Share This Page
Drug Price Trends for DIFICID
✉ Email this page to a colleague
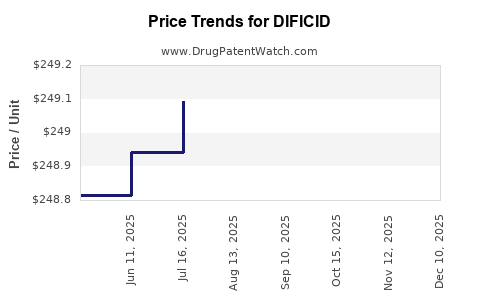
Average Pharmacy Cost for DIFICID
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| DIFICID 200 MG TABLET | 52015-0080-01 | 248.70697 | EACH | 2026-05-20 |
| DIFICID 200 MG TABLET | 52015-0080-01 | 248.69209 | EACH | 2026-04-22 |
| DIFICID 200 MG TABLET | 52015-0080-01 | 248.68579 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
What Is DIFICID and How Does It Fit Into the Market?
DIFICID (fidaxomicin) is an antibiotic approved primarily for the treatment of Clostridioides difficile infections (CDI). It is distinguished from other CDI treatments by its targeted activity, reduced recurrence rate, and favorable safety profile. Approved by the U.S. Food and Drug Administration (FDA) in 2011, DIFICID has gained market share owing to its efficacy in saving patients from recurrent CDI episodes, a major challenge in infection management.
The drug is produced by Merck & Co., with global distribution across multiple territories. The CDI market, though niche, remains sizable due to the high prevalence of CDI, especially in healthcare settings. In the U.S., an estimated 500,000 cases occur annually, with annual healthcare costs exceeding $1 billion.
DIFICID's penetration relies on clinician adoption, insurance coverage, and competition from alternative therapies, notably vancomycin, metronidazole, and newer agents like ridinilazole.
What Are the Current Market Dynamics for DIFICID?
The CDI treatment landscape is characterized by limited but focused competition. Vancomycin and metronidazole remain first-line treatments, with fidaxomicin positioned as an option for patients at higher risk of recurrence.
The approval of new agents and changes in clinical guidelines impact DIFICID's market share. For example, the Infectious Diseases Society of America recommends fidaxomicin for initial episodes of CDI with high recurrence risk. This positioning supports its use but limits broader adoption given cost considerations.
Insurance coverage influences access; DIFICID's high price—around $3,500 to $4,000 for a typical course—limits adoption, especially where payers favor older, cheaper drugs.
Recent trends include increased use in long-term care and hospital settings, with rising awareness of its benefits in preventing recurrence. The COVID-19 pandemic did not significantly alter CDI epidemiology but affected treatment patterns and hospital resource allocation.
How Is the Price of DIFICID Evolving?
DIFICID's list price, approximately $3,500 to $4,000 per course, has remained relatively stable over recent years. Price increases have been modest, often driven by inflation or adjustments for market conditions, but the absolute cost remains high relative to generic alternatives like vancomycin ($50-$100 per course).
The company has offered patient assistance programs and negotiated discounts to improve access, but no significant official rebate or price cuts have been publicly announced.
Price sensitivity among payers and providers persists, constraining broader use. Competitive pressure from generic vancomycin and emerging antibiotics continues to limit revenue growth.
Price Comparisons with Alternatives
| Drug | Typical Course Cost | Regulatory Status | Recurrent CDI Efficacy | Main Limitations |
|---|---|---|---|---|
| Fidaxomicin | $3,500-$4,000 | Prescription-only | Reduced recurrence (25%) | High cost, limited access |
| Vancomycin | $50-$100 | Generic | Higher recurrence (30-35%) | Less targeted, more resistance risk |
| Metronidazole | <$50 | Generic | Higher recurrence (35-40%) | Less effective for severe CDI |
What Are Future Price and Market Outlooks?
Projected market growth for DIFICID hinges on several factors:
- Clinical guidelines: Will favor its use in high-risk patients, expanding markets.
- Pricing negotiations: Payers and pharmacy benefit managers (PBMs) may secure discounts or formulary restrictions.
- New competitors: Emerging drugs may erode DIFICID's market share, especially if they achieve approval with better efficacy or lower prices.
- Patent status: Merck's expiry or expirations of exclusivity in various jurisdictions could introduce generics, dramatically reducing prices.
The overall CDI market is expected to grow annually at approximately 4-6% over the next five years, driven by aging populations and increased healthcare-associated infections.
Forecasts suggest the following for DIFICID:
- Sales will remain concentrated in hospital and long-term care settings.
- Price stabilization is likely absent; downward pressure from competition and generics is anticipated.
- Volume growth may be moderate, limited by clinical guidelines and payer restrictions.
Quantitative Projections (Assuming No Major Patent Erosion)
| Year | Estimated Market Size (USD) | DIFICID Market Share | Predicted Revenue (USD) |
|---|---|---|---|
| 2023 | 600 million | 20% | 120 million |
| 2025 | 700 million | 25% | 175 million |
| 2027 | 800 million | 25% | 200 million |
Decreased approval or reimbursement constraints could lower these projections. Conversely, if new indications or expanded use emerges, revenue could surpass estimates.
What Key Regulatory and Competitive Factors Impact Pricing?
Regulatory decisions influence pricing strategies. For example, if the FDA approves new formulations or expanded indications, price adjustments may follow. Conversely, approval of next-generation competitors with lower costs can lead to price erosion.
Patent protections provide exclusivity, but expiration deadlines threaten imminent generic entry:
- Patents originally filed in 2008 expire between 2023 and 2025 in major markets, risking generic erosion.
- Patent challenges or legal disputes could accelerate or delay generic approval.
Manufacturers may respond with tiered pricing, discounts, or new formulations to sustain revenues.
What Are the Main Takeaways?
- DIFICID is a niche drug with high efficacy for preventing CDI recurrence.
- The current price remains high at roughly $3,500-$4,000 per course.
- Market growth is constrained by cost, competition, and clinical practice guidelines.
- Patent expiration threatens price stability; generics are likely to enter in the next 1-3 years.
- Revenue projections suggest moderate growth, with several variables—including regulatory decisions and competitive dynamics—potentially significantly altering forecasts.
FAQs
1. When will generic versions of fidaxomicin likely enter the market?
Generic entry is expected between 2023 and 2025, aligning with patent expirations in major markets.
2. How does DIFICID compare to older CDI treatments in terms of cost-effectiveness?
While more expensive upfront, DIFICID reduces recurrence rates, potentially lowering overall treatment costs. However, high drug pricing remains a barrier to widespread adoption.
3. Are there ongoing efforts to reduce DIFICID’s price?
Yes. Merck and payers negotiate discounts and patient assistance programs to improve access, but no large-scale price reductions have been publicly announced.
4. How will new clinical guidelines impact DIFICID’s market share?
Guidelines favoring fidaxomicin for high-risk patients reinforce its niche. However, broader adoption may be limited by cost and availability of alternatives.
5. What is the outlook for DIFICID in emerging markets?
Lower income levels and cost sensitivities reduce adoption prospects. Pricing strategies and licensing agreements could influence penetration in these regions.
References
- CDC. "Clostridioides difficile infection (CDI): Surveillance and Statistics." (2022).
- Merck. "DIFICID (fidaxomicin) Prescribing Information." (2022).
- IQVIA. "Global Infectious Disease Therapeutics Market Report." (2022).
- FDA. "FDA Approval Documents for Fidaxomicin." (2011).
- EvaluatePharma. "Prescription Drug Market Trends." (2022).
More… ↓
