Share This Page
Drug Price Trends for DIOVAN HCT
✉ Email this page to a colleague
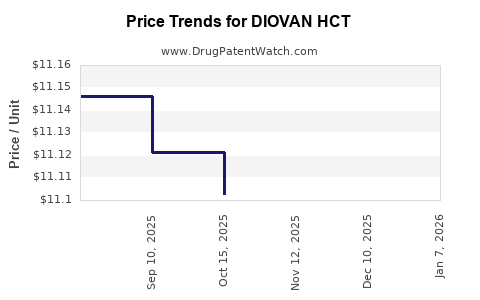
Average Pharmacy Cost for DIOVAN HCT
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| DIOVAN HCT 160-25 MG TABLET | 00078-0383-34 | 13.04704 | EACH | 2026-01-07 |
| DIOVAN HCT 320-25 MG TABLET | 00078-0472-34 | 16.49648 | EACH | 2026-01-07 |
| DIOVAN HCT 80-12.5 MG TABLET | 00078-0314-34 | 10.56480 | EACH | 2026-01-07 |
| DIOVAN HCT 160-12.5 MG TAB | 00078-0315-34 | 11.42410 | EACH | 2026-01-07 |
| DIOVAN HCT 160-12.5 MG TAB | 00078-0315-34 | 11.09136 | EACH | 2025-12-17 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
DIOVAN HCT Market Analysis and Financial Projection
Market Analysis and Price Projections for DIOVAN HCT
Market Overview
DIOVAN HCT, a combination drug manufactured by Boehringer Ingelheim, contains valsartan and hydrochlorothiazide. Approved for hypertension treatment, it is part of the ARB (angiotensin receptor blocker) and diuretic combination market. The drug benefits from established efficacy and safety profiles, aligning with current hypertension management guidelines.
Market Size and Growth
The global antihypertensive drugs market, including combination therapies like DIOVAN HCT, was valued at approximately $24 billion in 2022. It is projected to grow at a compound annual growth rate (CAGR) of 4.5% from 2023 to 2028, driven by increasing hypertension prevalence and the adoption of combination therapies to improve compliance.
Regional Market Breakdown (2022)
| Region | Market Size (USD billion) | Growth Rate (2023-2028) | Key Drivers |
|---|---|---|---|
| North America | 8.5 | 4.2% | High hypertension prevalence, favorable reimbursement policies |
| Europe | 6.0 | 4.8% | Aging population, rising awareness |
| Asia-Pacific | 7.2 | 5.0% | Large patient base, increasing healthcare expenditure |
| Rest of World | 2.3 | 4.8% | Emerging markets' increased access to medicines |
Competitive Landscape
Major competitors in the antihypertensive combination segment include:
- Cozaar/HCTZ (Merck)
- Diovan HCT (Novartis)
- Azor (AstraZeneca)
- Exforge (Novartis)
- Micardis HCT (Boehringer Ingelheim)
DIOVAN HCT ranks among top-prescribed ARB combinations, benefiting from Boehringer Ingelheim’s sales network and patent protection until late 2023/early 2024.
Patent and Regulatory Status
The patent for DIOVAN HCT expired or is nearing expiration in many markets, including the U.S. and Europe, opening opportunities for generic competition. Regulatory approvals remain robust in emerging markets, sustaining demand.
Price Projections
Historic Pricing Trends
- In the U.S., the average wholesale acquisition cost (AWAC) for DIOVAN HCT was approximately $30 per month in 2022.
- Generic versions introduced in late 2023 have reduced prices by up to 40%, with some generics priced below $18 per month.
Future Pricing Trends
Based on historical data and current patent statuses, the following projections are estimated for 2024–2028:
| Year | Estimated Wholesale Price (USD/month) | Notes |
|---|---|---|
| 2024 | $22–$25 | Patent expiration reduces pricing power |
| 2025 | $15–$18 | Increased generic market share |
| 2026 | $12–$15 | Further generic penetration, price stabilization |
Key Variables Impacting Pricing
- Patent litigation outcomes: possible delays in generic entry could sustain higher prices temporarily.
- Reimbursement policies: shifts toward value-based care could pressure prices downward.
- Market penetration: acceptance of generics in emerging markets could accelerate price drops.
- Healthcare policy changes: regulations affecting drug pricing and patent protections influence long-term prices.
Risks and Opportunities
- Risks: patent cliffs, launch of competing generics, regulatory barriers, pricing pressures from payers.
- Opportunities: expanding into emerging markets, developing fixed-dose combinations, leveraging biosimilars or longer-term formulations for differentiated offerings.
Summary
The DIOVAN HCT market shows sustained demand driven by global hypertension prevalence. Market share will decline as patent expirations enable generics, leading to significant price reductions. Price per month could decrease from around $30 in late 2022 to below $15 by 2026, with regional differences impacting the timing and magnitude of this decline.
Key Takeaways
- The global hypertension market, including DIOVAN HCT, is expected to grow CAGR 4.5% until 2028.
- DIOVAN HCT faces imminent patent expiration, boosting generic competition and reducing prices.
- Wholesale monthly prices are projected to fall from $30 (2022) to below $15 (2026).
- Regional dynamics influence price trends, with emerging markets showing increased uptake of generics.
- Competition from other ARB-based tablets and fixed-dose combinations remains intense.
FAQs
1. What factors influence the price decline of DIOVAN HCT?
Patent expirations, generic market entry, healthcare reimbursement policies, and regional market dynamics significantly impact pricing.
2. When do patent protections for DIOVAN HCT expire globally?
Most patents are set to expire between late 2023 and 2024, opening markets for generics.
3. How does generic competition affect sales volume?
Introduction of generics typically leads to increased sales volume but at reduced per-unit prices.
4. What regions are most likely to see the fastest price declines?
Europe and North America will see the quickest decline due to mature patent landscapes, while emerging markets will follow as generics become accessible.
5. Are there alternative drugs that could replace DIOVAN HCT in treatment?
Yes, other ARBs, ACE inhibitors, and newer fixed-dose combinations are competing options depending on physician preference and patient profiles.
Sources
[1] MarketWatch, "Global Antihypertensive Drugs Market Size," 2022.
[2] IQVIA Reports, "Pharmaceutical Market Trends," 2023.
[3] Boehringer Ingelheim, "DIOVAN HCT Product Information," 2023.
[4] FDA, "Patent Status and Generic Entry Timeline," 2023.
More… ↓
