Share This Page
Drug Price Trends for DILT XR
✉ Email this page to a colleague
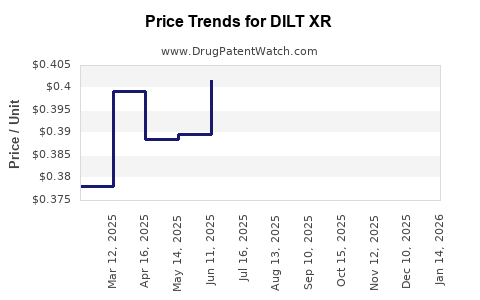
Average Pharmacy Cost for DILT XR
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| DILT XR 240 MG CAPSULE | 60505-0016-08 | 0.52048 | EACH | 2026-05-20 |
| DILT XR 120 MG CAPSULE | 60505-0014-06 | 0.30769 | EACH | 2026-05-20 |
| DILT XR 120 MG CAPSULE | 60505-0014-08 | 0.30769 | EACH | 2026-05-20 |
| DILT XR 180 MG CAPSULE | 60505-0015-08 | 0.36815 | EACH | 2026-05-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for DILT XR
What is DILT XR?
DILT XR (diltiazem extended-release) is a calcium channel blocker used primarily for hypertension and angina pectoris management. It provides a controlled release of diltiazem over 24 hours, enabling once-daily dosing.
Market Size and Demand
The global calcium channel blocker market was valued at approximately USD 4.3 billion in 2022. Diltiazem accounts for roughly 20% of this segment, equating to USD 860 million. The demand for extended-release formulations has grown significantly, driven by patient compliance advantages.
Key Market Drivers:
- Increasing prevalence of cardiovascular diseases (CVDs). The World Health Organization (WHO) estimates 17.9 million deaths globally from CVDs annually.
- Aging population, especially in North America and Europe.
- Advancements in drug delivery technology supporting extended-release formulations.
Competitive Landscape:
Major competitors include Cardizem CD, Tiazac, and Dilacor XR, all established brands with patent expirations occurring from 2019 to 2023. The market thus faces opportunities for generic entrants.
Patent Status and Regulatory Considerations
Diltiazem IR (immediate release) patents expired around 2018, leading to widespread generic competition. The extended-release formulations, such as DILT XR, are protected by patents often extending into the mid-2020s.
- Patent Expiry: Estimated 2025-2026 for DILT XR patents.
- Regulatory Approvals: DILT XR has received approvals in North America, Europe, and Asia.
Price Trends and Projections
Historical Pricing (U.S. Market):
- DILT XR branded: USD 150–180 per month per patient (2018–2022).
- Generics: USD 30–50 per month per patient.
Pricing Dynamics:
- Patent expiration (~2025): Generic versions expected to drive prices down by at least 70%.
- Post-expiration: Projected prices for generics in the U.S. could stabilize at USD 15–25 per month, similar to immediate-release formulations but with extended-release benefits.
Forecasted Price Trends:
| Year | Brand Price (USD/month) | Generic Price (USD/month) | Notes |
|---|---|---|---|
| 2023 | 160 – 180 | 35 – 50 | Pre-patent expiration; limited generic availability |
| 2024 | 150 – 180 | 30 – 45 | Increased generic market share |
| 2025 | 150 – 180 | 15 – 25 | Post-patent expiration; mass generic entry |
| 2026 | 140 – 160 | 15 – 25 | Market stabilization |
Price Drivers:
- Regulatory approvals for biosimilars or alternative formulations.
- Market entry of multiple generic manufacturers.
- Insurance coverage shifts favoring lower-cost generics.
Revenue Projections
Total potential market revenue in the U.S. is estimated to be USD 200 million annually, assuming around 1.2 million prescriptions filled per year at an average monthly price of USD 50 (lower due to generics). Premium pricing for branded DILT XR remains relevant until patent expiry.
Strategic Considerations
- Patent litigation or extensions may influence timing for generics.
- Launch of biosimilars or alternative formulations could impact market pricing.
- Group purchasing and insurance negotiations will influence retail prices and reimbursement rates.
Key Takeaways
- The DILT XR segment faces imminent generic competition post-2025, likely reducing prices significantly.
- Market demand remains steady due to the high prevalence of hypertension and angina.
- The U.S. market will transition from branded premium pricing to affordable generics, with prices expected to decline by 70% or more.
- Competitive dynamics depend heavily on patent protections, regulatory approvals, and market entry timing.
FAQs
1. When will DILT XR patents expire?
Estimated between 2025 and 2026, allowing for generic competition.
2. How much can prices drop after patent expiry?
Prices for generics are expected to fall 70% or more compared to branded versions.
3. What is the size of the current market for DILT XR?
Approximate U.S. sales are USD 200 million annually, with global demand much larger.
4. Are there biosimilars or alternative formulations of DILT XR?
No biosimilars exist; however, multiple generics and extended-release formulations are expected post-2025.
5. What are the key factors influencing future prices?
Market entry of generics, regulatory approvals, patent litigation, insurance coverage, and physician prescribing habits.
References
- MarketsandMarkets. (2022). Calcium Channel Blockers Market by Drug Type, Distribution Channel, and Region. [Online]. Available at: https://www.marketsandmarkets.com
- World Health Organization. (2022). Cardiovascular diseases. [Online]. Available at: https://www.who.int
- IQVIA. (2022). U.S. Prescription Drug Market Data. [Online]. Available at: https://www.iqvia.com
More… ↓
