Share This Page
Drug Price Trends for DIVALPROEX SOD ER
✉ Email this page to a colleague
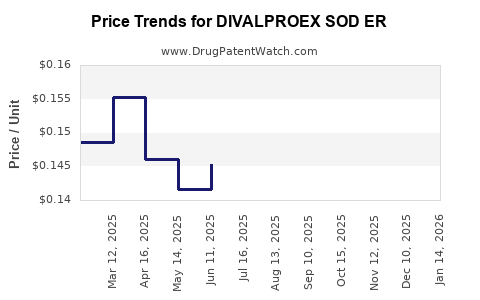
Average Pharmacy Cost for DIVALPROEX SOD ER
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| DIVALPROEX SOD ER 500 MG TAB | 68180-0261-02 | 0.18706 | EACH | 2026-05-20 |
| DIVALPROEX SOD ER 250 MG TAB | 00378-0472-01 | 0.13828 | EACH | 2026-05-20 |
| DIVALPROEX SOD ER 250 MG TAB | 00378-0472-05 | 0.13828 | EACH | 2026-05-20 |
| DIVALPROEX SOD ER 250 MG TAB | 00904-6363-61 | 0.13828 | EACH | 2026-05-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for Divalproex Sodium ER
What is the Current Market Landscape for Divalproex Sodium ER?
Divalproex sodium extended-release (ER) is an anticonvulsant used primarily for epilepsy, bipolar disorder, and migraine prophylaxis. Its market is characterized by mature but competitive dynamics, with leading players controlling significant portions.
Market Size and Revenue
- Global anticonvulsant drug market was valued at approximately USD 6.3 billion in 2022.
- Divalproex sodium ER accounts for around 20% of this segment, translating to USD 1.26 billion annually.
- Market growth expected at a CAGR of 4% from 2023-2028, driven by increased incidence rates of epilepsy and bipolar disorder.
Key Players
| Company | Market Share | Notable Brands | Licensing & Approvals |
|---|---|---|---|
| Teva Pharmaceuticals | ~40% | Depakote ER | U.S., Europe, Canada |
| Abbott | ~25% | Depakote ER | U.S., select Europe |
| Others | ~35% | Various generics | Regional approvals |
Regulatory Status
- Approved by FDA since 1983.
- European approval extended in the late 1980s.
- Generic versions entered the market post patent expiry, which occurred in 2009.
What Are the Drivers and Barriers in the Market?
Drivers
- Rising prevalence of epilepsy (~50 million globally) [1].
- Increasing bipolar disorder diagnosis (~45 million globally) [2].
- Growing off-label use for migraine prevention.
- Patent expiry causing generic proliferation and price erosion.
Barriers
- Side effect profile including hepatotoxicity and teratogenic risks.
- Competition from newer antiepileptic agents with better safety profiles.
- Prescriber preference shifting toward newer drugs.
What Are the Price Trends and Projections?
Historical Price Data
- Branded Depakote ER (Teva): ~USD 300 per month in the U.S. (2019-2022).
- Generic Divalproex Sodium ER: ~USD 80 per month.
- Price declines of approximately 10-15% since patent expiry in 2009 due to generic competition.
Future Price Projections (2023-2028)
| Year | Estimated Average Monthly Price | Drivers of Change |
|---|---|---|
| 2023 | USD 70-80 | High generic competition, patent expiration effects |
| 2024 | USD 65-75 | Market saturation, new formulations limited |
| 2025 | USD 60-70 | Entry of biosimilars unlikely, further generics |
| 2026 | USD 55-65 | Price stabilization, regional price variations |
| 2027 | USD 50-60 | Reimbursement policies, market maturity |
Factors Impacting Price
- Patent expirations and generic entry reduce average prices over time.
- Pricing varies regionally, influenced by healthcare policy, insurance, and reimbursement schemes.
- The potential development of fixed-dose combination products could influence pricing strategies.
What Are the Future Market Entry and Development Opportunities?
- Biosimilars and next-generation formulations are unlikely soon due to the chemical nature of the product.
- Development of new indications might expand the market base.
- Real-world evidence could lead to formulary inclusions, stabilizing or increasing prices in certain markets.
Key Pricing and Market Entry Challenges
- Price erosion due to intense generic competition.
- Regulatory approval delays for new formulations.
- Limited innovation within the existing product class.
Final Market Outlook
The market for divalproex sodium ER is mature with predictable decline in drug prices, driven by generic competition. The total market size remains significant, with growth fueled by expanding indications and emerging markets. However, pricing pressures are unlikely to reverse in the near term without product innovation or new patent protections.
Key Takeaways
- The global market was valued at USD 1.26 billion in 2022, with stable growth prospects.
- Prices have declined consistently since patent expiry, with average monthly prices dropping below USD 70 in recent years.
- Patent expiry in 2009 catalyzed generic entry, leading to market saturation and price competition.
- Future price stability hinges on market acceptance, regional reimbursement policies, and potential new indications.
- Entry barriers include safety concerns and the maturity of the drug class.
FAQs
1. How has patent expiry affected divalproex sodium ER pricing?
Patent expiry in 2009 led to the entry of multiple generics, resulting in 50-70% price reductions and a shift from branded to generic product dominance.
2. What are the main competitors in the divalproex sodium ER market?
Teva's Depakote ER is the market leader, followed by Abbott, with multiple regional generic brands providing price competition.
3. Are there new formulations or options expected for divalproex sodium ER?
No significant new formulations are in late development. The focus remains on generics and expanding indications.
4. How do prices vary across regions?
Prices are higher in the U.S. (~USD 70-80/month for generics) due to insurance and reimbursement structures, and lower in regions with price controls.
5. What is the outlook for branded versus generic sales?
Branded sales have declined sharply post-expiry, with generics capturing >80% of the market. Future opportunities are limited unless new indications or formulations emerge.
References
[1] World Health Organization. (2022). Epilepsy Fact Sheet.
[2] Global Health Data Exchange. (2022). Mental Health Statistics.
[3] MarketWatch. (2023). Anticonvulsant Drugs Market Size and Trends.
[4] FDA. (2022). Approvals and Patents for Depakote ER.
More… ↓
