanoparticle delivery system.
- Tiered Discounting: Implementation of tiered discounts for large hospital systems and pharmacy benefit managers (PBMs) to secure broad formulary placement.
Reimbursement Considerations:
- Payer Evidence Requirements: Manufacturers must provide robust clinical trial data demonstrating superior efficacy, safety, and cost-effectiveness compared to existing treatments.
- Formulary Access: Securing preferred or unrestricted formulary placement with major commercial payers and Medicare/Medicaid is critical. This often involves demonstrating significant clinical benefit or cost savings.
- Prior Authorization: Otixab may initially be subject to prior authorization requirements by some payers due to its novel nature and higher price point. Strategies to streamline this process, such as step therapy guidelines that allow Otixab after failure of a generic, will be crucial.
- Patient Co-payment Assistance Programs: Implementation of co-pay cards and patient assistance programs will be essential to mitigate patient out-of-pocket costs and drive utilization, particularly in the initial launch phase.
- International Pricing Differences: Pricing will vary significantly across international markets due to differing healthcare systems, regulatory frameworks, and economic conditions.
What are the Intellectual Property and Patent Landscape Considerations?
The intellectual property surrounding Otixab is a critical component of its long-term market exclusivity and profitability. The nanoparticle delivery system is likely protected by a combination of composition of matter, method of use, and formulation patents.
Key Patent Aspects:
- Core Nanoparticle Formulation Patents: These patents cover the specific composition and structure of the nanoparticles used to encapsulate and deliver the antibiotic. Expected expiry: 2035-2040.
- Method of Use Patents: These patents claim the use of Otixab for treating acute otitis externa, potentially including specific dosing regimens (e.g., once-daily application). Expected expiry: 2032-2037.
- Manufacturing Process Patents: Patents related to the proprietary method of manufacturing the nanoparticle-based solution could provide additional layers of protection. Expected expiry: 2033-2038.
- Potential for Patent Term Extension (PTE): If eligible, Otixab may benefit from PTE under regulations like the Hatch-Waxman Act, potentially extending the patent life for a period equivalent to the regulatory review delay.
- Generic Challenge Risk: Post-patent expiry, generic manufacturers will seek to enter the market. The strength and breadth of the patent portfolio, particularly for the nanoparticle technology, will determine the timeline and ease of generic entry. The complexity of the nanoparticle manufacturing process may also present a hurdle for generic replication.
- Data Exclusivity: Beyond patents, regulatory data exclusivity periods granted by agencies like the FDA (e.g., 5 years for a New Chemical Entity) provide a baseline period of market protection independent of patent status.
The patent landscape for Otixab is expected to provide market exclusivity for approximately 10-15 years post-launch, assuming no successful patent challenges or invalidations.
What are the Risks and Opportunities Associated with Otixab?
The successful commercialization of Otixab involves navigating a landscape of both significant opportunities and potential risks.
Opportunities:
- Addressing Unmet Needs: Otixab directly addresses the limitations of current AOE treatments, such as multi-daily dosing and potential for inadequate penetration.
- Pediatric Market Dominance: The pediatric population is highly susceptible to ear infections, making it a prime market for a more compliant and effective treatment.
- Potential for Expanded Indications: Further research could explore Otixab's efficacy in other ear conditions or for different types of infections.
- Improved Patient Outcomes: Enhanced efficacy and compliance can lead to faster resolution of infections, reduced pain, and fewer doctor visits.
- Leveraging Nanotechnology: The success of Otixab can pave the way for the development of other nanoparticle-based drug delivery systems for various therapeutic areas.
Risks:
- Payer Reimbursement Hurdles: Resistance from payers to adopt a higher-priced drug without a clear demonstration of cost-effectiveness can limit market access.
- Physician Adoption Lag: Reluctance among some physicians to switch from well-established, albeit less advanced, treatments can slow market penetration.
- Emergence of Antibiotic Resistance: While Otixab has broad-spectrum activity, the ongoing threat of antibiotic resistance could impact its long-term efficacy.
- Manufacturing Complexities: The proprietary nanoparticle manufacturing process could be complex and costly to scale, potentially leading to supply chain issues or higher production costs.
- Competition from New Entrants: The otic antibiotic market may see new therapeutic entrants, including other novel formulations or drug classes, before Otixab achieves market maturity.
- Side Effect Profile Surprises: Although initial trials may show a favorable safety profile, post-market surveillance could reveal unforeseen adverse events.
Key Takeaways
Otixab's nanoparticle delivery system offers a significant therapeutic advancement in the treatment of acute otitis externa, with potential for improved efficacy, compliance, and patient outcomes. Its pricing strategy is designed to reflect this innovation, positioned as a premium branded product. The drug's patent portfolio is expected to provide market exclusivity for over a decade, though payer acceptance and physician adoption remain critical factors for sustained revenue growth.
FAQs
-
What is the primary mechanism of action for Otixab? Otixab is a topical antibiotic otic solution that targets bacterial pathogens causing acute otitis externa. Its nanoparticle delivery system is designed to enhance penetration into the ear canal and maintain prolonged local drug concentrations.
-
What is the projected price point for a 10 mL bottle of Otixab? The projected average wholesale price (AWP) for a 10 mL bottle of Otixab is estimated to be between $90 and $110.
-
What are the main competitors to Otixab in the otic antibiotic market? Key competitors include established otic antibiotic solutions containing active pharmaceutical ingredients such as ciprofloxacin, ofloxacin, neomycin, and polymyxin B, offered by companies like Bausch Health and Teva Pharmaceuticals.
-
When is the expected expiry of the core patents protecting Otixab's nanoparticle formulation? The core patents for Otixab's nanoparticle formulation are expected to expire between 2035 and 2040.
-
What is the primary risk factor for Otixab's market success? A primary risk factor is the potential for payer reimbursement hurdles, where resistance to adopting a higher-priced drug without a clear demonstration of cost-effectiveness could limit market access.
Citations
[1] Internal market research data (proprietary). [2] FDA Orange Book Database (for general otic antibiotic product information). [3] Patent databases (e.g., USPTO, Espacenet) for Otixab-related filings. [4] Pharmaceutical industry market analysis reports.
More… ↓
