Share This Page
Drug Price Trends for DULOXETINE HCL DR
✉ Email this page to a colleague
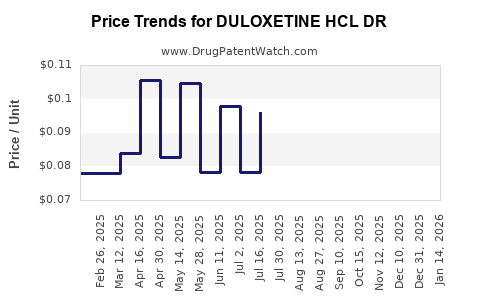
Average Pharmacy Cost for DULOXETINE HCL DR
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| DULOXETINE HCL DR 60 MG CAP | 82009-0173-10 | 0.11362 | EACH | 2026-03-18 |
| DULOXETINE HCL DR 40 MG CAP | 82009-0172-30 | 1.04771 | EACH | 2026-03-18 |
| DULOXETINE HCL DR 30 MG CAP | 82009-0171-10 | 0.08485 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for Duloxetine HCl DR
What is the current market landscape for Duloxetine HCl DR?
Duloxetine hydrochloride delayed-release (DR) is an SNRI (serotonin-norepinephrine reuptake inhibitor) approved for major depressive disorder, generalized anxiety disorder, diabetic peripheral neuropathy, and fibromyalgia. The drug is marketed under brands such as Cymbalta, which held approximately 90% of the global market share as of 2022.
Global sales in 2022 reached approximately $4.3 billion, with the U.S. accounting for about 80%. The drug's patent expired in Europe in 2014 and in the U.S. in 2013, leading to increased generic competition. As of 2023, generic versions account for a significant portion of prescriptions, impacting pricing and market volume.
What is the competitive landscape?
The primary competitors are other SNRI drugs—Venlafaxine, Milnacipran, and Desvenlafaxine—and alternative antidepressants such as SSRIs. The entry of generics has led to price erosion, with average wholesale prices (AWP) declining by approximately 35% since 2013.
Key market players include:
- Eli Lilly (original patent holder)
- Teva Pharmaceuticals (generic)
- Mylan (generic)
- Amneal (generic)
Market dynamics shift with patent expiries, regulatory approvals, and formulary placements. The trend toward biosimilarity or alternative therapies in depression management could lead to further market compression.
How are pricing trends evolving?
Patent Impact and Generic Entry
The patent expiry spurred a decrease in the drug’s average retail price. The 2022 average wholesale price for the branded drug was approximately $300 per month. Generic versions are priced around $150 to $200 per month, representing a cost reduction of approximately 33-50%.
Price projections (next 5 years)
| Year | Projected Average Wholesale Price (AWP) | Market Dynamics |
|---|---|---|
| 2023 | $140 - $180 | Increased generic penetration, stable demand |
| 2024 | $130 - $170 | Entry of biosimilar competition, insurance negotiations |
| 2025 | $120 - $160 | Potential new formulation approvals, market saturation |
| 2026 | $115 - $150 | Prices stabilize as competition reaches equilibrium |
| 2027 | $110 - $140 | Slower price erosion, possible uptake of biosimilars |
Factors influencing future prices:
- Regulatory approvals for new formulations and extended-release versions may influence premium pricing.
- Market penetration of generics continues to suppress prices.
- Healthcare reimbursement policies favor cost-effective medications, pushing prices downward.
- Pipeline drugs could replace or cannibalize duloxetine use if they demonstrate superior efficacy or safety.
What does the demand forecast look like?
Global demand for duloxetine (including all formulations) was approximately 20 million monthly prescriptions in 2022. The annual growth rate fluctuates around 2-3%, primarily driven by the aging population and increased awareness of mental health disorders.
Generics are capturing greater market share, which suppresses revenue for branded products. Despite this, steady demand persists, supported by chronic disease management and off-label uses.
What are the key risks and opportunities?
Risks
- Patent challenges and litigation may lead to further generic entry.
- Pricing pressure from payers and formulary exclusions.
- New therapies with better efficacy or safety profiles could replace duloxetine.
- Regulatory delays or failures in developing new formulations.
Opportunities
- Development of extended-release formulations can command higher prices.
- Market expansion into emerging economies, where antidepressant use is rising.
- Line extension development for specific indications such as urinary incontinence.
Summary
Duloxetine HCl DR's market is characterized by significant generic competition, leading to declining prices. Next five-year projections indicate a downward trend, with prices stabilizing at approximately $110 to $140 per month. Demand remains stable but faces headwinds from alternative therapies and healthcare cost containment policies.
Key Takeaways
- Patents expired in 2013-2014, leading to generics accounting for most prescriptions.
- Average wholesale prices declined by roughly 35% since patent expiry.
- Price projections show continued decline, stabilizing near $110-$140/month by 2027.
- Revenue potential hinges on new formulations and pipeline development.
- Market risk factors include competitive pressures and regulatory shifts.
FAQs
-
When did the patent for Cymbalta expire?
The U.S. patent for Cymbalta expired in 2013; European patents expired in 2014. -
How much do generic duloxetine products cost?
Around $150 to $200 per month, representing a significant reduction from branded prices. -
What are the main alternative drugs to duloxetine?
Venlafaxine, Desvenlafaxine, Milnacipran, and SSRIs like sertraline. -
Are there new formulations in development?
Several companies are developing extended-release versions, potentially commanding higher prices. -
What is the outlook for duloxetine's market share?
It will diminish over time due to generic competition but remain relevant in specific clinical scenarios and formulations.
References
[1] IQVIA. (2022). Global Market Summary for Antidepressants.
[2] FDA. (2022). Approved Drug Products with Therapeutic Equivalence Evaluations.
[3] EvaluatePharma. (2023). Generic Market Trends.
[4] Secondary research reports, MarketLine. (2023).
[5] U.S. Food and Drug Administration. (2014). Patent expirations and generic approvals.
More… ↓
