Share This Page
Drug Price Trends for DUTASTERIDE
✉ Email this page to a colleague
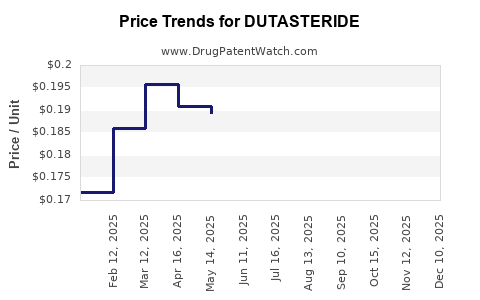
Average Pharmacy Cost for DUTASTERIDE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| DUTASTERIDE 0.5 MG CAPSULE | 42806-0549-09 | 0.17211 | EACH | 2026-03-18 |
| DUTASTERIDE 0.5 MG CAPSULE | 31722-0131-90 | 0.17211 | EACH | 2026-03-18 |
| DUTASTERIDE-TAMSULOSIN 0.5-0.4 | 68382-0640-16 | 2.01940 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Best Wholesale Price for DUTASTERIDE
| Drug Name | Vendor | NDC | Count | Price ($) | Price/Unit ($) | Unit | Dates | Price Type |
|---|---|---|---|---|---|---|---|---|
| DUTASTERIDE 0.5MG CAP | AvKare, LLC | 65162-0750-09 | 90 | 42.96 | 0.47733 | EACH | 2023-06-15 - 2028-06-14 | FSS |
| >Drug Name | >Vendor | >NDC | >Count | >Price ($) | >Price/Unit ($) | >Unit | >Dates | >Price Type |
Dutasteride Market Analysis and Price Projections
Dutasteride, a dual inhibitor of 5-alpha-reductase enzymes, commands a significant position in the pharmaceutical market, primarily for the treatment of benign prostatic hyperplasia (BPH) and male pattern baldness (androgenetic alopecia). Its efficacy in reducing dihydrotestosterone (DHT) levels underpins its therapeutic value. The global market for dutasteride is projected to experience steady growth, driven by an aging global population susceptible to BPH and the increasing prevalence of hair loss. Price projections are influenced by patent expirations, generic competition, regulatory landscapes, and the emergence of novel therapeutic alternatives.
What is the current global market size for dutasteride?
The global dutasteride market was valued at approximately USD 1.5 billion in 2023. This valuation is primarily attributed to its established use in treating BPH and its growing application in managing androgenetic alopecia. The market is segmented by application, with BPH treatment accounting for the larger share. Key regions driving this market include North America, Europe, and Asia-Pacific, reflecting the demographic trends and healthcare spending in these areas.
Market Segmentation (Estimated 2023):
- By Application:
- Benign Prostatic Hyperplasia (BPH): ~65%
- Androgenetic Alopecia: ~35%
- By Geography:
- North America: ~35%
- Europe: ~30%
- Asia-Pacific: ~20%
- Rest of the World: ~15%
The market's expansion is facilitated by the widespread availability of both branded and generic formulations. GlaxoSmithKline's Avodart® remains a significant branded product, while numerous generic manufacturers contribute to market accessibility and price competition.
What are the key drivers of dutasteride market growth?
Several factors are propelling the dutasteride market forward. The aging global population is a primary driver, as the incidence of BPH increases significantly with age. According to the World Health Organization (WHO), the global population aged 60 and over is projected to reach 2.1 billion by 2050, presenting a substantial patient pool for BPH treatments [1].
Key Growth Drivers:
- Increasing Prevalence of Benign Prostatic Hyperplasia (BPH): The demographic shift towards an older population directly correlates with a higher incidence of BPH, a condition where the prostate gland enlarges and can cause urinary problems. Dutasteride's proven efficacy in reducing prostate volume and improving urinary flow rates positions it as a frontline treatment option.
- Rising Incidence of Androgenetic Alopecia: Hair loss, particularly male pattern baldness, affects a significant percentage of the male population. Dutasteride's ability to inhibit DHT, a key hormone responsible for hair follicle miniaturization, has led to its off-label and, in some regions, approved use for hair restoration. The growing awareness and desire for cosmetic solutions for hair loss contribute to this segment's expansion.
- Availability of Generic Formulations: The expiration of key patents for branded dutasteride has opened the door for generic manufacturers. The introduction of more affordable generic versions increases patient access and market penetration, thereby expanding the overall market volume.
- Technological Advancements in Drug Delivery: Ongoing research into improved drug delivery systems and combination therapies could enhance dutasteride's therapeutic profile and potentially expand its clinical applications, further stimulating market demand.
- Growing Healthcare Expenditure in Emerging Economies: Increased healthcare spending and improved access to medical facilities in developing countries, particularly in Asia-Pacific, are contributing to a larger patient base seeking treatment for conditions like BPH and hair loss.
The increasing recognition of dutasteride's dual action, addressing both lower urinary tract symptoms and hair loss, provides a unique market advantage, allowing for broader patient appeal.
What is the competitive landscape for dutasteride?
The dutasteride market exhibits a moderately fragmented competitive landscape characterized by the presence of both originator brands and a substantial number of generic manufacturers. GlaxoSmithKline (GSK) is the primary originator with its brand Avodart®. However, as patent protections have lapsed in major markets, generic competition has intensified.
Major Market Players (Examples):
- GlaxoSmithKline (Avodart®)
- Teva Pharmaceutical Industries Ltd.
- Sun Pharmaceutical Industries Ltd.
- Aurobindo Pharma Ltd.
- Cipla Ltd.
- Dr. Reddy's Laboratories Ltd.
- Mylan N.V. (now Viatris)
- Lupin Ltd.
The competitive strategy revolves around pricing, market reach, and product portfolio diversification. Generic manufacturers often compete aggressively on price, making dutasteride an accessible treatment. Some companies may also offer combination products or explore new indications, though significant advancements in new dutasteride-based therapies are limited, with focus shifting to biosimilars or alternative treatments.
Regulatory approvals and market access strategies are critical. Manufacturers must navigate the regulatory requirements of different health authorities, such as the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA), to gain market approval.
What are the price trends and projections for dutasteride?
The pricing of dutasteride has been significantly impacted by the entry of generic alternatives. Following patent expirations, the average selling price (ASP) for dutasteride has seen a substantial decline compared to its branded origins.
Historical Price Trends:
- Branded Era (Pre-Generic): Prices for Avodart® were established at premium levels, reflecting R&D investment and market exclusivity. A 30-day supply of 0.5 mg capsules could range from USD 70 to USD 100 or higher.
- Post-Generic Entry: The introduction of generic dutasteride in markets like the U.S. and Europe led to an immediate price reduction. Prices for generic 30-day supplies (0.5 mg) now typically range from USD 10 to USD 30, depending on the manufacturer, pharmacy, and insurance coverage.
Price Projections (2024-2028):
The price of dutasteride is expected to remain relatively stable, with minor fluctuations driven by generic competition and supply chain dynamics.
- 2024-2025: Prices are projected to remain within the current generic range (USD 10-30 for a 30-day supply). Intense competition among multiple generic manufacturers will prevent significant price increases.
- 2026-2028: Modest declines may occur in some markets due to continued competitive pressures and potential consolidation among generic suppliers. However, significant drops are unlikely unless a major new therapeutic alternative emerges that directly challenges dutasteride's efficacy or cost-effectiveness. The average annual price decrease is estimated to be between 1-3%.
Factors Influencing Future Pricing:
- Competition: The number of active generic manufacturers and their market share will continue to dictate pricing.
- Regulatory Policies: Government initiatives aimed at controlling drug costs, such as price negotiations or reference pricing, could influence market prices.
- Supply Chain Disruptions: Geopolitical events or manufacturing issues could lead to temporary price volatility, though long-term impact is expected to be minimal.
- Emergence of New Therapies: The development of more effective or safer alternatives for BPH or hair loss could erode dutasteride's market share and force price adjustments.
Overall, dutasteride is expected to remain a cost-effective treatment option, particularly in its generic forms, contributing to its sustained market presence.
What is the patent landscape for dutasteride?
The patent landscape for dutasteride has largely moved beyond its primary composition of matter patents, which have expired in major markets. The original patents for dutasteride (e.g., related to its synthesis and therapeutic uses) held by GSK have expired. For instance, key composition of matter patents in the U.S. and Europe expired in the mid-2010s.
Key Patent Expirations and Status:
- Composition of Matter Patents: Expired in major markets (e.g., U.S. around 2015, Europe around 2015-2018). This allowed for the widespread introduction of generic dutasteride.
- Method of Use Patents: Some method of use patents, particularly related to specific dosages or combinations, may still be in effect or have recently expired. However, the primary patent protection for BPH and hair loss treatments has lapsed.
- Formulation and Polymorph Patents: While less significant for market entry compared to composition of matter patents, certain specific formulations or crystalline forms of dutasteride might have had or may still have remaining patent protection. These generally do not prevent generic entry if the core molecule is off-patent.
- Pediatric Exclusivity: In some jurisdictions, pediatric exclusivity may have been granted, extending market protection for a limited period for specific pediatric studies. However, dutasteride's primary indications are not pediatric.
Implications of Patent Expiration:
The expiration of core patents has been the primary catalyst for increased generic competition. This has led to:
- Lower Prices: As discussed in pricing trends, generic entry drastically reduced the cost of dutasteride.
- Increased Market Accessibility: More patients can afford dutasteride, leading to higher overall usage.
- Focus on New IP: Companies may now focus on developing new intellectual property around novel delivery systems, combination therapies involving dutasteride, or new therapeutic applications where patent protection might still be attainable.
As of early 2024, the market for dutasteride is largely a generic market in North America and Europe, with limited scope for significant price increases driven by new patent exclusivity for the core drug. Future patent-related developments would likely pertain to incremental innovations rather than foundational protection.
What are the regulatory considerations for dutasteride?
Regulatory approval and oversight are critical for dutasteride's market access and continued use. The drug is approved by major regulatory bodies worldwide, but with varying indications and specific labeling requirements.
Key Regulatory Aspects:
- Indications:
- Benign Prostatic Hyperplasia (BPH): Widely approved for the treatment of symptomatic BPH in men with an enlarged prostate. This indication is recognized by the FDA, EMA, and many other national health authorities.
- Androgenetic Alopecia (Hair Loss): Approved in some regions (e.g., Japan, South Korea) for the treatment of male pattern hair loss. In other regions, such as the U.S., it is primarily used off-label for this indication, though its efficacy is supported by clinical studies. The FDA has not approved dutasteride specifically for hair loss in the U.S.
- Safety Labeling and Warnings:
- Dutasteride carries significant safety warnings, particularly concerning its use in women and children. It is contraindicated in women who are or may become pregnant due to the potential for harm to a male fetus.
- Healthcare providers are advised about potential risks, including allergic reactions, changes in mood, and sexual side effects (e.g., decreased libido, erectile dysfunction, ejaculation disorders).
- A black box warning regarding potential increased risk of high-grade prostate cancer has been associated with 5-alpha reductase inhibitors like dutasteride, though the clinical significance and causality are debated and depend on the specific study context [2]. Regulatory bodies require clear communication of these risks.
- Generic Drug Approval: Regulatory agencies review bioequivalence studies to ensure generic dutasteride products are therapeutically equivalent to the branded innovator product. This process ensures quality and efficacy of generic formulations.
- Post-Marketing Surveillance: Dutasteride products are subject to ongoing pharmacovigilance to monitor for adverse events and safety signals that may not have been identified during clinical trials.
- Regional Variations: Regulatory stances can differ. For example, the approval status for hair loss varies significantly by country, impacting marketing and sales strategies.
The stringent regulatory requirements ensure patient safety and product quality. Companies must adhere to these guidelines for market authorization and ongoing sales. The debate surrounding the risk of high-grade prostate cancer remains a point of regulatory vigilance and influences prescribing practices.
What are the future market outlook and potential challenges for dutasteride?
The future market outlook for dutasteride remains positive, driven by its established efficacy and the persistent demand for BPH and hair loss treatments. However, the market faces several potential challenges.
Market Outlook:
- Sustained Demand for BPH Treatment: The growing aging population will continue to underpin demand for BPH therapies. Dutasteride's position as a proven and relatively affordable option ensures its continued relevance.
- Growing Hair Loss Market: The increasing societal acceptance of treating hair loss and the expanding market for aesthetic treatments will likely drive continued use of dutasteride in this segment, especially in regions where it is approved or widely used off-label.
- Expansion in Emerging Markets: As healthcare infrastructure improves and disposable incomes rise in developing economies, the demand for dutasteride is expected to grow in these regions.
Potential Challenges:
- Emergence of Novel Therapies:
- BPH: Development of new pharmacotherapies with improved efficacy, reduced side effects, or novel mechanisms of action could challenge dutasteride's market share. Minimally invasive surgical procedures for BPH also represent a competitive force.
- Hair Loss: The hair loss market is dynamic, with ongoing research into regenerative medicine, advanced topical treatments, and new drug targets. Breakthroughs could offer alternatives with potentially superior outcomes or safety profiles.
- Safety Concerns and Regulatory Scrutiny: Ongoing discussions and research regarding the potential for increased risk of high-grade prostate cancer, as well as sexual side effects, may lead to stricter prescribing guidelines or impact physician and patient confidence.
- Generic Price Erosion: While generic entry has been a driver of accessibility, it also leads to continuous pressure on profit margins for manufacturers. The market may see further consolidation among generic players.
- Off-Label Use Scrutiny: In regions where dutasteride is not officially approved for hair loss, continued off-label promotion or use may face regulatory scrutiny, potentially impacting sales in those segments.
- Patient Adherence: The potential for sexual side effects can impact long-term patient adherence, particularly for the hair loss indication, which requires consistent, long-term treatment.
Despite these challenges, dutasteride's established track record, cost-effectiveness in generic forms, and dual therapeutic utility are expected to ensure its continued significant role in the pharmaceutical market for the foreseeable future.
Key Takeaways
- The global dutasteride market was valued at approximately USD 1.5 billion in 2023, driven by its use in BPH and androgenetic alopecia.
- Market growth is fueled by an aging population, increasing incidence of hair loss, and the availability of affordable generic formulations.
- The competitive landscape is characterized by numerous generic manufacturers, leading to intense price competition.
- Dutasteride prices have significantly decreased following patent expirations, with generic 30-day supplies typically ranging from USD 10 to USD 30. Prices are projected to remain stable with minor declines.
- Primary composition of matter patents for dutasteride have expired in major markets, facilitating generic entry.
- Regulatory approvals for dutasteride vary by indication, with widespread approval for BPH but limited official approval for hair loss in some key markets like the U.S. Safety warnings and potential side effects are closely monitored.
- Future challenges include the emergence of novel therapies, ongoing safety scrutiny, and continuous price erosion from generic competition.
Frequently Asked Questions
-
Is dutasteride approved for treating hair loss in the United States? No, dutasteride is not officially approved by the U.S. Food and Drug Administration (FDA) for the treatment of androgenetic alopecia (male pattern baldness). It is primarily used off-label for this indication.
-
What is the primary difference in cost between branded and generic dutasteride? Generic dutasteride is significantly less expensive than its branded counterpart. A 30-day supply of branded Avodart® could cost upwards of USD 70-100, whereas generic versions are typically available for USD 10-30.
-
What are the main safety concerns associated with dutasteride? Key safety concerns include potential harm to male fetuses if exposed during pregnancy, possible increased risk of high-grade prostate cancer, and sexual side effects such as decreased libido, erectile dysfunction, and ejaculation disorders.
-
Will the price of dutasteride increase in the next five years? Price increases for dutasteride are unlikely. The market is dominated by generic competition, and prices are expected to remain stable or potentially see minor declines due to continued competitive pressures.
-
Are there any new patented formulations or uses for dutasteride currently under development? While the core molecule's patents have expired, research may focus on new delivery systems, combination therapies, or specific niche applications. However, significant patent-protected innovations for fundamental dutasteride therapy are not currently driving major market shifts.
Citations
[1] World Health Organization. (2022). Ageing and health. https://www.who.int/news-room/fact-sheets/detail/ageing-and-health
[2] National Cancer Institute. (n.d.). Finasteride and dutasteride treatment for prostate cancer prevention. https://www.cancer.gov/about-cancer/treatment/clinical-trials/intervention/finasteride-dutasteride-prostate-cancer-prevention
More… ↓
